中国组织工程研究 ›› 2021, Vol. 25 ›› Issue (13): 2070-2078.doi: 10.3969/j.issn.2095-4344.2196
• 干细胞综述 stem cell review • 上一篇 下一篇
间充质干细胞在心脏移植免疫耐受诱导中的重要作用
毛 鑫1,余丽梅2,3,4,5,王 峰1
- 1遵义医科大学第二附属医院胸心外科,贵州省遵义市 563000;2遵义医科大学附属医院,贵州省遵义市 563003;3贵州省细胞工程重点实验室,贵州省遵义市 563003;4贵州省生物治疗人才基地,贵州省遵义市 563003;5遵义市干细胞与再生医学工程研究中心,贵州省遵义市 563003
-
收稿日期:2020-04-22修回日期:2020-04-25接受日期:2020-06-03出版日期:2021-05-08发布日期:2020-12-28 -
通讯作者:王峰,副教授,硕士,硕士研究生导师,遵义医科大学第二附属医院胸心外科,贵州省遵义市 563000 -
作者简介:毛鑫,男,1995年生,贵州省遵义市人,汉族,遵义医科大学在读硕士,主要从事胸心血管外科基础研究。 -
基金资助:国家自然科学基金(81660074),项目负责人:王峰
Important role of mesenchymal stem cells in immune tolerance induction in heart transplantation
Mao Xin1, Yu Limei2, 3, 4, 5, Wang Feng1
- Department of Cardiothoracic Surgery, Second Affiliated Hospital of
-
Received:2020-04-22Revised:2020-04-25Accepted:2020-06-03Online:2021-05-08Published:2020-12-28 -
Contact:Wang Feng, Associate professor, Master, Master’s supervisor, Department of Cardiothoracic Surgery, Second Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Mao Xin, Master candidate, Department of Cardiothoracic Surgery, Second Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81660074 (to WF)
摘要:

文题释义:
心脏移植免疫耐受:心脏移植后,受者对供者来源的MHC抗原能产生细胞和体液免疫排斥反应,一般以细胞免疫为主。通过不同方式对宿主免疫系统识别同种异体抗原进行干扰,从而达到宿主对移植物异型抗原产生特异性免疫无反应(免疫耐受),达到移植物长期存活。
间充质干细胞来源的胞外囊泡:主要包括外泌体和微囊泡,它们包裹着大量的生物活性因子,包括蛋白质(细胞因子、膜受体、生长因子和酶)和遗传物质(mRNA和microRNAs)。间充质干细胞来源胞外囊泡是间充质干细胞发挥免疫调节和组织再生功能的主要生物因子,在心脏移植免疫耐受诱导过程中也发挥重要作用。
背景:心脏移植是终末期心脏疾病的重要治疗方式,移植后的维持离不开免疫抑制药物的使用。鉴于免疫抑制药物的不良反应较大,甚至可能对宿主生活质量产生巨大影响,人们开始探寻新的诱导移植耐受的方法,来尽量规避免疫抑制药物的使用。间充质干细胞的免疫调节特性受到了研究人员的关注,并开展了大量的相关研究。
目的:总结并探讨间充质干细胞的免疫调节特点和心脏移植免疫耐受诱导方面的问题,以及它们之间的关系。
方法:检索PubMed数据库中相关文献,时间不限。检索词包括“stem cell; mesenchymal stem cells;MSC;MSCs;heart transplantation;cardiac allograft;heart allograft;immune tolerance;tolerance;immunological rejection”。筛选排除重复研究,对相关研究文献进行整理总结并综述。
结果与结论:一方面,间充质干细胞对免疫系统各细胞有着不同的作用;另一方面,间充质干细胞调控或直接产生的众多生物活性因子构成了复杂的网络,创造了一个利于免疫调节的微环境。间充质干细胞在心脏移植中的应用多局限在动物实验,且结果都相对乐观。研究人员对心脏移植免疫耐受诱导的探究,有助于人们对该领域的进一步了解,并给相关临床工作更多的启示。
https://orcid.org/0000-0002-3556-3933(毛鑫)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
毛 鑫, 余丽梅, 王 峰. 间充质干细胞在心脏移植免疫耐受诱导中的重要作用[J]. 中国组织工程研究, 2021, 25(13): 2070-2078.
Mao Xin, Yu Limei, Wang Feng. Important role of mesenchymal stem cells in immune tolerance induction in heart transplantation[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2070-2078.
2.1.1 T细胞 间充质干细胞可以启动同种异体反应性T细胞的激活,但不能引起T细胞的增殖反应[5]。在间充质干细胞的刺激下,T细胞被阻滞在G1期;在分子水平上,细胞周期蛋白D2的表达被明显抑制,而p27(Kip1)的表达则被上调[6]。研究表明,间充质干细胞通过诱导活化的T细胞凋亡来抑制T细胞的增殖,但对静止的T细胞没有影响。这种凋亡可能与间充质干细胞在干扰素γ存在下表达的吲哚胺2,3-双加氧酶(IDO)将色氨酸转化为犬尿氨酸有关[7]。吲哚胺2,3-双加氧酶介导的色氨酸分解代谢是人间充质干细胞中一种新的T细胞抑制效应机制[8]。研究证明,间充质干细胞不仅可以抑制白细胞介素2诱导的细胞增殖,还可以抑制细胞裂解活性和细胞因子的产生,而这种抑制主要是可溶性因子吲哚胺2,3-双加氧酶和前列腺素E2的协同作用[9]。B7-H1(程序性死亡配体1)是B7家族分子中的新成员,B7-H1与其受体PD-1的结合可以在体外抑制T细胞的增殖和某些细胞因子的分泌。有研究发现,间充质干细胞的免疫抑制能力也与此途径有关。人脐带来源间充质干细胞结构性表达T细胞激活的负调控因子B7-H1,干扰素γ处理后其表达增加;同时,干扰素γ还诱导人脐带来源间充质干细胞表达吲哚胺2,3-双加氧酶和HLA-DR。这使得人脐带来源间充质干细胞具有T细胞免疫抑制活性[10]。此外,间充质干细胞治疗可能使免疫平衡从Th1细胞(主要产生白细胞介素2和干扰素γ)向Th2细胞(主要产生白细胞介素4和白细胞介素10)转移[11],这对免疫耐受的产生具有重要意义。
调节性T细胞(Treg细胞)是免疫调节机制的新兴关键角色,它是T辅助细胞的亚群,专门抑制T细胞介导的免疫反应,并特异性地表达FoxP3转录因子[12]。同种异体移植物免疫耐受的诱导与Treg细胞比例增加关系密切[13]。CD4+CD25+FoxP3+Treg细胞亚群具有免疫调节功能,能够抑制抗原特异性的T细胞反应,并利于诱导和维持移植物特异性免疫耐受。间充质干细胞分泌白细胞介素10依赖的可溶性HLA-G5亚型,可以促进间充质干细胞诱导Treg细胞生成。中和抗HLA-G5抗体的阻断实验表明,HLA-G5首先抑制异基因T细胞的增殖,然后促进CD4+CD25+FoxP3+Treg细胞的扩增[14]。
此外,间充质干细胞诱导的树突状细胞(MSCs-DC)吞噬能力强,免疫原性低,免疫调节功能强,它能从CD4+CD25-Foxp3-T细胞中高效地产生CD4+CD25+Foxp3+Treg细胞[15]。间充质干细胞能够增强CD8+CD28-Treg细胞的调节功能,具体表现在增加CD8+CD28-T细胞的比例,增强CD8+CD28-T细胞抑制初始CD4+T细胞增殖和活化的能力,减少活化的CD4+T细胞产生干扰素γ,诱导活化的CD4+T细胞凋亡[16]。
2.1.2 树突状细胞 间充质干细胞通过接触诱导具有T细胞抑制特性的调节性抗原提呈细胞(APC)来间接抑制T细胞。不同的APC群体的发育受到组织细胞微环境的调节。间充质干细胞与MLR体系共培养分析显示,干扰素γ和白细胞介素10的表达上调,肿瘤坏死因子α的表达下调[5,17]。树突状细胞是启动适应性免疫反应的最有效的抗原提呈细胞,它能介导包括针对同种异体细胞或自身抗原的免疫应答。因此,通过增强其吞噬能力和抑制其刺激淋巴细胞增殖的能力来调节其功能特性,降低或调节免疫和炎症反应,这在逻辑上是可行的。另外,间充质干细胞被证明可干扰单核细胞或CD34+造血前体细胞的树突状细胞成熟[18-20]。间充质干细胞抑制单核细胞分化及产生树突状细胞的关键介质可能是前列腺素E2[21-22]。间充质干细胞诱导的树突状细胞的抑制功能可通过转化生长因子β1介导,而且还通过诱导Treg细胞或T细胞无能的产生而发挥作用[15]。诱导多能干细胞是一种无限制、无侵袭性的间充质干细胞来源,可通过分泌因子白细胞介素10和直接接触细胞来抑制人单核细胞来源树突状细胞的分化[19]。也有研究认为间充质干细胞可通过改变细胞骨架组织来干扰激活过程,进而调节树突状细胞功能;经间充质干细胞处理的树突状细胞不能形成活跃的免疫突触;不能有效参与T细胞的免疫突触的形成[23]。此外,间充质干细胞的培养上清液也可干扰树突状细胞的内吞作用,降低树突状细胞分泌白细胞介素12和激活同种异体反应性T细胞的能力,这似乎也进一步证实了间充质干细胞分泌因子的重要作
用[24]。
2.1.3 B细胞 研究发现人间充质干细胞通过产生可溶性因子阻滞细胞周期的G0/G1期来抑制B细胞的增殖,抑制B细胞的分化和趋化细胞因子的产生来抑制B淋巴细胞相关的体液免疫反应。众所周知,IgG作为一种由浆细胞分泌的免疫球蛋白,在活化的B细胞培养过程中加入间充质干细胞可以抑制IgG的产生[25]。通过间充质干细胞输注增加CD5+调节性B细胞(Breg)产生白细胞介素10,可改善难治性慢性移植物抗宿主病。同时,间充质干细胞可以促进CD5+调节性B细胞的存活和增殖,吲哚胺2,3-双加氧酶在其中发挥了重要作用[26]。
2.1.4 巨噬细胞 间充质干细胞通过转化生长因子β1募集巨噬细胞来减轻损伤部位的病理改变[27]。间充质干细胞的吲哚胺2,3-双加氧酶活性与单核细胞向分泌白细胞介素10的M2免疫抑制巨噬细胞(CD14+/CD206+)分化有关。这些单核细胞来源的M2反过来以白细胞介素10非依赖的方式参与T细胞增殖的抑制,从而放大了间充质干细胞产生的免疫抑制效应。同时,间充质干细胞与CD14+单核细胞共培养可诱导CD206上调和单核细胞分泌白细胞介素10[28]。骨髓来源的巨噬细胞与骨髓间充质干细胞共培养后巨噬细胞表型由M1向M2转变,表现为白细胞介素6、白细胞介素1β、单核细胞趋化蛋白1、诱导型一氧化氮合成酶等M1标志物明显降低,而M2标志物白细胞介素10、白细胞介素4、CD206和Arg1明显升高[29]。
2.1.5 微环境 间充质干细胞调控或直接产生的生物活性因子(如免疫抑制因子、生长因子、趋化因子、表面黏附分子等)构成了一个复杂的网络,并创造了一个利于细胞稳定、再生及免疫调节的微环境。在对间充质干细胞的了解过程中发现,间充质干细胞免疫调节特性需要在促炎环境中激活,其功能是与微环境相互作用的结果。已有研究表明白细胞介素1、干扰素γ和肿瘤坏死因子α参与间充质干细胞的启动[2,30]。
间充质干细胞释放白细胞介素10支持调节性T细胞的分化,还能抑制促炎细胞因子的产生(如干扰素γ)和抑制抗原递呈细胞的功能[31-32],其中抑制因子的释放在使用肿瘤坏死因子α和干扰素γ刺激后得到增强[12]。另外,干扰素γ联合肿瘤坏死因子α预处理骨髓间充质干细胞对T细胞增殖的抑制作用较未处理时增强[33],这可能与干扰素介导的吲哚胺2,3-双加氧酶上调有关[28]。另外,表面黏附分子的表达,如血管细胞黏附分子1、细胞间黏附分子1等,也参与间充质干细胞与免疫细胞的相互作用[34]。干扰素γ联合促炎细胞因子肿瘤坏死因子α、白细胞介素1α或白细胞介素1β可以刺激间充质干细胞诱发非常高水平的免疫抑制因子(如诱导型一氧化氮合酶)、趋化因子以及黏附分子的大量表达,它们的协同作用导致了免疫细胞的聚集,制造了一种微环境,在这种微环境中间充质干细胞产生的局部作用因子的作用被放大(T细胞可被诱导型一氧化氮合酶产生的一氧化氮抑制),各因子的协同作用产生了强大的免疫抑制效果[35-36]。
学者们发现间充质干细胞抑制免疫细胞活动的能力要通过炎症环境来实现。炎症平衡是许多疾病的一个关键点,必须在对抗微生物感染、控制过度炎症及其有害影响之间取得平衡。研究表明间充质干细胞能够重新校准炎症效应T细胞和抗炎Treg细胞之间的平衡,这一过程与单核细胞向抗炎(M2)巨噬细胞的极化密切相关[12],而且间充质干细胞很容易被高浓度的炎症递质吸引到组织损伤部位。研究发现,当受到炎症环境信号刺激时,其释放的抑制免疫反应分子使其逃避免疫监视,容易迁移到损伤部位[2]。同时,骨髓间充质干细胞可能通过其衍生的白细胞介素6保护储存池中的中性粒细胞免于凋亡,保护其效应功能[37]。
2.1.6 体外共培养及细胞毒性T细胞 适应性免疫应答是体内抗原特异性淋巴细胞接受抗原刺激后,产生的一系列生物学效应。CD4+辅助T淋巴细胞和CD8+细胞毒性T淋巴细胞可在抗原处理和抗原呈递细胞呈递抗原后,提供特定的抗原特异性免疫反应[12]。许多研究探讨了间充质干细胞对免疫细胞的抑制作用。外周血单核细胞与间充质干细胞共培养时可产生强大的调节性CD4+或CD8+淋巴细胞,这些调节细胞可能进一步放大了间充质干细胞介导的免疫抑制效应[38]。另外,间充质干细胞能抑制和刺激混合淋巴细胞培养和有丝分裂反应,不依赖主要组织相容性复合体,这似乎更多地依赖于细胞剂量,可能是刺激机制的“过载”造成的[39]。间充质干细胞能抑制混合淋巴细胞培养中细胞毒性T细胞的形成,这种T细胞抑制与其产生的可溶性因子有关[40-41]。而间充质干细胞在混合淋巴细胞培养中早期添加才会表现出抑制细胞毒性的能力。虽然,间充质干细胞不是细胞毒性T细胞或NK细胞杀伤的靶点,但研究发现白细胞介素2激活的NK细胞可以有效杀伤异体和自体间充质干细胞,原因也许在于间充质干细胞低水平表达HLA-Ⅰ类抗原,同时也表达几种激活NK细胞受体识别的配体[5,9]。另外,间充质干细胞可以通过细胞间的接触和可溶性因子介导两种不同方式改变NK细胞的表型,并抑制增殖、细胞因子分泌和对HLA-Ⅰ类表达靶点的细胞毒作用[42]。吲哚胺2,3-双加氧酶和前列腺素E2则是间充质干细胞诱导NK细胞抑制的关键介质。
2.1.7 旁分泌(胞外囊泡) 如今,人们也注意到旁分泌信号调节免疫反应的地位。人们发现RAP1缺乏引起的旁分泌功能障碍损害了同种异体心脏移植排斥反应中间充质干细胞的免疫抑制能力[43]。间充质干细胞来源的胞外囊泡作为间充质干细胞发挥免疫调节和组织再生功能的主要生物因子,也得到了广泛关注。间充质干细胞来源的胞外囊泡(主要包括外泌体和微囊泡)包裹着大量的生物活性因子,包括蛋白质(细胞因子、膜受体、生长因子和酶)和遗传物质(mRNA和microRNAs)[44]。研究表明,间充质干细胞来源外泌体在免疫调节中起重要作用,能抑制免疫细胞的增殖和调节B细胞的功能[45]。
由此可见,间充质干细胞的作用广泛,对免疫系统中多种细胞,包括T细胞、B细胞、树突状细胞、NK细胞和巨噬细胞等均有不同作用。间充质干细胞联合各因子所构成的微环境,在免疫调节方面也产生了巨大的影响。不难看出,间充质干细胞在免疫系统中的应用潜力较大,希望在未来的努力中能将其变成治疗相关疾病的有利工具。
2.2 间充质干细胞移植改善心脏移植预后 虽然干细胞在理论上具有良好的应用前景,但在临床上的应用情况并不乐观,除了造血干细胞在血液学上的应用外,目前其他类型干细胞的临床结果并不理想。干细胞在心脏移植中的应用更多局限于动物实验。经过各种途径将供体的抗原转移到受体内,对受体的免疫细胞采取各种方式的预处理,以便减低受体免疫系统对外来抗原的辨识能力,是诱导产生免疫耐受的发展方向。心脏移植免疫耐受诱导情况见图1。通过对间充质干细胞在大鼠心脏移植模型中免疫抑制作用行蛋白质组学分析,发现供体来源的骨髓间充质干细胞明显减轻了大鼠心脏移植后的急性排斥反应,炎性细胞浸润明显减轻,移植物存活时间明显延长[46]。
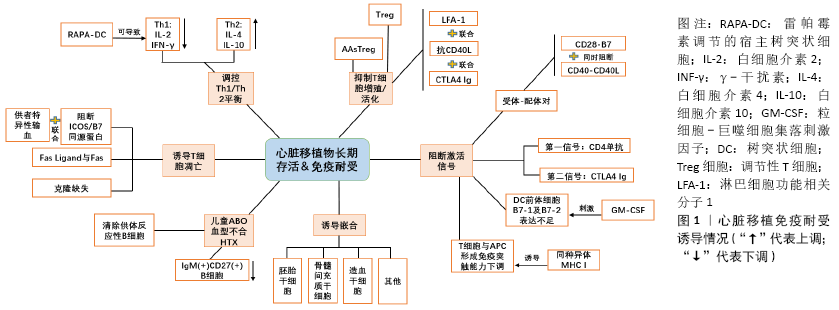
在相关的异位心脏移植模型中,供者来源的间充质干细胞诱导耐受的诱导期(移植前)和维持期(移植后>100 d)均与供者特异性CD4+CD25+Foxp3+Treg细胞扩增和抗供者Th1活性降低有关。另外,间充质干细胞的免疫调节特性需要去除造血干细胞,需在供者和受者之间部分共享MHCs-Ags,并在移植前输注[47]。骨髓间充质干细胞在体外和体内都能抑制同种异体T细胞反应。静脉注射间充质干细胞可延长移植心脏存活时间,可能是通过改变Th1/Th2平衡向Th2偏移来诱导同种异体移植耐受[48]。另外,骨髓间充质干细胞与骨髓来源树突状细胞共培养后上调了树突状细胞的CD45RB表达,通过鼠尾静脉注射这种CD45RBB+树突状细胞可增强T细胞的免疫抑制能力,增强同种异体大鼠心脏移植的免疫耐受
性[49]。
白细胞介素35是新近发现的一种细胞因子,通过增强调节性T细胞的功能,抑制Th1和Th17的增殖和功能,发挥强大的免疫抑制作用。过表达白细胞介素35基因的脂肪间充质干细胞(IL-35-MSCs)注射到同种异位腹部心脏移植模型小鼠体内,结果发现这种细胞在体内、外均能持续分泌白细胞介素35,成功缓解同种异体移植排斥反应,延长移植物存活时间。此外,过表达白细胞介素35基因的脂肪间充质干细胞在心脏移植小鼠中表现出更强的免疫抑制能力,进一步降低了Th17细胞的比例,增加了CD4Foxp3 T细胞的比例,调节了Th1/Th2的平衡,提示过表达白细胞介素35基因的脂肪间充质干细胞在抑制移植排斥反应方面更具优势,可能为诱导移植免疫耐受提供新的途径[50]。
B7-H1是B7家族分子的成员,具有免疫抑制特性。WANG等[51]用抗B7-H1单抗阻断间充质干细胞表达B7-H1,发现其扰乱了基于间充质干细胞治疗产生的心脏移植排斥反应防御,这与耐受性树突状细胞、Treg细胞和Breg细胞的增长不足以及抗原刺激的T细胞增殖的恢复、抗供体抗体产生和B细胞增殖有关,也证实了间充质干细胞的免疫耐受诱导功能部分依赖于B7-H1的表达。同时,研究还发现间充质干细胞联合雷帕霉素可成功诱导心脏移植的免疫耐受,然而这种耐受也可被抗B7-H1单抗阻断。另外,失去代谢和分泌活性的间充质干细胞对移植物存活率无明显影响,这可能进一步说明抑制T细胞介导的同种异体移植排斥反应需要具备完整功能的间充质干细胞[52]。
有研究发现单纯的间充质干细胞治疗对异位心脏移植物的存活率没有显著影响。然而间充质干细胞与相关免疫抑制药物的联合应用却产生了不错的效果,间充质干细胞联合短期霉酚酸酯可显著延长移植物的存活时间[53]。间充质干细胞与亚治疗剂量的雷帕霉素联合使用时,减弱同种异体免疫反应,促进心脏移植耐受[54]。
外泌体近年来受到了广泛的关注,间充质干细胞来源外泌体在介导间充质干细胞的生物学功能中起着重要作用。此外,外泌体被证实在细胞动力学中具有细胞间通讯工具的功能。外泌体是细胞内信号传导的重要介质,控制着靶细胞的功能行为。近期有学者提出猜想,表达miR-126-5p和
miR-126-5p的间充质干细胞外泌体可能在预防异基因心脏移植排斥反应方面具有潜力,并延长移植物存活时间,其机制可能与沉默高迁移率族蛋白1(HMGB1)信号通路有关,这种蛋白可以激活炎症反应,最终导致心脏移植后的排斥反应[55]。吲哚胺2,3-双加氧酶在骨髓间充质干细胞中的表达被认为与间充质干细胞介导的免疫抑制有关。为此,有人采用慢病毒转基因系统获得稳定表达吲哚胺2,3-双加氧酶的骨髓间充质干细胞(IDO-BMSCs),并进一步获取该细胞来源的外泌体,将其注射到心脏移植大鼠体内,发现移植物免疫排斥反应降低,心脏移植物功能改善。随后进一步的研究发现该细胞本身也可通过增加树突状细胞和Treg细胞的产生和活性来抑制心脏移植排斥反应[56-57]。
胸腺内注射/静脉注射间充质干细胞可延长大鼠同种异体心脏移植物存活时间,其机制可能与下调miR-155表达、改变Th1/Th2平衡、上调Treg细胞表达有关。他们的研究也发现,胸腺联合静脉注射间充质干细胞是提高CD4+、CD25+、Foxp3+T细胞百分率的最佳途径。另外还导致白细胞介素2和干扰素γ水平降低,白细胞介素4和白细胞介素10水平升高[58]。
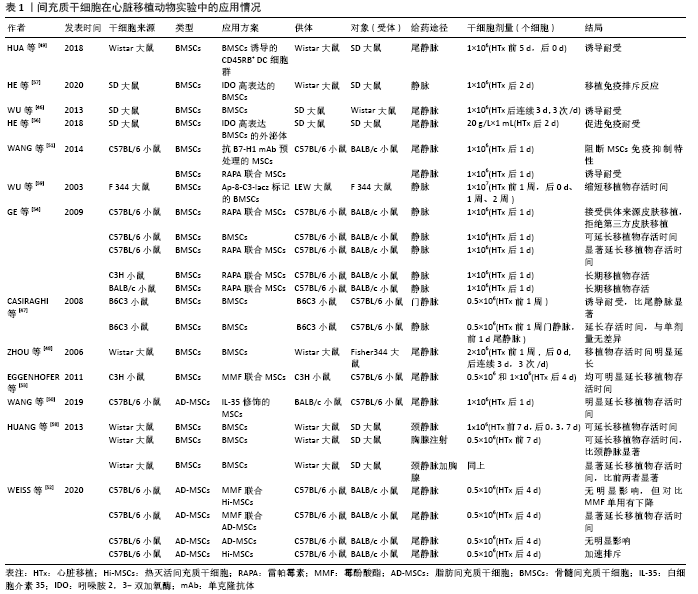
然而,也并不是所有研究都得到了支持移植物存活的结果。WU等[59]发现间充质干细胞参与了同种异体移植物的组织修复,它在排斥反应损伤后的器官移植组织修复过程中导致了移植物纤维化。骨髓间充质干细胞在同种异体移植排斥反应部位有较强的迁移能力,在慢性排斥反应期间积极参与组织修复。而间充质干细胞较强的向成纤维细胞迁移的能力,也抑制了其向肌细胞分化。间充质干细胞在心脏移植动物模型中的应用情况见表1。
2.3 心脏移植免疫耐受诱导的探索 心脏移植受体对MHC抗原能产生细胞和体液免疫排斥反应,一般以细胞免疫为主。学者们对移植心脏MHC抗原被宿主免疫系统识别引发排斥的机制及对策作了大量的研究,期望能诱导宿主对移植物的异型抗原产生特异性免疫无反应(免疫耐受),这样不仅能保证移植器官的长期存活,还可以保证移植后患者免疫系统的正常功能,避免免疫功能低下导致的不良后果。
2.3.1 心脏移植免疫耐受与免疫细胞凋亡 在如今的研究中,人们进一步认识了移植免疫耐受与免疫细胞凋亡之间关系。在细胞凋亡与心脏等器官移植耐受的作用机制中,“否决现象”曾被学者提出[60],即一种含同种异体抗原的细胞,在其活动的过程中,使识别它们的T细胞失活,使具有自身反应性的细胞克隆经凋亡而清除。另外,随着Fas Ligand作为一种保护机制被发现时,人们对免疫赦免的概念也有了新的认识。Fas Ligand存在赫免区并与进入该区的活化淋巴细胞上表达的Fas抗原结合,导致淋巴细胞凋亡,从而使得在异基因移植时保护组织免受免疫攻击[61]。这些观点在器官移植免疫耐受的研究中有着深远意义。有研究发现,供体特异性输血预处理联合诱导共刺激因子(ICOS)/B7同源蛋白(B7h)同种异体识别受损可促进受体引流淋巴结CD8+T细胞凋亡,并参与同种异体心脏移植的无反应性[62]。
免疫系统可通过删除自身反应性淋巴细胞来维持自身的耐受性,而对于T细胞来说,这一步骤发生在胸腺,在那里发育的T细胞首先表达它们的抗原特异性受体。胸腺中成熟的胸腺细胞在遇到树突状细胞呈递的自身或异体抗原时,会发生克隆性缺失/凋亡。树突状细胞来源的一氧化氮也许在其中发挥重要作用[63]。在大鼠中,胸腺内注射同种异体抗原可诱导同种异体心脏移植耐受。诱导胸腺内耐受的细胞和分子机制尚不清楚,其主要机制可能是通过程序性细胞死亡或凋亡的过程消除反应性T细胞,即克隆缺失[64]。
2.3.2 心脏移植免疫耐受与T细胞 T细胞在促进机体产生免疫耐受的过程中起着重要作用。Treg细胞作为T细胞的亚群,通过主动抑制自身反应性T细胞的活化和扩增来维持外周的自我耐受性,且有多个Treg细胞种群参与维持外周耐受性。然而对于同种异体移植物存活率与循环中Treg细胞数量的关系,学者们得出了不同的结论[65-66]。有研究表明,Treg细胞可与未成熟的供体来源树突状细胞共培养来选择有效的同种抗原特异性Treg细胞。学者认为同种抗原特异性Treg细胞具有高度抑制功能,如抑制了T细胞的增殖。配合保护其功能的干预(如暂时性的雷帕霉素靶蛋白抑制)可促进心脏移植长期存活[67]。
1986年,MOSMANN等[68]发现CD4+ T辅助细胞可产生Th1型和Th2型细胞因子,并探讨了两种因子相互抑制的状态。Th2型细胞因子在心脏移植免疫耐受的诱导和维持中也发挥着重要作用。有研究表明,骨髓间充质干细胞能抑制同种异体T细胞反应。静脉注射间充质干细胞可延长移植心脏存活时间,而其可能的机制是通过改变Th1/Th2平衡来实现的,实时定量PCR也显示Th1/Th2平衡向Th2偏移[48]。大量的研究似乎都认为免疫耐受诱导依赖于CD4+T细胞向Th2型细胞(白细胞介素4、白细胞介素10)倾斜和偏离Th1型细胞(白细胞介素2、干扰素γ)。但这种依赖于Th1/Th2平衡的免疫耐受诱导也存在争议。该理论在器官移植中是否一定对器官长期存活有益,这是否是维持耐受性所必须的,曾有学者对此持怀疑态度[69-70]。
2.3.3 阻断激活信号 有学者研究表示,受体-配体对CD28-B7和CD40-gp39对于启动和放大T细胞依赖的免疫应答是必不可少的。CD28-B7相互作用提供最佳T细胞激活和白细胞介素2产生所需的“第二信号”,而CD40-gp39信号共同刺激B细胞、巨噬细胞、内皮细胞和T细胞激活,但这两条通路需要同时阻断,才能有效中止T细胞在体外和体内的克隆性增殖,抑制原发血管化心脏移植物的慢性血管排斥反应的发展[71]。CD4单抗(mAb)诱导的活化信号1或CTLA4Ig介导的共刺激信号2的阻断可诱导致敏大鼠心脏移植耐受[69]。研究表明,在粒细胞-巨噬细胞集落刺激因子刺激下,细胞表面共刺激分子B7-1(CD80)和B7-2(CD86)表达不足的小鼠骨髓来源MHCⅡ+树突状细胞前体细胞在体外可以诱导同种异体抗原特异性T细胞无能。而这些共刺激分子缺陷的树突状细胞前体细胞(MHCⅡ+、CD80dim、CD86-)可延长非免疫抑制受体的同种异体心脏移植存活时间[72]。由此可见,激活信号的阻断在心脏移植耐受诱导中具有着重要作用。
2.3.4 心脏移植免疫耐受与树突状细胞 除了抗原和淋巴细胞,抗原提呈树突状细胞亦是免疫应答的关键。树突状细胞在诱导免疫耐受以及调节T细胞介导的免疫应答类型中可能起重要作用[73]。树突状细胞所介导的免疫反应在延长心脏移植存活方面具有重要意义[74],通过对树突状细胞的预处理,能达到延长移植物存活的效果。有研究发现,雷帕霉素调节的宿主树突状细胞在移植前与供体抗原调制并给予宿主,移植术后再进行短暂的雷帕霉素治疗,可以无限期延长心脏移植的存活时间。这与Treg细胞对移植物的浸润以及移植物血管病变的消失有关[75]。未成熟状态的树突状细胞致耐受特性较强,药物操作是一种前景良好且具备临床适用性的未成熟树突状细胞产出方法。研究发现,亲免疫配体雷帕霉素可以抑制树突状细胞的成熟及其效应功能。经雷帕霉素处理的同种异体抗原致敏的树突状细胞诱导T细胞产生白细胞介素2和干扰素γ的能力降低,并减弱随后的攻击反应。静脉注射这些雷帕霉素处理的树突状细胞能显著延长同种异体抗原特异性心脏移植物的存活时间[76]。此外,表现为未成熟表型的骨髓来源树突状细胞群体不仅能显著延长同种异体心脏移植物的存活时间,还能降低抗供者的体液和细胞免疫反应[77]。随后他们的研究进一步说明,供者特异性同种异体移植耐受是通过给予受体来源的未成熟树突状细胞和次优免疫抑制来实现的[78]。不仅系统注射供者来源的树突状细胞前体细胞可以延长非免疫抑制宿主的同种异体心脏移植存活时间,而且局部注射粒细胞-巨噬细胞集落刺激因子也可诱导移植耐受,通过将树突状细胞直接输送到心肌来诱导对同种异体心脏移植物的耐受性,从而降低全身不良反应的可能性,这可能比全身注射树突状细胞更有效[79]。
2.3.5 嵌合现象的诱导 嵌合现象是接受异体或异种移植物一段时间后,供、受体细胞相互移行、相互存在的现象,稳定的嵌合与供体器官或组织移植的永久耐受有关。嵌合细胞包括树突状细胞、T细胞、B细胞及巨噬细胞,其中树突状细胞的作用尤其重要。间充质干细胞在诱导嵌合体形成方面也有着良好的效果。有研究者用胚胎干细胞建立嵌合体,成功诱导了心脏移植免疫耐受。同时,造血干细胞及骨髓间充质干细胞亦可建立心脏嵌合,且前者较后者更容易。骨髓间充质干细胞在心脏移植的混合嵌合体发展过程中也起着关键的作用[80-81]。
2.3.6 儿童ABO血型不合的心脏移植 儿童相较成人在心脏移植后表现出更好的存活率,并且可以安全地接受ABO血型不合的捐赠者的心脏移植。儿童对供者ABH抗原产生免疫耐受,表现为特异性抗体的持续缺乏。对这一机制进行研究也许能帮助找到稳定诱导免疫耐受的方法。机体对ABH抗原的应答是由IgM+CD27+B细胞介导的,而儿童ABO血型不合移植供者ABO特异性耐受是由于这些细胞的低比重所促进
的[82]。另外有学者将ABO血型不合的婴儿心脏移植成功的机制描述为“外源细胞的主动获得性耐受”,产生耐受性需消除供体反应性B淋巴细胞,并且可能依赖于某种程度的抗原表达的持续性[83]。
2.3.7 其他 对于传统的药物治疗,有学者探讨他克莫司和环孢素A的免疫抑制疗法对心脏移植受者树突状细胞和排斥反应发生的影响,他克莫司治疗的患者浆细胞样树突状细胞值较高,而高浆细胞样树突状细胞的心脏移植患者发生排斥反应的风险较低[84]。此外, CTLA-4免疫球蛋白、抗CD40L和抗LFA-1联合治疗接受完全MHC不匹配、带血管蒂心脏移植的小鼠,可部分抑制T细胞增殖,从而限制移植物反应性T细胞的数量,使其产生耐受状态。同时,移植物内的
Foxp3+CD4+T细胞的积聚可局部抑制移植物的反应活性,防止排斥反应的发生[85]。同种异体移植排斥反应中的免疫反应涉及一系列分子事件,导致T细胞和抗原提呈细胞之间形成免疫突触。同种异体MHC-Ⅰ诱导心脏移植耐受过程中T细胞极性和运动性相关基因下调,即它们的移动、识别和与抗原提呈细胞形成功能性免疫突触的能力[86]。
| [1] KRAMPERA M, GALIPEAU J, SHI Y, et al. Immunological characterization of multipotent mesenchymal stromal cells--The International Society for Cellular Therapy (ISCT) working proposal. Cytotherapy. 2013;15(9): 1054-1061. [2] QUARANTA P, FOCOSI D, FREER G, et al. Tweaking Mesenchymal Stem/Progenitor Cell Immunomodulatory Properties with Viral Vectors Delivering Cytokines. Stem Cells Dev. 2016;25(18):1321-1341. [3] VOISIN C, CAUCHOIS G, REPPEL L, et al. Are the Immune Properties of Mesenchymal Stem Cells from Wharton’s Jelly Maintained during Chondrogenic Differentiation? J Clin Med. 2020;9(2):423. [4] JIANG X, LIU C, HAO J, et al. CD4(+)CD25 (+) regulatory T cells are not required for mesenchymal stem cell function in fully MHC-mismatched mouse cardiac transplantation. Cell Tissue Res. 2014;358(2):503-514. [5] KLYUSHNENKOVA E, MOSCA JD, ZERNETKINA V, et al. T cell responses to allogeneic human mesenchymal stem cells: immunogenicity, tolerance, and suppression. J Biomed Sci. 2005;12(1):47-57. [6] GLENNIE S, SOEIRO I, DYSON PJ, et al. Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells. Blood. 2005;105(7): 2821-2827. [7] PLUMAS J, CHAPEROT L, RICHARD MJ, et al. Mesenchymal stem cells induce apoptosis of activated T cells. Leukemia. 2005;19(9):1597-1604. [8] MEISEL R, ZIBERT A, LARYEA M, et al. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood. 2004;103(12):4619-4621. [9] SPAGGIARI GM, CAPOBIANCO A, ABDELRAZIK H, et al. Mesenchymal stem cells inhibit natural killer-cell proliferation, cytotoxicity, and cytokine production: role of indoleamine 2,3-dioxygenase and prostaglandin E2. Blood. 2008;111(3):1327-1333. [10] TIPNIS S, VISWANATHAN C, MAJUMDAR AS. Immunosuppressive properties of human umbilical cord-derived mesenchymal stem cells: role of B7-H1 and IDO. Immunol Cell Biol. 2010;88(8):795-806. [11] ZHANG X, REN X, LI G, et al. Mesenchymal stem cells ameliorate experimental autoimmune uveoretinitis by comprehensive modulation of systemic autoimmunity. Invest Ophthalmol Vis Sci. 2011;52(6):3143-3152. [12] BERNARDO ME, FIBBE WE. Mesenchymal stromal cells: sensors and switchers of inflammation. Cell Stem Cell. 2013;13(4):392-402. [13] KAUNDAL U, BAGAI U, RAKHA A. Immunomodulatory plasticity of mesenchymal stem cells: a potential key to successful solid organ transplantation. J Transl Med. 2018;16(1):31. [14] SELMANI Z, NAJI A, ZIDI I, et al. Human leukocyte antigen-G5 secretion by human mesenchymal stem cells is required to suppress T lymphocyte and natural killer function and to induce CD4+CD25highFOXP3+ regulatory T cells. Stem Cells. 2008;26(1):212-222. [15] ZHAO ZG, XU W, SUN L, et al. Immunomodulatory function of regulatory dendritic cells induced by mesenchymal stem cells. Immunol Invest. 2012; 41(2):183-198. [16] LIU Q, ZHENG H, CHEN X, et al. Human mesenchymal stromal cells enhance the immunomodulatory function of CD8(+)CD28(-) regulatory T cells. Cell Mol Immunol. 2015;12(6):708-718. [17] BEYTH S, BOROVSKY Z, MEVORACH D, et al. Human mesenchymal stem cells alter antigen-presenting cell maturation and induce T-cell unresponsiveness. Blood. 2005;105(5):2214-2219. [18] SPAGGIARI GM, MORETTA L. Interactions between mesenchymal stem cells and dendritic cells. Adv Biochem Eng Biotechnol. 2013;130:199-208. [19] GAO WX, SUN YQ, SHI J, et al. Effects of mesenchymal stem cells from human induced pluripotent stem cells on differentiation, maturation, and function of dendritic cells. Stem Cell Res Ther. 2017;8(1):48. [20] SAEIDI M, MASOUD A, SHAKIBA Y, et al. Immunomodulatory effects of human umbilical cord Wharton’s jelly-derived mesenchymal stem cells on differentiation, maturation and endocytosis of monocyte-derived dendritic cells. Iran J Allergy Asthma Immunol. 2013;12(1):37-49. [21] SPAGGIARI GM, ABDELRAZIK H, BECCHETTI F, et al. MSCs inhibit monocyte-derived DC maturation and function by selectively interfering with the generation of immature DCs: central role of MSC-derived prostaglandin E2. Blood. 2009;113(26):6576-6583. [22] JIANG XX, ZHANG Y, LIU B, et al. Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells. Blood. 2005;105(10):4120-4126. [23] ALDINUCCI A, RIZZETTO L, PIERI L, et al. Inhibition of immune synapse by altered dendritic cell actin distribution: a new pathway of mesenchymal stem cell immune regulation. J Immunol. 2010;185(9):5102-5110. [24] ZHANG W, GE W, LI C, et al. Effects of mesenchymal stem cells on differentiation, maturation, and function of human monocyte-derived dendritic cells. Stem Cells Dev. 2004;13(3):263-271. [25] CORCIONE A, BENVENUTO F, FERRETTI E, et al. Human mesenchymal stem cells modulate B-cell functions. Blood. 2006;107(1):367-372. [26] PENG Y, CHEN X, LIU Q, et al. Mesenchymal stromal cells infusions improve refractory chronic graft versus host disease through an increase of CD5+ regulatory B cells producing interleukin 10. Leukemia. 2015;29(3):636-646. [27] LIU W, ZHANG S, GU S, et al. Mesenchymal stem cells recruit macrophages to alleviate experimental colitis through TGFβ1. Cell Physiol Biochem. 2015; 35(3):858-865. [28] FRANÇOIS M, ROMIEU-MOUREZ R, LI M, et al. Human MSC suppression correlates with cytokine induction of indoleamine 2,3-dioxygenase and bystander M2 macrophage differentiation. Mol Ther. 2012;20(1):187-195. [29] CHO DI, KIM MR, JEONG HY, et al. Mesenchymal stem cells reciprocally regulate the M1/M2 balance in mouse bone marrow-derived macrophages. Exp Mol Med. 2014;46(1):e70. [30] LE BLANC K, MOUGIAKAKOS D. Multipotent mesenchymal stromal cells and the innate immune system. Nat Rev Immunol. 2012;12(5):383-396. [31] PATEL SA, SHERMAN L, MUNOZ J, et al. Immunological properties of mesenchymal stem cells and clinical implications. Arch Immunol Ther Exp (Warsz). 2008;56(1):1-8. [32] FONTAINE MJ, SHIH H, SCHÄFER R, et al. Unraveling the Mesenchymal Stromal Cells’ Paracrine Immunomodulatory Effects. Transfus Med Rev. 2016;30(1):37-43. [33] CUERQUIS J, ROMIEU-MOUREZ R, FRANÇOIS M, et al. Human mesenchymal stromal cells transiently increase cytokine production by activated T cells before suppressing T-cell proliferation: effect of interferon-γ and tumor necrosis factor-α stimulation. Cytotherapy. 2014;16(2):191-202. [34] NAJAR M, RAICEVIC G, ID BOUFKER H, et al. Modulated expression of adhesion molecules and galectin-1: role during mesenchymal stromal cell immunoregulatory functions. Exp Hematol. 2010;38(10):922-932. [35] REN G, ZHANG L, ZHAO X, et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell. 2008;2(2):141-150. [36] MA S, XIE N, LI W, et al. Immunobiology of mesenchymal stem cells. Cell Death Differ. 2014;21(2):216-225. [37] RAFFAGHELLO L, BIANCHI G, BERTOLOTTO M, et al. Human mesenchymal stem cells inhibit neutrophil apoptosis: a model for neutrophil preservation in the bone marrow niche. Stem Cells. 2008;26(1):151-162. [38] PREVOSTO C, ZANCOLLI M, CANEVALI P, et al. Generation of CD4+ or CD8+ regulatory T cells upon mesenchymal stem cell-lymphocyte interaction. Haematologica. 2007;92(7):881-888. [39] LE BLANC K, TAMMIK L, SUNDBERG B, et al. Mesenchymal stem cells inhibit and stimulate mixed lymphocyte cultures and mitogenic responses independently of the major histocompatibility complex. Scand J Immunol. 2003;57(1):11-20. [40] RASMUSSON I, RINGDÉN O, SUNDBERG B, et al. Mesenchymal stem cells inhibit the formation of cytotoxic T lymphocytes, but not activated cytotoxic T lymphocytes or natural killer cells. Transplantation. 2003;76(8):1208-1213. [41] DI NICOLA M, CARLO-STELLA C, MAGNI M, et al. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood. 2002;99(10):3838-3843. [42] SOTIROPOULOU PA, PEREZ SA, GRITZAPIS AD, et al. Interactions between human mesenchymal stem cells and natural killer cells. Stem Cells. 2006; 24(1):74-85. [43] DING Y, LIANG X, ZHANG Y, et al. Rap1 deficiency-provoked paracrine dysfunction impairs immunosuppressive potency of mesenchymal stem cells in allograft rejection of heart transplantation. Cell Death Dis. 2018;9(3):386. [44] REIS M, MAVIN E, NICHOLSON L, et al. Mesenchymal Stromal Cell-Derived Extracellular Vesicles Attenuate Dendritic Cell Maturation and Function. Front Immunol. 2018;9:2538. [45] KHARE D, OR R, RESNICK I, et al. Mesenchymal Stromal Cell-Derived Exosomes Affect mRNA Expression and Function of B-Lymphocytes. Front Immunol. 2018;9:3053. [46] WU SM, ZHANG WX, WANG MH, et al. Proteomic analysis of the immunosuppressive effects of mesenchymal stem cells in a rat heart transplantation model. Adv Clin Exp Med. 2013;22(6):785-794. [47] CASIRAGHI F, AZZOLLINI N, CASSIS P, et al. Pretransplant infusion of mesenchymal stem cells prolongs the survival of a semiallogeneic heart transplant through the generation of regulatory T cells. J Immunol. 2008; 181(6):3933-3946. [48] ZHOU HP, YI DH, YU SQ, et al. Administration of donor-derived mesenchymal stem cells can prolong the survival of rat cardiac allograft. Transplant Proc. 2006;38(9):3046-3051. [49] HUA F, CHEN Y, YANG Z, et al. Protective action of bone marrow mesenchymal stem cells in immune tolerance of allogeneic heart transplantation by regulating CD45RB+ dendritic cells. Clin Transplant. 2018;32(4):e13231. [50] WANG W, ZHAO N, LI B, et al. Inhibition of cardiac allograft rejection in mice using interleukin-35-modified mesenchymal stem cells. Scand J Immunol. 2019;89(4):e12750. [51] WANG H, QI F, DAI X, et al. Requirement of B7-H1 in mesenchymal stem cells for immune tolerance to cardiac allografts in combination therapy with rapamycin. Transpl Immunol. 2014;31(2):65-74. [52] WEISS ARR, LEE O, EGGENHOFER E, et al. Differential effects of heat-inactivated, secretome-deficient MSC and metabolically active MSC in sepsis and allogenic heart transplantation. Stem Cells. 2020;38(6):797-807. [53] EGGENHOFER E, RENNER P, SOEDER Y, et al. Features of synergism between mesenchymal stem cells and immunosuppressive drugs in a murine heart transplantation model. Transpl Immunol. 2011;25(2-3):141-147. [54] GE W, JIANG J, BAROJA ML, et al. Infusion of mesenchymal stem cells and rapamycin synergize to attenuate alloimmune responses and promote cardiac allograft tolerance. Am J Transplant. 2009;9(8):1760-1772. [55] MORMILE R. Prevention and management of allograft rejection in heart transplantation: are miR-126-5p and miR-126-5p-expressing mesenchymal stem cell exosomes the Ariadne’s thread? Cardiovasc Pathol. 2020;47:107208. [56] HE JG, XIE QL, LI BB, et al. Exosomes Derived from IDO1-Overexpressing Rat Bone Marrow Mesenchymal Stem Cells Promote Immunotolerance of Cardiac Allografts. Cell Transplant. 2018;27(11):1657-1683. [57] HE JG, LI BB, ZHOU L, et al. Indoleamine 2,3-dioxgenase-transfected mesenchymal stem cells suppress heart allograft rejection by increasing the production and activity of dendritic cells and regulatory T cells. J Investig Med. 2020;68(3):728-737. [58] HUANG H, HE J, TENG X, et al. Combined intrathymic and intravenous injection of mesenchymal stem cells can prolong the survival of rat cardiac allograft associated with decrease in miR-155 expression. J Surg Res. 2013; 185(2):896-903. [59] WU GD, NOLTA JA, JIN YS, et al. Migration of mesenchymal stem cells to heart allografts during chronic rejection. Transplantation. 2003;75(5): 679-685. [60] GEORGE JF, THOMAS JM. The molecular mechanisms of veto mediated regulation of alloresponsiveness. J Mol Med (Berl). 1999;77(7):519-526. [61] GRIFFITH TS, FERGUSON TA. The role of FasL-induced apoptosis in immune privilege. Immunol Today. 1997;18(5):240-244. [62] DU JF, SHEN XF, JI XQ, et al. Apoptosis of peripheral T cells in rodent cardiac allograft recipients induced by donor-specific transfusion with impaired inducible costimulator/B7 homologous protein allorecognition. Transplant Proc. 2013;45(2):564-568. [63] AIELLO S, NORIS M, PICCININI G, et al. Thymic dendritic cells express inducible nitric oxide synthase and generate nitric oxide in response to self- and alloantigens. J Immunol. 2000;164(9):4649-4658. [64] OLUWOLE SF, CHOWDHURY NC, JIN MX, et al. Induction of transplantation tolerance to rat cardiac allografts by intrathymic inoculation of allogeneic soluble peptides. Transplantation. 1993;56(6):1523-1527. [65] DEMIRKIRAN A, KOK A, KWEKKEBOOM J, et al. Low circulating regulatory T-cell levels after acute rejection in liver transplantation. Liver Transpl. 2006; 12(2):277-284. [66] HUANG Y, SHAN J, ZHANG C, et al. Peripheral blood T regulatory cell counts may not predict transplant rejection. BMC Immunol. 2010;11:40. [67] RAIMONDI G, SUMPTER TL, MATTA BM, et al. Mammalian target of rapamycin inhibition and alloantigen-specific regulatory T cells synergize to promote long-term graft survival in immunocompetent recipients. J Immunol. 2010;184(2):624-636. [68] MOSMANN TR, CHERWINSKI H, BOND MW, et al. Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J Immunol. 1986;136(7):2348-2357. [69] ONODERA K, CHANDRAKER A, VOLK HD, et al. Distinct tolerance pathways in sensitized allograft recipients after selective blockade of activation signal 1 or signal 2. Transplantation. 1999;68(2):288-293. [70] ZHENG XX, MARKEES TG, HANCOCK WW, et al. CTLA4 signals are required to optimally induce allograft tolerance with combined donor-specific transfusion and anti-CD154 monoclonal antibody treatment. J Immunol. 1999;162(8):4983-4990. [71] LARSEN CP, ELWOOD ET, ALEXANDER DZ, et al. Long-term acceptance of skin and cardiac allografts after blocking CD40 and CD28 pathways. Nature. 1996;381(6581):434-438. [72] FU F, LI Y, QIAN S, et al. Costimulatory molecule-deficient dendritic cell progenitors (MHC class II+, CD80dim, CD86-) prolong cardiac allograft survival in nonimmunosuppressed recipients. Transplantation. 1996;62(5): 659-665. [73] BANCHEREAU J, BRIERE F, CAUX C, et al. Immunobiology of dendritic cells. Annu Rev Immunol. 2000;18:767-811. [74] DIETERLEN MT, JOHN K, REICHENSPURNER H, et al. Dendritic Cells and Their Role in Cardiovascular Diseases: A View on Human Studies. J Immunol Res. 2016;2016:5946807. [75] MORELLI AE, THOMSON AW. Tolerogenic dendritic cells and the quest for transplant tolerance. Nat Rev Immunol. 2007;7(8):610-621. [76] TANER T, HACKSTEIN H, WANG Z, et al. Rapamycin-treated, alloantigen-pulsed host dendritic cells induce ag-specific T cell regulation and prolong graft survival. Am J Transplant. 2005;5(2):228-236. [77] PÊCHE H, TRINITÉ B, MARTINET B, et al. Prolongation of heart allograft survival by immature dendritic cells generated from recipient type bone marrow progenitors. Am J Transplant. 2005;5(2):255-267. [78] BÉRIOU G, PÊCHE H, GUILLONNEAU C, et al. Donor-specific allograft tolerance by administration of recipient-derived immature dendritic cells and suboptimal immunosuppression. Transplantation. 2005;79(8):969-972. [79] HIKAWA H, LOTZE MT, ROSENBLUM WD. Induction of peripheral tolerance by local delivery of dendritic cell progenitors to cardiac allografts in a murine heterotopic heart transplantation model. Gen Thorac Cardiovasc Surg. 2007;55(8):307-314. [80] THIELE J, VARUS E, WICKENHAUSER C, et al. Mixed chimerism of cardiomyocytes and vessels after allogeneic bone marrow and stem-cell transplantation in comparison with cardiac allografts. Transplantation. 2004;77(12):1902-1905. [81] FÄNDRICH F, LIN X, CHAI GX, et al. Preimplantation-stage stem cells induce long-term allogeneic graft acceptance without supplementary host conditioning. Nat Med. 2002;8(2):171-178. [82] URSCHEL S, RYAN LA, LARSEN IM, et al. Development of B-cell memory in early childhood and the impact on antigen-specific tolerance after heart transplantation. J Heart Lung Transplant. 2016;35(4):491-499. [83] FAN X, ANG A, POLLOCK-BARZIV SM, et al. Donor-specific B-cell tolerance after ABO-incompatible infant heart transplantation. Nat Med. 2004;10(11):1227-1233. [84] JOHN K, DIETERLEN MT, TARNOK A, et al. Role of dendritic cells in the context of acute cellular rejection: comparison between tacrolimus- or cyclosporine A-treated heart transplanted recipients. Cytometry B Clin Cytom. 2014;86(5):362-367. [85] ODERUP C, MALM H, EKBERG H, et al. Costimulation blockade-induced cardiac allograft tolerance: inhibition of T cell expansion and accumulation of intragraft cD4(+)Foxp3(+) T cells. Transplantation. 2006;82(11): 1493-1500. [86] LISIK W, TEJPAL N, GONG Y, et al. Down regulation of genes involved in T cell polarity and motility during the induction of heart allograft tolerance by allochimeric MHC I. PLoS One. 2009;4(12):e8020. [87] MENARD C, PACELLI L, BASSI G, et al. Clinical-grade mesenchymal stromal cells produced under various good manufacturing practice processes differ in their immunomodulatory properties: standardization of immune quality controls. Stem Cells Dev. 2013;22(12):1789-1801. |
| [1] | 蒲 锐, 陈子扬, 袁凌燕. 不同细胞来源外泌体保护心脏的特点与效应[J]. 中国组织工程研究, 2021, 25(在线): 1-. |
| [2] | 林清凡, 解一新, 陈婉清, 叶振忠, 陈幼芳. 人胎盘源间充质干细胞条件培养液可上调缺氧状态下BeWo细胞活力和紧密连接因子的表达[J]. 中国组织工程研究, 2021, 25(在线): 4970-4975. |
| [3] | 张 超, 吕 欣. 髋臼骨折固定后的异位骨化:危险因素、预防及其治疗进展[J]. 中国组织工程研究, 2021, 25(9): 1434-1439. |
| [4] | 周继辉, 李新志, 周 游, 黄 卫, 陈文瑶. 髌骨骨折修复内植物选择的多重问题[J]. 中国组织工程研究, 2021, 25(9): 1440-1445. |
| [5] | 王德斌, 毕郑刚. 尺骨鹰嘴骨折-脱位解剖力学、损伤特点、固定修复及3D技术应用的相关问题[J]. 中国组织工程研究, 2021, 25(9): 1446-1451. |
| [6] | 姬志祥, 蓝常贡. 尿酸盐转运蛋白在痛风中的多态性和治疗相关性[J]. 中国组织工程研究, 2021, 25(8): 1290-1298. |
| [7] | 袁 美, 张新新, 郭祎莎, 毕 霞. 循环microRNA在血管性认知障碍诊断中的应用[J]. 中国组织工程研究, 2021, 25(8): 1299-1304. |
| [8] | 张秀梅, 翟运开, 赵 杰, 赵 萌. 类器官模型国内外数据库近10年文献研究热点分析[J]. 中国组织工程研究, 2021, 25(8): 1249-1255. |
| [9] | 王正东, 黄 娜, 陈婧娴, 郑作兵, 胡鑫宇, 李 梅, 苏 晓, 苏学森, 颜 南. 丁酸钠抑制氟中毒可诱导小胶质细胞活化及炎症因子表达增多[J]. 中国组织工程研究, 2021, 25(7): 1075-1080. |
| [10] | 汪显耀, 关亚琳, 刘忠山. 提高间充质干细胞治疗难愈性创面的策略[J]. 中国组织工程研究, 2021, 25(7): 1081-1087. |
| [11] | 万 然, 史 旭, 刘京松, 王岩松. 间充质干细胞分泌组治疗脊髓损伤的研究进展[J]. 中国组织工程研究, 2021, 25(7): 1088-1095. |
| [12] | 廖成成, 安家兴, 谭张雪, 王 倩, 刘建国. 口腔鳞状细胞癌干细胞的治疗靶点及应用前景[J]. 中国组织工程研究, 2021, 25(7): 1096-1103. |
| [13] | 赵 敏, 冯柳祥, 陈 垚, 顾 霞, 王平义, 李一梅, 李文华. 低氧环境下外泌体可作为疾病的标志物[J]. 中国组织工程研究, 2021, 25(7): 1104-1108. |
| [14] | 谢文佳, 夏天娇, 周卿云, 刘羽佳, 顾小萍. 小胶质细胞介导神经元损伤在神经退行性疾病中的作用[J]. 中国组织工程研究, 2021, 25(7): 1109-1115. |
| [15] | 李珊珊, 郭笑霄, 尤 冉, 杨秀芬, 赵 露, 陈 曦, 王艳玲. 感光细胞替代治疗视网膜变性疾病[J]. 中国组织工程研究, 2021, 25(7): 1116-1121. |
细胞治疗被认为是修复边缘器官、减少缺血再灌注损伤和诱导实体器官移植免疫耐受的新方法。希望通过具有免疫调节性能细胞的干预,可以改变移植受者效应通路和调节通路之间的平衡,最终提高宿主免疫系统控制同种异体移植物免疫应答的潜力。如今间充质干细胞在器官移植实验模型中的应用得到了相对乐观的结果。越来越多的证据表明,间充质干细胞移植可以作为一种有效的免疫调节策略,诱导各种免疫相关疾病的耐受。但是该领域大量的研究数据,有一部分是缺乏可比性的、多余的,甚至是矛盾的。国际细胞治疗协会(International Society for Celluar Therapy,ISCT)在2005年对间充质干细胞进行了最低标准规范[1]。这些多能细胞可从骨髓、脂肪、脐带、胎盘等各种组织中分离出来,参与调节器官内环境平衡、组织重塑和损伤修复,通过各种方式于机体中形成一个平衡的微环境。
大量的研究表明,在器官移植后使用间充质干细胞进行免疫抑制或耐受诱导是有效和安全的。间充质干细胞的发展,为异体器官移植提供了更多的可能性,也使间充质干细胞拥有了临床应用的价值。但从动物基础实验到实际的临床应用,还有很长的路要走,需要相关领域科研人员的共同努力。文章主要从间充质干细胞、免疫细胞及心脏移植免疫耐受角度出发,综述间充质干细胞在心脏移植免疫耐受诱导中的重要作用。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
mesenchymal stem cells;MSC;MSCs;heart transplantation;
cardiac allograft;heart allograft;immune tolerance;
tolerance;immunological rejection”。
1.2 入选标准 ①间充质干细胞与心脏移植免疫耐受、延长心脏移植物存活时间相关的文献;②具有原创性,研究方法可靠,背景明确;③明确研究对象、干预措施、对照、结局及研究类型。
1.3 质量评估 共检索到358篇参考文献。按入选标准进行人工筛选,排除相关性差的研究,最终纳入87篇文献。纳入研究的文献包括研究原著、综述等。
干细胞在心脏移植免疫耐受诱导方面的研究相对较少,且多局限于动物实验。间充质干细胞的单独使用效果理论上是可观的,实际的应用中也取得了不少乐观的结果,但也不乏否定的结果出现。对间充质干细胞的预处理以及与药物联合使用似乎也激起了人们的兴趣。旨在通过这种方式尽量规避间充质干细胞的潜在危险、放大间充质干细胞的作用效果以及减少免疫抑制药物使用等。心脏移植免疫耐受的诱导途径并不算少,相关学者在此方面进行了大量的探究,期望能够进一步延长心脏移植物的存活时间,也希望它能给临床中所有终末期心脏病、心脏移植适应证的患者带来福音。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程

文题释义:#br# 心脏移植免疫耐受:心脏移植后,受者对供者来源的MHC抗原能产生细胞和体液免疫排斥反应,一般以细胞免疫为主。通过不同方式对宿主免疫系统识别同种异体抗原进行干扰,从而达到宿主对移植物异型抗原产生特异性免疫无反应(免疫耐受),达到移植物长期存活。#br# 间充质干细胞来源的胞外囊泡:主要包括外泌体和微囊泡,它们包裹着大量的生物活性因子,包括蛋白质(细胞因子、膜受体、生长因子和酶)和遗传物质(mRNA和microRNAs)。间充质干细胞来源胞外囊泡是间充质干细胞发挥免疫调节和组织再生功能的主要生物因子,在心脏移植免疫耐受诱导过程中也发挥重要作用。#br#
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||