中国组织工程研究 ›› 2021, Vol. 25 ›› Issue (13): 2079-2086.doi: 10.3969/j.issn.2095-4344.2193
• 干细胞综述 stem cell review • 上一篇 下一篇
脐带间充质干细胞在骨组织工程中的应用与进展
韩宁宁,左进富,孙 淼,唐胜建,刘方军
- 潍坊医学院整形外科研究所,山东省潍坊市 261000
-
收稿日期:2020-05-11修回日期:2020-05-16接受日期:2020-06-17出版日期:2021-05-08发布日期:2020-12-29 -
通讯作者:刘方军,教授,硕士生导师,潍坊医学院整形外科研究所,山东省潍坊市 261000 -
作者简介:韩宁宁,女,1994年生,山东省梁山县人,汉族,潍坊医学院在读硕士,主要从事骨缺损修复方面的研究。
Application and progress of umbilical cord mesenchymal stem cells in bone tissue engineering
Han Ningning, Zuo Jinfu, Sun Miao, Tang Shengjian, Liu Fangjun
- Plastic Surgery Institute, Weifang Medical University, Weifang 261000, Shandong Province, China
-
Received:2020-05-11Revised:2020-05-16Accepted:2020-06-17Online:2021-05-08Published:2020-12-29 -
Contact:Liu Fangjun, Professor, Master’s supervisor, Plastic Surgery Institute, Weifang Medical University, Weifang 261000, Shandong Province, China -
About author:Han Ningning, Master candidate, Plastic Surgery Institute, Weifang Medical University, Weifang 261000, Shandong Province, China
摘要:
 文题释义:
文题释义:
人脐带间充质干细胞:是源于新生儿脐带组织的一种多功能干细胞,具备较强的增殖、自我更新能力。同时,由于其具有多向分化潜能,成为骨、软骨、肌肉、肌腱、神经、肝、心肌等组织工程种子细胞的优良选择。目前,可从整条脐带及脐带不同组织(羊膜上皮细胞、Wharton’s jelly及脐动静脉)分别提取出间充质干细胞。
生物打印技术:采用特殊打印装置,对生物材料、活细胞及生长因子等活性因子进行程序化打印,可精确调控宏观及微观结构,最终得到三维立体组织、器官等仿生结构。此方法为细胞生物学、再生医学和组织工程重建等领域做出重要贡献。
背景:由于脐带间充质干细胞作为种子细胞具有来源广泛、取材方便、免疫原性低、成骨分化潜能及较强的增殖和自我更新能力等诸多优点,近年来关于其在骨组织工程中的应用研究越来越多。
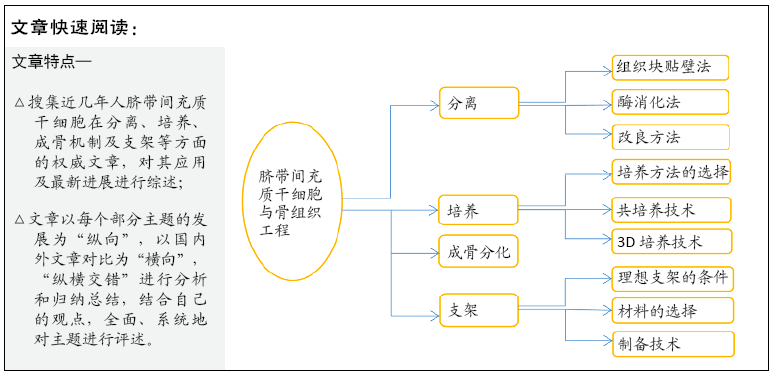
目的:对脐带间充质干细胞在分离培养、成骨诱导及其支架等方面进行综述。
方法:由第一作者检索CNKI数据库和PubMed数据库的相关文章,检索词为“人脐带间充质干细胞,分离,培养,成骨分化,支架,骨组织工程”“human umbilical cord stem cell,culture,isolation,osteogenic differentiation,scaffold,bone tissue engineering”,查阅2004至2020年来有关的文献,最终纳入104篇文献。
结果与结论:脐带间充质干细胞的分离、培养方法不一;无血清或动物血清替代物培养体系及细胞共培养技术取得了很大进展;3D培养系统将是未来发展的方向。脐带间充质干细胞成骨诱导分化机制仍不明确,需深入研究阐明。至今,开发取长补短、性能更优的复合支架仍是研究的焦点;生物打印技术突破了微米级精确调控支架内部复杂结构打造个体化支架的难题,为骨组织工程带来巨大希望;设计和制造出具有多方面理想组成(包括生物相容性、微观及宏观高孔隙率、机械性能和相关生物可吸收性等)及减少临床治疗不良反应的支架仍是骨组织工程中的重要挑战之一。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
韩宁宁, 左进富, 孙 淼, 唐胜建, 刘方军. 脐带间充质干细胞在骨组织工程中的应用与进展[J]. 中国组织工程研究, 2021, 25(13): 2079-2086.
Han Ningning, Zuo Jinfu, Sun Miao, Tang Shengjian, Liu Fangjun. Application and progress of umbilical cord mesenchymal stem cells in bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2079-2086.
2.1.1 人脐带间充质干细胞的分离方法 关于人脐带间充质干细胞的分离尚无系统特定的方法[10]。目前主要有酶消化法、组织块贴壁法及各种改良分离方法。刘奉等[11]采用组织块贴壁法和胶原酶-胰酶消化法分离培养人脐带间充质干细胞,结果发现组织贴壁法培养成功率更高、纯度较好,更适用于临床应用。WIDOWATI等[12]探讨了酶消化法和组织块贴壁法分别获取Wharton’s jelly间充质干细胞的差异,认为两者均可分离培养出间充质干细胞,且均具有三系分化能力;尽管两者表面标记物表达基本相同,但总体上组织块贴壁法的所有标记物的表达均高于酶消化法;同时,群体倍增时间实验证实组织块贴壁法获得的干细胞在传代早期具有较高的增殖能力。BOEY等[13]采用酶消化法、组织块贴壁法及机械解离法分别从冷冻保存的Wharton’s jelly中分离间充质干细胞,结果显示除机械解离法提取细胞失败外,酶消化法和组织块贴壁法均获得形态相似的间充质干细胞,但酶消化法细胞融合速度更快、产量更高且具有一致性,更适合低温保存脐带的间充质干细胞获取。为了解决临床生产环境、组织收集运输过程、先天组织变异性等原因造成的早期处理人脐带组织的难控制性,IFTIMIA-MANDER等[14]比较了组织块贴壁法和酶消化法在不同时间段对不同位置的脐带进行分离提取间充质干细胞,研究更适合实际操作条件的提取方法,在分离、运输及储备脐带组织严格操作条件的挑战下,组织块贴壁法提取人脐带间充质干细胞表现出更加稳定的性质,是更可行的方法。MORI等[15]研究者在传统组织块贴壁法的基础上添加了带有直径1 mm针孔的不锈钢网进行改良,使组织块与培养皿底部紧密相贴,结果显示分离出人脐带间充质干细胞所需时间更短、数量更多,且对细胞无损伤,这不但解决了传统组织块贴壁法中组织块易漂浮的问题,同时此方法更加简单、快捷,适用于临床。SMITH等[10]设计利用米特尼C管封闭系统机械解离脐带及酶消化方法成功分离出人脐带间充质干细胞,此种改良方法避免了剔除血管或切碎脐带等繁琐步骤带来的污染风险及产量低下等不足。
酶消化法可在短时间内获得更多细胞,缩短了原代培养时间,但其价格昂贵,步骤繁琐,污染概率高,同时掌握不好酶的浓度和消化时间,易损伤细胞,降低活性,影响细胞质量和数量。组织块贴壁法分离细胞原代培养的耗时较长,但简单易行、细胞纯度较高、活性较好。因此,目前分离培养人脐带间充质干细胞的最优方法仍未达成共识,需要进一步的研究来建立和优化标准化的分离程序。
2.1.2 人脐带间充质干细胞培养方法的选择 培养方法的选择直接影响人脐带间充质干细胞培养的成功性及其以后的临床应用。至今,研究者更多采用加适当体积分数胎牛血清的培养基[16-17]。然而,由于动物源血清培养基成分不明确,具体来源不固定,易导致免疫原性反应,甚至会携带病毒危及生物安全[18],且不同血清批次存在差异,血清质量难以控制,从而影响实验结果的可靠性。近年来,研究者们转向动物源血清替代物(包括人源血清、血浆[19]、人脐血血清和血浆[20-21]、
血小板裂解物等[22-23]、血小板血浆[24-25])或无血清培养体系培养人脐带间充质干细胞的研究。
张学娟等[25]比较了含体积分数10%胎牛血清的培养基和含体积分数15%血清替代物的培养基培养人脐带间充质干细胞,发现胎牛血清组的细胞增殖能力效果更好,但其随着细胞代数的增加出现了细胞分化或老化现象,血清替代物组则出现较均一的符合间充质干细胞国际标准的细胞,在免疫表型和分化能力上,两组无明显差异。KANDOI等[22]采用血小板裂解物替代胎牛血清的培养基培养通过组织块贴壁法和酶消化法分离获取的人脐带间充质干细胞,血小板裂解物体系培养得到的细胞均符合间充质干细胞鉴定的ISCT标准,具有典型的免疫表型、黏附性及多系分化能力,且组织块贴壁法培养的干细胞产量增加,通过实验证实经血小板裂解物长期培养后,两种方法得到的人脐带间充质干细胞仍能保持染色体稳定性。结果表明,血小板裂解物是胎牛血清的一种可行替代物,这与MOHAMMADI等[23]得出的结论相一致。杨锴等[25]将第5代人脐带间充质干细胞分别培养于添加或不添加活化富血小板血浆的培养基以及含最佳浓度富血小板血浆和成骨诱导液组合的培养基中,发现富血小板血浆亦具有促进人脐带间充质干细胞增殖和成骨分化的作用。李世梅等[26]
设计了含不同浓度鹿瓜多肽的完全培养基用于人脐带间充质干细胞的体外培养实验,通过EdU细胞增殖实验检测显示未添加鹿瓜多肽的培养基所获取培养细胞数量明显少于实验组,且随着添加鹿瓜多肽浓度增加,实验组获得的细胞数量亦明显增多,表明鹿瓜多肽对人脐带间充质干细胞的增殖有浓度梯度依赖的促进作用。
另外,细胞共培养技术应用于细胞体外培养诱导分化亦备受关注。ZHENG等[27]将兔软骨细胞和人骨髓间充质干细胞体外共培养成功诱导分化出人源性软骨细胞,在此基础上,LI等[28]通过实验研究证实低密度软骨细胞与人脐带间充质干细胞间接共培养有望成为修复软骨缺损的新方法。CHEN等[29]首次将不同来源的人脐血内皮细胞分别和4种不同来源的间充质干细胞(人脐带间充质干细胞、人骨髓间充质干细胞、人诱导性多能干细胞源性间充质干细胞和人胚胎干细胞源性间充质干细胞)共培养在磷酸钙骨水泥支架上,体外实验发现4组均成功形成了微毛细血管样结构,成骨和血管生成基因表达增高,且血管长度和细胞矿化都与培养时间呈正比;体内实验也证实细胞共培养实验组在体内获得良好的成骨和血管生成能力。这些实验为解决脐带间充质干细胞诱导效率低、缺乏生长因子或基因传递系统引导干细胞成骨分化以及血管网络化困难等缺点提供新思路、新方法,因此细胞共培养方法在骨重建中将具有良好的应用前景。
近年来,为解决传统2D培养方法细胞产量不能满足临床需求,培养微环境与人体微环境的差异导致培养出的干细胞生物学特性和功能改变,以及传统3D培养过程复杂、成本高昂等缺陷,已有研究者成功建立一种新的人脐带间充质干细胞三维培养系统[30],研究者将悬浮于无血清培养基中的原代人脐带间充质干细胞培养在表面进行硅化处理的球形玻璃培养皿中,利用悬挂摇杆系统进行摇动培养,结果得到更多数量的细胞,并具有较高的干细胞特异性标记物表达水平、良好的自我更新能力以及较强的分化能力。ZHANG等[31]利用酸溶/碱固化自球型成型方法(AASS)简单、高效地制备了京尼平交联壳聚糖/氧化石墨烯(GCS/GO)混合微球三维细胞培养支架,通过培养人脐带间充质干细胞证实干细胞可大量扩增、生长,且长期维持其特性,同时支架具有良好的细胞相容性、可控的降解性以及较低的膨胀率。三维培养系统模拟细胞生长的复杂微环境,为工业化大批生产提供新思路,具备独特的优势,将会越来越受到关注。
2.2 人脐带间充质干细胞的成骨分化 既往研究已表明人脐带间充质干细胞具有成骨分化潜能[32-33],但其具体的成骨分化机制仍不明确[34]。在基于Toll样受体可以调节间充质干细胞分化的实验基础上[35-36],KIM等[37]研究者将人脐带间充质干细胞培养在含有Toll样受体和NLRs受体激活剂的标准成骨培养基中培养,发现添加有激活剂的成骨诱导液培养的干细胞呈现出更好的成骨分化作用,且激动剂均表现出ERK磷酸化作用,同时用U0126(MEK1/2抑制剂)处理细胞后会抑制激动剂所增强的成骨分化作用,这表明NLRs和Toll样受体可能参与了间充质干细胞成骨诱导分化的分子调控,并且ERK信号通路可能对Toll样受体和NLR刺激诱导人脐带间充质干细胞的成骨分化起到了关键作用。LI等[34]通过比较添加和不添加成骨诱导培养基进行脐带Wharton’s jelly间充质干细胞的成骨诱导实验,发现诱导组碱性磷酸酶活性及骨钙素基因的表达呈升高的表现。另外使用ERK信号通路抑制剂处理干细胞后,各项成骨分化指标均显著下降,当除去抑制剂后上述指标恢复。该结果表明人脐带间充质干细胞在合适的成骨诱导刺激下具有成骨分化能力,且ERK信号通路的激活在人脐带间充质干细胞成骨分化过程中发挥了不可或缺的作用。NA等[38]使用siNotch1、硼替佐米及2.5 μmol/L GSI-I
处理成骨诱导的人脐带间充质干细胞,测定与成骨分化相关的RGC32的转录,发现只有siNotch1和硼替佐米共同处理人脐带间充质干细胞时才出现与2.5 μmol/L GSI-I处理人脐带间充质干细胞导致RGC32转录水平降低的情况一致,表明GSI-I减少人脐带间充质干细胞的成骨分化,且Notch抑制和蛋白酶体抑制都参与了此过程。这提示Notch信号可能与蛋白酶体靶向蛋白在调控人脐带间充质干细胞成骨分化过程中发挥着重要作用。尽管人脐带间充质干细胞的成骨分化潜能已得到广泛的证实,但其具体的诱导分化机制仍需全面深入地研究阐述。
2.3 支架材料 目前,寻找理想的组织工程用生物支架材料亦成为骨组织工程研究的热点之一。生物支架材料作为种子细胞黏附的载体,不仅为其提供足够的三维生长、增殖空间,同时亦作为细胞间传递生物信号的基础,以调节种子细胞的生长形态、结构特点、细胞表型及其定向诱导分化等[39-40]。
2.3.1 理想的支架材料 骨具有复杂的层次结构,支持其多种机械、生物和化学功能[41]。因此,理想的支架材料需要具备生物学、结构及实验和临床等方面的条件。
(1)生物学特征:支架必须具有良好的生物相容性才能使细胞对支架具有黏附性,保证其成活率以及增殖、分化等功能发挥正常,还要求支架及其降解产物对细胞无毒性,并且对受体无免疫原性。同样,可控的生物降解性保证支架降解与骨组织再生同时发生[41],从而防止因降解速度过快导致新生骨组织不足以承载负荷而出现骨折等后果,或由于降解速度过慢影响骨组织生成。此外生物活性亦是理想支架必须具备的重要特征之一。良好的生物活性促进骨传导、骨诱导作用等。
(2)结构特征:支架的机械性能(包括杨氏模量、抗压强度、疲劳强度和断裂韧性等)是评定支架材料是否合适的标准之一,其应与移植部位骨组织相匹配,力学过小不足以承受压力而发生骨折;力学过大导致周围骨组织的应力遮挡,导致骨组织松动等[42]。支架必须具有高孔隙率、高孔隙互连率,且具有血管化性能,利于细胞向支架迁移、氧气和营养物质输入、有害废物排出等。近年,支架的结构设计和表面性能方面受到关注[43],结构的重塑性满足不同部位骨缺损的结构仿生,改变表面性能、改善机械性能、增强骨生成等[44]。
(3)实验与临床要求:支架材料的研究都要经过验证并最终投入临床使用。理想的支架需要在实验中易观察,在临床中操作简便、手术创伤小、患者经济负担小等。
2.3.2 支架材料选择 目前,骨组织工程的支架材料主要分为聚合物、生物活性陶瓷、金属和复合材料。
至今,已有大量关于将人脐带间充质干细胞植入单独一种支架材料用于骨再生的实验研究[45-46]。由于单一的材料很难满足构建骨组织工程细胞-材料复合体的要求,故将现有材料混合以取长补短,制造出具有更优性能的复合支架成为研究的焦点。KARGOZAR等[47]分别将人骨髓间充质干细胞、人脐带间充质干细胞和人脂肪间充质干细胞装载于3D生物活性玻璃/明胶(BaG/Gel)复合支架。体外培养证实3种细胞与支架有良好的黏附和增殖能力,说明生物活性玻璃/明胶复合支架具有较好的生物相容性;在体内实验中,将其分别植入大鼠颅骨直径7 cm的缺损中,于4周和12周进行检测,结果示3组均有新骨形成,人骨髓间充质干细胞/支架组的成骨能力最强,人脐带间充质干细胞/支架组次之,但其新血管形成能力最突出,这说明将机械强度好、骨结合能力强的生物活性玻璃与生物相容性好、无免疫原性的明胶相结合的复合支架可接种人脐带间充质干细胞用于骨组织工程。WANG等[48]已首次成功将聚乳酸(丙交酯-乙交酯共聚物)(PLGA)、纳米羟基磷灰石(nHA)和壳聚糖(CS)结合在一起制备成复合支架,经体外和裸鼠皮下实验证实该复合支架对人脐带间充质干细胞的黏附率及增殖率最高,成骨分化作用及机械强度最强。为进一步评价植入细胞-支架修复裸鼠颅骨缺损的效果,研究者们通过比较了6组(PLGA支架+人脐带间充质干细胞;nHA/PLGA支架+人脐带间充质干细胞;CS/PLGA支架+人脐带间充质干细胞;nHA/CS/PLGA支架+人脐带间充质干细胞;仅有nHA/CS/PLGA支架;无支架对照组)裸鼠,发现于4周和8周时,植入人脐带间充质干细胞的nHA/CS/PLGA支架均比其他组形成的骨样组织和骨组织数量多,表明其对早期成骨有促进作用[49];8周时Van Gieson染色显示:nHA/CS/PLGA +人脐带间充质干细胞组大部分骨缺损被新生骨替代,支架大量降解;对照组未发现新生骨形成;仅有nHA/CS/PLGA支架组新生骨组织沿周边生长,支架仍有残余;而剩余3组新生骨呈岛状、块状分布,且支架仍明显残留。结果表明,nHA/CS/PLGA支架对人脐带间充质干细胞的成骨诱导性更强,支架生物降解性更好,能更好地修复大型骨缺损。纳米羟基磷灰石增强基材的机械强度以及纳米羟基磷灰石颗粒促进骨形成,同时具有多个活性基团的壳聚糖材料不仅能提供细胞识别的信号,而且其表面所携带正电荷有利于吸引带负电荷细胞的黏附[50],此外,纳米羟基磷灰石和壳聚糖亦增加了聚乳酸(丙交酯-乙交酯共聚物)表面的粗糙度,可能也是促进人脐带间充质干细胞附着、增殖和成骨分化的原因之一。研究者将无机材料、天然生物材料和合成高分子材料结合在一起,扬长避短,制造出了更加优质的复合支架。
每种材料有其各自的优缺点,见表1[51-65]。

近年来,研究者们亦越来越关注生物支架的表面性能[43],通过改变表面性能来弥补支架的缺点。已有报道通过有机相(如聚多巴胺[66]、铜离子等[67])和无机相(明胶[68]、生长因子等[69])表面涂层修饰对支架表面进行改性,优化支架功能,增强骨组织工程支架与周围环境的相互反应[44]。刘雨杰等[70]将人脐带间充质干细胞接种到多聚赖氨酸(PLL)接枝的海藻酸钠-壳聚糖(SA-CS)多孔复合支架上进行体外培养,结果表明经多聚赖氨酸优化的海藻酸钠-壳聚糖复合支架上的细胞数目及增殖速度更具有优越性,这是由于多聚赖氨酸所携带的正电荷能够促进细胞的黏附。马敏先等[71]通过将支架材料浸泡在完全培养基中来提高材料表面的的亲水性能,最终得到的细胞数量增多以及细胞增殖速度加快。同时,由于微创手术在临床外科手术中不可忽略的优点以及发展潮流,可注射式支架成为一个重要的研究方向。ZHAO 等[72-73]
已研究出将包埋于藻酸盐、水凝胶或者壳聚糖中的人脐带间充质干细胞融入磷酸钙,制成可注射的磷酸钙复合支架材料,实验证实干细胞在该支架具有良好的生存能力和成骨分化能力。WANG等[74]研发了一种可注射性混合型羟基磷灰石纳米颗粒胶体凝胶支架,其通过静电力将带负电荷的无机羟基磷灰石纳米粒和带正电荷的有机聚(D,L-乳酸-羟基乙酸)(PLGA)纳米粒子组装成具有黏性且呈三维多孔结构的胶体凝胶,正是由于静电力被剪切破坏及其可恢复性,来促进材料的挤压、成型和注射。研究者们将人脐带间充质干细胞接种于此支架上发现其对细胞的毒性可忽略不计,生物相容性好,使其成为可注射骨填充材料的理想候选者。目前,设计和制造具有多方面理想组成(包括生物相容性、微观及宏观高孔隙率、机械性能和相关生物可吸收性等)及减少临床治疗不良反应的生物支架材料成为骨组织工程研究中的重要挑战之一。
2.3.3 支架制造技术 支架制造技术主要分为传统制造技术和快速成型技术。传统制备技术存在许多限制,不能满足个体定制化支架制备的需求,近年来研究者们转向对快速成型技术的研究,将其运用于骨组织工程,提供了一种快速制造个性化仿生支架的有效方法。几种主要制造方法优缺点见表2[75-92] 。
快速成型技术是以切层-堆积自下而上为理论,利用计算机辅助设计(CAD)和/或计算机辅助制造(CAM)程序辅助的一种快速成型及构建具有可控的内部结构和外部形状等复杂结构细胞化支架的技术[93-94],其中生物3D打印具有重要的应用前景。
生物3D打印克服了传统制造的缺陷,是一种增材式的制造技术,可在微米级条件下精确调控细胞和支架结构,模拟了体内环境和天然细胞外基质的复杂性[41,95]。生物3D打印技术主要包括喷墨打印、激光辅助打印、微阀打印和挤压生物打印等[96]。目前,这种技术用于骨组织工程的油墨材料主要是水凝胶[97]。水凝胶材料细胞相容性好,具备细胞可分配性及一定黏弹性,可保证打印结构的稳定性[98],同时其在物理上约束悬浮细胞,保持细胞活性并可根据需要进行定
制[95],但其机械性能较差[99],可能会对成骨分化有抑制作
用[100],使生物打印技术受到一定程度的限制[101]。因此,研究出新型优质油墨材料成为亟待解决的问题。此外,生物打印技术为制备出具有血管化功能的支架提供了可能。有研究者通过优化两种类型甲基丙烯酰明胶(GelMA)水凝胶生物墨水分别生物打印出血管和成骨龛[102]。位于中心的可灌注血管是由可快速降解的低刚度甲基丙烯酰明胶水凝胶打印形成,并装载了人脐静脉内皮细胞和人骨髓间充质干细胞共培养,促进血管形成。周围的成骨龛则由装载有人骨髓间充质干细胞和骨诱导硅酸盐纳米血小板的高硬度甲基丙烯酰明胶水凝胶打印,所构成的生物膜预先培养1周,促进成骨分化。另外,研究者们在周围成骨龛中引入呈浓度梯度分布的化学结合血管内皮生长因子,不仅满足血管的扩张功能需求,同时促进人骨髓间充质干细胞的成骨分化。此方法生物打印出具有骨组织结构及血管系统的工程骨,为治疗大型骨缺损提供了重要治疗策略。CHEN等[103]通过生物3D打印技术将Wharton’s jelly间充质干细胞负载的水凝胶生物墨水通过压电喷嘴喷射到分层的熔融聚多巴胺修饰的硅酸钙(PDACS)/聚己内酯(PCL)支架链上制成三维复合支架。结果证实Wharton’s jelly间充质干细胞具有更高的增殖和生存能力,同时该生物支架机械性能良好且利于细胞成骨分化和血管生成。
生物打印技术具有高度空间控制和细胞传递作用[104],为骨组织工程带来巨大希望,同时其在提高空间控制率、精度和分辨率、加强支架机械性能以及实现复杂血管交通的仿生支架并应用于临床等方面仍面临巨大挑战。

| [1] GHASSEMI T, SHAHROODI A, EBRAHIMZADEH MH, et al. Current Concepts in Scaffolding for Bone Tissue Engineering. Arch Bone Jt Surg. 2018;6(2):90-99. [2] MORADI SL, GOLCHIN A, HAJISHAFIEEHA Z, et al. Bone tissue engineering: Adult stem cells in combination with electrospun nanofibrous scaffolds. J Cell Physiol. 2018;233(10):6509-6522. [3] 余炳田, 郑季南, 曾丽萍.交锁髓内钉加髂骨植骨治疗胫骨骨缺损的临床应用[J].创伤外科杂志,2017,19 (1):52-54. [4] AGARWAL R, GARCÍA AJ. Biomaterial strategies for engineering implants for enhanced osseointegration and bone repair. Adv Drug Deliv Rev. 2015;94:53-62. [5] GHANAATI S, BARBECK M, BOOMS P, et al. Potential lack of “standardized” processing techniques for production of allogeneic and xenogeneic bone blocks for application in humans. Acta Biomater. 2014;10(8):3557-3562. [6] GARCÍA-GARETA E, COATHUP MJ, BLUNN GW. Osteoinduction of bone grafting materials for bone repair and regeneration. Bone. 2015;81: 112-121. [7] LIU M, ZENG X, MA C, et al. Injectable hydrogels for cartilage and bone tissue engineering. Bone Res. 2017;5:17014. [8] JIN YZ, LEE JH. Mesenchymal Stem Cell Therapy for Bone Regeneration. Clin Orthop Surg. 2018;10(3):271-278. [9] KILLINGTON K, MAFI R, MAFI P, et al. A Systematic Review of Clinical Studies Investigating Mesenchymal Stem Cells for Fracture Non-Union and Bone Defects. Curr Stem Cell Res Ther. 2018;13(4):284-291. [10] SMITH JR, CROMER A, WEISS ML. Human Umbilical Cord Mesenchymal Stromal Cell Isolation, Expansion, Cryopreservation, and Characterization. Curr Protoc Stem Cell Biol. 2017;41:1F.18.1-1F.18.23. [11] 刘奉,蒋祥林,刘明明.不同培养方法对人脐带间充质干细胞的影响[J].中国组织工程研究,2016,20(28):4136-4141. [12] WIDOWATI W, GUNANEGARA R, RIZAL R,et al. Comparative Analysis of Wharton’s Jelly Mesenchymal Stem Cell (WJ-MSCs) Isolated Using Explant and Enzymatic Methods. Journal of Physics Conference Series. 2019;1374:012024. [13] BOEY PYK, LIM SLD, TANG KF, et al. Comparative study of the methods of extracting mesenchymal stem cells from cryopreserved Wharton’s Jelly. J Stem Cells Regen Med. 2017;13(1):29-32. [14] IFTIMIA-MANDER A, HOURD P, DAINTY R, et al. Mesenchymal stem cell isolation from human umbilical cord tissue: understanding and minimizing variability in cell yield for process optimization. Biopreserv Biobank. 2013;11(5):291-298. [15] MORI Y, OHSHIMO J, SHIMAZU T, et al. Improved explant method to isolate umbilical cord-derived mesenchymal stem cells and their immunosuppressive properties. Tissue Eng Part C Methods. 2015; 21(4):367-372. [16] SHEN C, YANG C, XU S, et al. Comparison of osteogenic differentiation capacity in mesenchymal stem cells derived from human amniotic membrane (AM), umbilical cord (UC), chorionic membrane (CM), and decidua (DC). Cell Biosci. 2019;9:17. [17] ZHANG Y, XING Y, JIA L, et al. An In Vitro Comparative Study of Multisource Derived Human Mesenchymal Stem Cells for Bone Tissue Engineering. Stem Cells Dev. 2018;27(23):1634-1645. [18] VAN DER VALK J, BIEBACK K, BUTA C, et al. Fetal Bovine Serum (FBS): Past - Present - Future. ALTEX. 2018;35(1):99-118. [19] BLÁZQUEZ-PRUNERA A, DÍEZ JM, GAJARDO R, et al. Human mesenchymal stem cells maintain their phenotype, multipotentiality, and genetic stability when cultured using a defined xeno-free human plasma fraction. Stem Cell Res Ther. 2017;8(1):103. [20] HASSAN G, KASEM I, SOUKKARIEH C, et al. A Simple Method to Isolate and Expand Human Umbilical Cord Derived Mesenchymal Stem Cells: Using Explant Method and Umbilical Cord Blood Serum. Int J Stem Cells. 2017;10(2):184-192. [21] CASEIRO AR, IVANOVA G, PEDROSA SS, et al. Human umbilical cord blood plasma as an alternative to animal sera for mesenchymal stromal cells in vitro expansion - A multicomponent metabolomic analysis. PLoS One. 2018;13(10):e0203936. [22] KANDOI S, L PK, PATRA B, et al. Evaluation of platelet lysate as a substitute for FBS in explant and enzymatic isolation methods of human umbilical cord MSCs. Sci Rep. 2018;8(1):12439. [23] MOHAMMADI S, NIKBAKHT M, MALEK MOHAMMADI A, et al. Human Platelet Lysate as a Xeno Free Alternative of Fetal Bovine Serum for the In Vitro Expansion of Human Mesenchymal Stromal Cells. Int J Hematol Oncol Stem Cell Res. 2016;10(3):161-171. [24] 杨锴,程亚栋,王鹏,等.富血小板血浆体外培养人脐带间充质干细胞:细胞增殖及成骨分化的能力[J].中国组织工程研究,2017, 21(17):2652-2658. [25] 张学娟,刘高米洋,刘菊芬,等.无血清和有血清培养人脐带间充质干细胞对比[J].中国组织工程研究,2018,22(13):2020-2026. [26] 李世梅,王黎明,李铭,等.鹿瓜多肽对脐带间充质干细胞增殖、迁移和抗炎因子分泌的影响[J].医学综述,2019,25(19):3917-3921. [27] ZHENG P, JU L, JIANG B, et al. Chondrogenic differentiation of human umbilical cord blood‑derived mesenchymal stem cells by co‑culture with rabbit chondrocytes. Mol Med Rep. 2013;8(4):1169-1182. [28] LI X, DUAN L, LIANG Y, et al. Human Umbilical Cord Blood-Derived Mesenchymal Stem Cells Contribute to Chondrogenesis in Coculture with Chondrocytes. Biomed Res Int. 2016;2016:3827057. [29] CHEN W, LIU X, CHEN Q, et al. Angiogenic and osteogenic regeneration in rats via calcium phosphate scaffold and endothelial cell co-culture with human bone marrow mesenchymal stem cells (MSCs), human umbilical cord MSCs, human induced pluripotent stem cell-derived MSCs and human embryonic stem cell-derived MSCs. J Tissue Eng Regen Med. 2018;12(1):191-203. [30] LI Y, GUO G, LI L, et al. Three-dimensional spheroid culture of human umbilical cord mesenchymal stem cells promotes cell yield and stemness maintenance. Cell Tissue Res. 2015;360(2):297-307. [31] ZHANG S, MA B, WANG S,et al. Mass-production of fluorescent chitosan/graphene oxide hybrid microspheres for in vitro 3D expansion of human umbilical cord mesenchymal stem cells. Chemical Engineering Journal. 2018;331:675-684 . [32] BEIKI B, ZEYNALI B, TAGHIABADI E, et al. Osteogenic differentiation of Wharton’s jelly-derived mesenchymal stem cells cultured on WJ-scaffold through conventional signalling mechanism. Artif Cells Nanomed Biotechnol. 2018;46(sup3):S1032-S1042. [33] 王刚,李东升,管玉涛,等.两种不同来源间充质干细胞的生物学特性比较[J].中国组织工程研究,2018,22(17):2705-2710. [34] LI CS, ZHENG Z, SU XX, et al. Activation of the Extracellular Signal-Regulated Kinase Signaling Is Critical for Human Umbilical Cord Mesenchymal Stem Cell Osteogenic Differentiation. Biomed Res Int. 2016;2016:3764372. [35] HWA CHO H, BAE YC, JUNG JS. Role of toll-like receptors on human adipose-derived stromal cells. Stem Cells. 2006;24(12):2744-2752. [36] PEVSNER-FISCHER M, MORAD V, COHEN-SFADY M, et al. Toll-like receptors and their ligands control mesenchymal stem cell functions. Blood. 2007;109(4):1422-1432. [37] KIM HS, SHIN TH, YANG SR, et al. Implication of NOD1 and NOD2 for the differentiation of multipotent mesenchymal stem cells derived from human umbilical cord blood. PLoS One. 2010;5(10):e15369. [38] NA T, LIU J, ZHANG K, et al. The notch signaling regulates CD105 expression, osteogenic differentiation and immunomodulation of human umbilical cord mesenchymal stem cells. PLoS One. 2015;10(2): e0118168. [39] HODGKINSON T, YUAN XF, BAYAT A. Adult stem cells in tissue engineering. Expert Rev Med Devices. 2009;6(6):621-640. [40] CORDONNIER T, LAYROLLE P, GAILLARD J, et al. 3D environment on human mesenchymal stem cells differentiation for bone tissue engineering. J Mater Sci Mater Med. 2010;21(3):981-987. [41] ROSETI L, PARISI V, PETRETTA M, et al. Scaffolds for Bone Tissue Engineering: State of the art and new perspectives. Mater Sci Eng C Mater Biol Appl. 2017;78:1246-1262. [42] NIINOMI M, NAKAI M. Titanium-Based Biomaterials for Preventing Stress Shielding between Implant Devices and Bone. Int J Biomater. 2011;2011:836587. [43] MOTAMEDIAN SR, HOSSEINPOUR S, AHSAIE MG, et al. Smart scaffolds in bone tissue engineering: A systematic review of literature. World J Stem Cells. 2015;7(3):657-668. [44] 方旭,董军峰.骨组织工程植入物的表面修饰[J].中国组织工程研究,2020,24(22):3573-3578. [45] XUE R, QIAN Y, LI L, et al. Polycaprolactone nanofiber scaffold enhances the osteogenic differentiation potency of various human tissue-derived mesenchymal stem cells. Stem Cell Res Ther. 2017;8(1):148. [46] CHEN W, LIU J, MANUCHEHRABADI N, et al. Umbilical cord and bone marrow mesenchymal stem cell seeding on macroporous calcium phosphate for bone regeneration in rat cranial defects. Biomaterials. 2013;34(38):9917-9925. [47] KARGOZAR S, MOZAFARI M, HASHEMIAN SJ, et al. Osteogenic potential of stem cells-seeded bioactive nanocomposite scaffolds: A comparative study between human mesenchymal stem cells derived from bone, umbilical cord Wharton’s jelly, and adipose tissue. J Biomed Mater Res B Appl Biomater. 2018;106(1):61-72. [48] WANG F, ZHANG YC, ZHOU H, et al. Evaluation of in vitro and in vivo osteogenic differentiation of nano-hydroxyapatite/chitosan/poly(lactide-co-glycolide) scaffolds with human umbilical cord mesenchymal stem cells. J Biomed Mater Res A. 2014;102(3):760-768. [49] WANG F, SU XX, GUO YC, et al. Bone regeneration by nanohydroxyapatite/chitosan/poly(lactide-co-glycolide) scaffolds seeded with human umbilical cord mesenchymal stem cells in the calvarial defects of the nude mice. Biomed Res Int. 2015;2015:261938. [50] TANASE CE, POPA MI, VERESTIUC L. Biomimetic chitosan-calcium phosphate composites with potential applications as bone substitutes: preparation and characterization. J Biomed Mater Res B Appl Biomater. 2012;100(3):700-708. [51] OGUERI KS, JAFARI T, ESCOBAR IVIRICO JL, et al. Polymeric biomaterials for scaffold-based bone regenerative engineering. Regen Eng Transl Med. 2019;5(2):128-154. [52] JAYAKUMAR R, NWE N, TOKURA S, et al. Sulfated chitin and chitosan as novel biomaterials. Int J Biol Macromol. 2007;40(3):175-181. [53] AHMED S, AHMAD M, JAYACHANDRAN M, et al. Chitosan Based Dressings for Wound Care. Immunochem Immunopathol. 2015;1:106-117. [54] ORYAN A, SAHVIEH S. Effectiveness of chitosan scaffold in skin, bone and cartilage healing. Int J Biol Macromol. 2017;104(Pt A):1003-1011. [55] PREETHI SOUNDARYA S, HARITHA MENON A, VIJI CHANDRAN S, et al. Bone tissue engineering: Scaffold preparation using chitosan and other biomaterials with different design and fabrication techniques. Int J Biol Macromol. 2018;119:1228-1239. [56] MATASSI F, NISTRI L, CHICON PAEZ D, et al. New biomaterials for bone regeneration. Clin Cases Miner Bone Metab. 2011;8(1):21-24. [57] TANG Z, LI X, TAN Y, et al. The material and biological characteristics of osteoinductive calcium phosphate ceramics. Regen Biomater. 2018; 5(1):43-59. [58] LI Y, JIANG T, ZHENG L, et al. Osteogenic differentiation of mesenchymal stem cells (MSCs) induced by three calcium phosphate ceramic (CaP) powders: A comparative study. Mater Sci Eng C Mater Biol Appl. 2017; 80:296-300. [59] CHAMPION E. Sintering of calcium phosphate bioceramics. Acta Biomater. 2013;9(4):5855-5875. [60] TIAN Y, LU T, HE F, et al. β-tricalcium phosphate composite ceramics with high compressive strength, enhanced osteogenesis and inhibited osteoclastic activities. Colloids Surf B Biointerfaces. 2018;167:318-327. [61] BEJARANO J, DETSCH R, BOCCACCINI AR, et al. PDLLA scaffolds with Cu- and Zn-doped bioactive glasses having multifunctional properties for bone regeneration. J Biomed Mater Res A. 2017;105(3):746-756. [62] KACZMAREK B, SIONKOWSKA A, GOŁYŃSKA M, et al. In vivo study on scaffolds based on chitosan, collagen, and hyaluronic acid with hydroxyapatite. Int J Biol Macromol. 2018;118(Pt A):938-944. [63] DING W. Opportunities and challenges for the biodegradable magnesium alloys as next-generation biomaterials. Regen Biomater. 2016;3(2):79-86. [64] GLENSKE K, DONKIEWICZ P, KÖWITSCH A, et al. Applications of Metals for Bone Regeneration. Int J Mol Sci. 2018;19(3):826. [65] ALVAREZ K, NAKAJIMA H. Metallic Scaffolds for Bone Regeneration. Materials. 2009;2(3):790-832. [66] KAO CT, LIN CC, CHEN YW, et al. Poly(dopamine) coating of 3D printed poly(lactic acid) scaffolds for bone tissue engineering. Mater Sci Eng C Mater Biol Appl. 2015;56:165-173. [67] YANG Y, ZHENG K, LIANG R, et al. Cu-releasing bioactive glass/polycaprolactone coating on Mg with antibacterial and anticorrosive properties for bone tissue engineering. Biomed Mater. 2017;13(1): 015001. [68] WANG J, LI D, LI T, et al. Gelatin Tight-Coated Poly(lactide-co-glycolide) Scaffold Incorporating rhBMP-2 for Bone Tissue Engineering. Materials (Basel). 2015;8(3):1009-1026. [69] MIN J, CHOI KY, DREADEN EC, et al. Designer Dual Therapy Nanolayered Implant Coatings Eradicate Biofilms and Accelerate Bone Tissue Repair. ACS Nano. 2016;10(4):4441-4450. [70] 刘雨杰,赵锦,何志旭,等.优化海藻酸钠-壳聚糖多孔复合支架用于人脐带来源间充质干细胞的体外扩增[J].基础医学与临床, 2019,39(5):630-635. [71] 马敏先,何志旭,王梅,等.PHBHHx与人脐带间充质干细胞的体内异位成骨研究[J].重庆医学,2016,45(20):2740-2744. [72] ZHAO L, WEIR MD, XU HH. An injectable calcium phosphate-alginate hydrogel-umbilical cord mesenchymal stem cell paste for bone tissue engineering. Biomaterials. 2010;31(25):6502-6510. [73] ZHAO L, WEIR MD, XU HH. Human umbilical cord stem cell encapsulation in calcium phosphate scaffolds for bone engineering. Biomaterials. 2010;31(14):3848-3857. [74] WANG Q, GU Z, JAMAL S, et al. Hybrid hydroxyapatite nanoparticle colloidal gels are injectable fillers for bone tissue engineering. Tissue Eng Part A. 2013;19(23-24):2586-2593. [75] THAVORNYUTIKARN B, CHANTARAPANICH N, SITTHISERIPRATIP K, et al. Bone tissue engineering scaffolding: computer-aided scaffolding techniques. Prog Biomater. 2014;3:61-102. [76] BAJAJ P, SCHWELLER RM, KHADEMHOSSEINI A, et al. 3D biofabrication strategies for tissue engineering and regenerative medicine. Annu Rev Biomed Eng. 2014;16:247-276. [77] TILKORN DJ, LOKMIC Z, CHAFFER CL, et al. Disparate companions: tissue engineering meets cancer research. Cells Tissues Organs. 2010; 192(3):141-157. [78] CAI SJ, LI CW, WEIHS D, et al. Control of cell proliferation by a porous chitosan scaffold with multiple releasing capabilities. Sci Technol Adv Mater. 2017;18(1):987-996. [79] LIU L, LIU J, WANG M, et al. Preparation and characterization of nano-hydroxyapatite/silk fibroin porous scaffolds. J Biomater Sci Polym Ed. 2008;19(3):325-338. [80] AHMED S, ANNU, ALI A, et al. A review on chitosan centred scaffolds and their applications in tissue engineering. Int J Biol Macromol. 2018; 116:849-862. [81] DI LUCA A, DE WIJN JR, VAN BLITTERSWIJK CA, et al. Tailorable Surface Morphology of 3D Scaffolds by Combining Additive Manufacturing with Thermally Induced Phase Separation. Macromol Rapid Commun. 2017;38(16):1700186. [82] COLOSI C, COSTANTINI M, BARBETTA A, et al. Morphological comparison of PVA scaffolds obtained by gas foaming and microfluidic foaming techniques. Langmuir. 2013;29(1):82-91. [83] MANAVITEHRANI I, LE TYL, DALY S, et al. Formation of porous biodegradable scaffolds based on poly(propylene carbonate) using gas foaming technology. Mater Sci Eng C Mater Biol Appl. 2019;96: 824-830. [84] SCHIFFMAN JD, SCHAUER CL. One-step electrospinning of cross-linked chitosan fibers. Biomacromolecules. 2007;8(9):2665-2667. [85] DALTON PD, VAQUETTE C, FARRUGIA BL, et al. Electrospinning and additive manufacturing: converging technologies. Biomater Sci. 2013; 1(2):171-185. [86] REZVANI Z, VENUGOPAL JR, URBANSKA AM, et al. A bird’s eye view on the use of electrospun nanofibrous scaffolds for bone tissue engineering: Current state-of-the-art, emerging directions and future trends. Nanomedicine. 2016;12(7):2181-2200. [87] CASANOVA MR, REIS RL, MARTINS A, et al. The Use of Electrospinning Technique on Osteochondral Tissue Engineering. Adv Exp Med Biol. 2018;1058:247-263. [88] 吴懋亮,刘廷章,方明伦,等.快速成形技术在仿生支架制造中的应用[J].机械与电子,2004(9):68-71. [89] KIM K, YEATTS A, DEAN D, et al. Stereolithographic bone scaffold design parameters: osteogenic differentiation and signal expression. Tissue Eng Part B Rev. 2010;16(5):523-539. [90] ALIHEIDARI N, TRIPURANENI R, AMELI A, et al. Fracture resistance measurement of fused deposition modeling 3D printed polymers. Polymer Testing.2017;60:94-101. [91] DU X. 3D Bio-Printing Review. Materials Science and Engineering. 2018; 301:012023. [92] ZHANG B, LUO Y, MA L, et al. 3D bioprinting: an emerging technology full of opportunities and challenges. Bio-Design and Manufacturing. 2018;1:2-13. [93] 周茜.计算机辅助骨组织工程技术的研究进展[J].现代生物医学进展,2014,14(23):4582-4585. [94] MANDRYCKY C, WANG Z, KIM K, et al. 3D bioprinting for engineering complex tissues. Biotechnol Adv. 2016;34(4):422-434. [95] STANTON MM, SAMITIER J, SÁNCHEZ S. Bioprinting of 3D hydrogels. Lab Chip. 2015;15(15):3111-3115. [96] TURNBULL G, CLARKE J, PICARD F, et al. 3D bioactive composite scaffolds for bone tissue engineering. Bioact Mater. 2017;3(3):278-314. [97] SAWKINS MJ, MISTRY P, BROWN BN, et al. Cell and protein compatible 3D bioprinting of mechanically strong constructs for bone repair. Biofabrication. 2015;7(3):035004. [98] PATI F, JANG J, HA DH, et al. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat Commun. 2014;5:3935. [99] FAN R, PIOU M, DARLING E, et al. Bio-printing cell-laden Matrigel-agarose constructs. J Biomater Appl. 2016;31(5):684-692. [100] LI CC, KHARAZIHA M, MIN C, et al. Microfabrication of Cell-Laden Hydrogels for Engineering Mineralized and Load Bearing Tissues. Adv Exp Med Biol. 2015;881:15-31. [101] MORONI L, BOLAND T, BURDICK JA, et al. Biofabrication: A Guide to Technology and Terminology. Trends Biotechnol. 2018;36(4):384-402. [102] BYAMBAA B, ANNABI N, YUE K, et al. Bioprinted Osteogenic and Vasculogenic Patterns for Engineering 3D Bone Tissue. Adv Healthc Mater. 2017;6(16):1700015. [103] CHEN YW, SHEN YF, HO CC, et al. Osteogenic and angiogenic potentials of the cell-laden hydrogel/mussel-inspired calcium silicate complex hierarchical porous scaffold fabricated by 3D bioprinting. Mater Sci Eng C Mater Biol Appl. 2018;91:679-687. [104] TRICOMI BJ, DIAS AD, CORR DT. Stem cell bioprinting for applications in regenerative medicine. Ann N Y Acad Sci. 2016;1383(1):115-124. |
| [1] | 蒲 锐, 陈子扬, 袁凌燕. 不同细胞来源外泌体保护心脏的特点与效应[J]. 中国组织工程研究, 2021, 25(在线): 1-. |
| [2] | 林清凡, 解一新, 陈婉清, 叶振忠, 陈幼芳. 人胎盘源间充质干细胞条件培养液可上调缺氧状态下BeWo细胞活力和紧密连接因子的表达[J]. 中国组织工程研究, 2021, 25(在线): 4970-4975. |
| [3] | 张 超, 吕 欣. 髋臼骨折固定后的异位骨化:危险因素、预防及其治疗进展[J]. 中国组织工程研究, 2021, 25(9): 1434-1439. |
| [4] | 周继辉, 李新志, 周 游, 黄 卫, 陈文瑶. 髌骨骨折修复内植物选择的多重问题[J]. 中国组织工程研究, 2021, 25(9): 1440-1445. |
| [5] | 王德斌, 毕郑刚. 尺骨鹰嘴骨折-脱位解剖力学、损伤特点、固定修复及3D技术应用的相关问题[J]. 中国组织工程研究, 2021, 25(9): 1446-1451. |
| [6] | 姬志祥, 蓝常贡. 尿酸盐转运蛋白在痛风中的多态性和治疗相关性[J]. 中国组织工程研究, 2021, 25(8): 1290-1298. |
| [7] | 袁 美, 张新新, 郭祎莎, 毕 霞. 循环microRNA在血管性认知障碍诊断中的应用[J]. 中国组织工程研究, 2021, 25(8): 1299-1304. |
| [8] | 张秀梅, 翟运开, 赵 杰, 赵 萌. 类器官模型国内外数据库近10年文献研究热点分析[J]. 中国组织工程研究, 2021, 25(8): 1249-1255. |
| [9] | 王正东, 黄 娜, 陈婧娴, 郑作兵, 胡鑫宇, 李 梅, 苏 晓, 苏学森, 颜 南. 丁酸钠抑制氟中毒可诱导小胶质细胞活化及炎症因子表达增多[J]. 中国组织工程研究, 2021, 25(7): 1075-1080. |
| [10] | 汪显耀, 关亚琳, 刘忠山. 提高间充质干细胞治疗难愈性创面的策略[J]. 中国组织工程研究, 2021, 25(7): 1081-1087. |
| [11] | 万 然, 史 旭, 刘京松, 王岩松. 间充质干细胞分泌组治疗脊髓损伤的研究进展[J]. 中国组织工程研究, 2021, 25(7): 1088-1095. |
| [12] | 廖成成, 安家兴, 谭张雪, 王 倩, 刘建国. 口腔鳞状细胞癌干细胞的治疗靶点及应用前景[J]. 中国组织工程研究, 2021, 25(7): 1096-1103. |
| [13] | 赵 敏, 冯柳祥, 陈 垚, 顾 霞, 王平义, 李一梅, 李文华. 低氧环境下外泌体可作为疾病的标志物[J]. 中国组织工程研究, 2021, 25(7): 1104-1108. |
| [14] | 谢文佳, 夏天娇, 周卿云, 刘羽佳, 顾小萍. 小胶质细胞介导神经元损伤在神经退行性疾病中的作用[J]. 中国组织工程研究, 2021, 25(7): 1109-1115. |
| [15] | 李珊珊, 郭笑霄, 尤 冉, 杨秀芬, 赵 露, 陈 曦, 王艳玲. 感光细胞替代治疗视网膜变性疾病[J]. 中国组织工程研究, 2021, 25(7): 1116-1121. |
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.2 纳入标准 ①文章所述内容与人脐带间充质干细胞的分离、培养、成骨分化及支架材料和制备方法密切相关;②同一领域选择近期发表或在权威杂志上发表的文章。
1.3 排除标准 重复性研究与无关研究。
1.4 数据的提取 阅读初检得到文章的标题和摘要,排除与文章目的不符和重复性研究,共保留其中的104篇归纳总结。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
 文题释义:
文题释义:人脐带间充质干细胞:是源于新生儿脐带组织的一种多功能干细胞,具备较强的增殖、自我更新能力。同时,由于其具有多向分化潜能,成为骨、软骨、肌肉、肌腱、神经、肝、心肌等组织工程种子细胞的优良选择。目前,可从整条脐带及脐带不同组织(羊膜上皮细胞、Wharton’s jelly及脐动静脉)分别提取出间充质干细胞。
生物打印技术:采用特殊打印装置,对生物材料、活细胞及生长因子等活性因子进行程序化打印,可精确调控宏观及微观结构,最终得到三维立体组织、器官等仿生结构。此方法为细胞生物学、再生医学和组织工程重建等领域做出重要贡献。#br#
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||