[1] TIELAND M, TROUWBORST I, CLARK BC. Skeletal muscle performance and ageing. J Cachexia Sarcopenia Muscle. 2018;9(1):3-19.
[2] SCHMIDT M, SCHULER SC, HUTTNER SS, et al. Adult stem cells at work: regenerating skeletal muscle. Cell Mol Life Sci. 2019;76(13): 2559-2570.
[3] DAVIES OG, YANG L, PLAYER DJ, et al. Defining the Balance between Regeneration and Pathological Ossification in Skeletal Muscle Following Traumatic Injury. Front Physiol. 2017;8:194.
[4] QAZI TH, DUDA GN, ORT MJ, et al. Cell therapy to improve regeneration of skeletal muscle injuries. J Cachexia Sarcopenia Muscle. 2019;10(3):501-516.
[5] Yamashita N. Retrograde signaling via axonal transport through signaling endosomes. J Pharmacol Sci. 2019;141(2):91-96.
[6] HOFMANN K, ZWEIER M, STICHT H, et al. Biallelic SEMA3A defects cause a novel type of syndromic short stature.Am J Med Genet A. 2013;161A(11):2880-2889.
[7] Kanemaru H, Yamada Y, Ohazama A, et al. Semaphorin 3A Inhibits Nerve Regeneration During Early Stage after Inferior Alveolar Nerve Transection. Sci Rep. 2019;9(1):4245.
[8] Liu LN, Li XM, Ye DQ, et al. Emerging role of semaphorin-3A in autoimmune diseases. Inflammopharmacology. 2018;26(3):655-665.
[9] ACEVEDO LM, BARILLAS S, WEIS SM, et al. Semaphorin 3A suppresses VEGF-mediated angiogenesis yet acts as a vascular permeability factor. Blood. 2008;111(5):2674-2680.
[10] LECOURT S, LEPELLETIER Y, VANNEAUX V, et al. Human Muscle Progenitor Cells Displayed Immunosuppressive Effect through Galectin-1 and Semaphorin-3A. Stem Cells Int. 2012;2012:412610.
[11] HAYASHI M, NAKASHIMA T, TANIGUCHI M, et al. Osteoprotection by semaphorin 3A. Nature. 2012;485(7396):69-74.
[12] WEN HZ, JIANG H, LI L, et al. Semaphorin 3A Attenuates Electrical Remodeling at Infarct Border Zones in Rats after Myocardial Infarction. Tohoku J Exp Med. 2011;225(1):51-57.
[13] ANDERSON JE, DO MQ, DANESHVAR N, et al. The role of semaphorin3A in myogenic regeneration and the formation of functional neuromuscular junctions on new fibres. Biol Rev Camb Philos Soc. 2017;92(3):1389-1405.
[14] JONGBLOETS BC, PASTERKAMP RJ. Semaphorin signalling during development. Development. 2014;141(17):3292-3297.
[15] Rohm B, Ottemeyer A, Lohrum M, et al. Plexin/neuropilin complexes mediate repulsion by the axonal guidance signal semaphorin 3A. 2000;93(1):95-104.
[16] Guo HF, Vander Kooi CW. Neuropilin Functions as an Essential Cell Surface Receptor. J Biol Chem. 2015;290(49):29120-29126.
[17] PASCOE HG, WANG Y, ZHANG X. Structural mechanisms of plexin signaling. Prog Biophys Mol Biol. 2015;118(3):161-168.
[18] TATSUMI R, SANKODA Y, ANDERSON JE, et al. Possible implication of satellite cells in regenerative motoneuritogenesis: HGF upregulates neural chemorepellent Sema3A during myogenic differentiation. Am J Physiol Cell Physiol. 2009;297(2):C238-252.
[19] HENNINGSEN J, RIGBOLT K T, BLAGOEV B, et al. Dynamics of the skeletal muscle secretome during myoblast differentiation. Mol Cell Proteomics. 2010;9(11):2482-2496.
[20] Lee AS, Anderson JE, Joya JE, et al. Aged skeletal muscle retains the ability to fully regenerate functional architecture. Bioarchitecture. 2013;3(2):25-37.
[21] SATO Y, DO MK, SUZUKI T, et al. Satellite cells produce neural chemorepellent semaphorin 3A upon muscle injury.Anim Sci J. 2013; 84(2):185-189.
[22] Wokke JH, Van den Oord CJ, Leppink GJ, et al.Perisynaptic satellite cells in human external intercostal muscle: A quantitative and qualitative study. Anat Rec. 1989;223(2):174-180.
[23] Christov C, Chrétien F, Abou-Khalil R, et al. Muscle satellite cells and endothelial cells: close neighbors and privileged partners.Mol Biol Cell. 2007;18(4):1397-1409.
[24] WOSCZYNA MN, RANDO TA. A Muscle Stem Cell Support Group: Coordinated Cellular Responses in Muscle Regeneration. Developmental Cell. 2018;46(2):135-143.
[25] DAS DK, GRAHAM ZA, CARDOZO CP. Myokines in Skeletal Muscle Physiology and Metabolism: Recent Advances and Future Perspectives. Acta Physiologica. 2019;228(2):e13367.
[26] YIN H, PRICE F, RUDNICKI MA. Satellite cells and the muscle stem cell niche. Physiol Rev. 2013;93(1):23-67.
[27] 张潜英.调控骨骼肌干细胞功能的骨骼肌分泌因子的系统筛选 [D].北京:北京协和医学院, 2018.
[28] QAHAR M, TAKUMA Y, MIZUNOYA W, et al. Semaphorin 3A promotes activation of Pax7, Myf5, and MyoD through inhibition of emerin expression in activated satellite cells. FEBS Open Bio. 2016;6(6): 529-539.
[29] KAWABE Y, WANG YX, MCKINNELL IW, et al. Carm1 regulates Pax7 transcriptional activity through MLL1/2 recruitment during asymmetric satellite stem cell divisions. Cell Stem Cell. 2012;11(3):333-345.
[30] ASFOUR HA, ALLOUH MZ, SAID RS. Myogenic regulatory factors: The orchestrators of myogenesis after 30 years of discovery. Exp Bio Med(Maywood). 2018;243(2):118-128.
[31] SU Y, YU Y, LIU C, et al. Fate decision of satellite cell differentiation and self-renewal by miR-31-IL34 axis. Cell Death Differ. 2019;27(3):949-965.
[32] BENTZINGER CF, WANG YX, DUMONT NA, et al. Cellular dynamics in the muscle satellite cell niche. EMBO Rep. 2013;14(12):1062-1072.
[33] KUANG S, GILLESPIE MA, RUDNICKI MA. Niche Regulation of Muscle Satellite Cell Self-Renewal and Differentiation. Cell Stem Cell. 2008;2(1): 22-31.
[34] KUANG S, KURODA K, LE GRAND F, et al. Asymmetric self-renewal and commitment of satellite stem cells in muscle. Cell. 2007;129(5): 999-1010.
[35] LEBRASSEUR NK, TCHKONIA T, KIRKLAND JL. Cellular Senescence and the Biology of Aging, Disease, and Frailty.Nestle Nutr Inst Workshop Ser. 2015;83:11-18.
[36] MIKIHITO, HAYASHI, TOMOKI, et al. Autoregulation of Osteocyte Sema3A Orchestrates Estrogen Action and Counteracts Bone Aging.Cell Metab. 2019;29(3):627-637.
[37] KALINA V, ALEXANDER C, ZARINE K, et al. Semaphorin 3A Signaling Through Neuropilin-1 Is an Early Trigger for Distal Axonopathy in the SOD1G93A Mouse Model of Amyotrophic Lateral Sclerosis. J Neuropathol Exp Neurol. 2014;73(7):702-713.
[38] SCHIAFFINO S, REGGIANI C. Fiber Types in Mammalian Skeletal Muscles. Physiological Reviews. 2011;91(4):1447-1531.
[39] TATSUMI R, SUZUKI T, DO MQ, et al. Slow-Myofiber Commitment by Semaphorin 3A Secreted from Myogenic Stem Cells. Stem Cells.2017; 35(7):1815-1834.
[40] HUGHES SM, TAYLOR JM, TAPSCOTT SJ, et al. Selective accumulation of MyoD and Myogenin mRNA in fast and slow adult skeletal muscle is controlled by innervation and hormones. Development. 1993;118(4): 1137-1147.
[41] LAGORD C, SOULET L, BONAVAUD S, et al. Differential myogenicity of satellite cells isolated from extensor digitorum longus (EDL) and soleus rat muscles revealed in vitro. Cell Tissue Res. 1998;291(3):455-468.
[42] SUZUKI T, DO MK, SATO Y, et al. Comparative analysis of semaphorin 3A in soleus and EDL muscle satellite cells in vitro toward understanding its role in modulating myogenin expression.Int J Biochem Cell Biol. 2013;45(2):476-482.
[43] LIU N, GARRY GA, LI S, et al. A Twist2-dependent progenitor cell contributes to adult skeletal muscle. Nature Cell Biology. 2017;19(3): 202-213.
[44] LI S, KARRI D, SANCHEZ-ORTIZ E, et al. Sema3a-Nrp1 Signaling Mediates Fast-Twitch Myofiber Specificity of Tw2(+) Cells. Dev Cell. 2019;51(1): 89-98 e4.
[45] TURNER NJ, BADYLAK SF. Regeneration of skeletal muscle.Cell Tissue Res. 2012;347(3):759-774.
[46] Yin P, Lv H, Zhang L, et al.Semaphorin 3A: A Potential Target for Low Back Pain.Crit Rev Eukaryot Gene Expr. Front Aging Neurosci. 2015;7:216.
[47] 尹鹏滨,唐佩福,张里程,等.信号素3A在骨自稳态与骨重塑中的作用[J].解放军医学院学报,2014,35(12):1272-1274.
[48] ZHANG L, ZHANG L, MAO Z, et al. Semaphoring 3A: an association between traumatic brain injury and enhanced osteogenesis. Med Hypotheses. 2013;81(4):713-714.
[49] Fukuda T, Takeda S, Xu R, et al. Sema3A regulates bone-mass accrual through sensory innervations. Nature. 2013;497(7450):490-493.
[50] MCLOON LK. A new role for satellite cells: control of reinnervation after muscle injury by semaphorin 3A. Focus on “Possible implication of satellite cells in regenerative motoneuritogenesis: HGF upregulates neural chemorepellent Sema3A during myogenic differentiation” . Am J Physiol Cell Physiol. 2009;297(2):C227-230.
[51] Wu JH, Zhou YF, Hong CD, et al. Semaphorin-3A protects against neointimal hyperplasia after vascular injury.EBioMedicine. 2019;39: 95-108.
[52] Chen B, Shan T. The role of satellite and other functional cell types in muscle repair and regeneration. J Muscle Res Cell Motil. 2019;40(1): 1-8.
[53] YANG W, HU P. Skeletal muscle regeneration is modulated by inflammation. J Orthop Translat. 2018;13:25-32.
[54] ANDERSON JE. Hepatocyte Growth Factor and Satellite Cell Activation.Adv Exp Med Biol. 2016;900:1-25.
[55] SAKAGUCHI S, SHONO J, SUZUKI T, et al. Implication of anti-inflammatory macrophages in regenerative moto-neuritogenesis: promotion of myoblast migration and neural chemorepellent semaphorin 3A expression in injured muscle.Int J Biochem Cell Biol. 2014;54:272-285.
[56] SAWANO S, SUZUKI T, DO MK, et al. Supplementary immunocytochemistry of hepatocyte growth factor production in activated macrophages early in muscle regeneration. Anim Sci J. 2014; 85(12):994-1000.
[57] SYVERUD BC, VANDUSEN KW, LARKIN LM. Growth Factors for Skeletal Muscle Tissue Engineering. Cells Tissues Organs. 2016;202(3-4): 169-179.
[58] DO MK, SUZUKI T, GERELT B, et al. Time-coordinated prevalence of extracellular HGF, FGF2 and TGF-beta3 in crush-injured skeletal muscle.Anim Sci J. 2012;83(10):712-717.
[59] DO MK, SATO Y, SHIMIZU N, et al. Growth factor regulation of neural chemorepellent Sema3A expression in satellite cell cultures. Am J Physiol Cell Physiol. 2011;301(5):C1270-1279.
[60] RP R, RM J, CR R, et al. Satellite cell-mediated angiogenesis in vitro coincides with a functional hypoxia-inducible factor pathway. Am J Physiol Cell Physiol. 2009;296(6):C1321-1328.
[61] VERMA M, ASAKURA Y, MURAKONDA BSR, et al. Muscle Satellite Cell Cross-Talk with a Vascular Niche Maintains Quiescence via VEGF and Notch Signaling. Cell Stem Cell. 2018;23(4):530-543 e9.
[62] Duchesne E, Dufresne SS, Dumont NA. Impact of Inflammation and Anti-inflammatory Modalities on Skeletal Muscle Healing: From Fundamental Research to the Clinic. Phys Ther. 2017;97(8):807-817.
[63] MEYER LA, FRITZ J, PIERDANT-MANCERA M, et al. Current drug design to target the Semaphorin/Neuropilin/Plexin complexes. Cell Adh Migr. 2016;10(6):700-708.
[64] NEUFELD G, MUMBLAT Y, SMOLKIN T, et al.The semaphorins and their receptors as modulators of tumor progression. Drug Resist Updat. 2016;29:1-12.
|

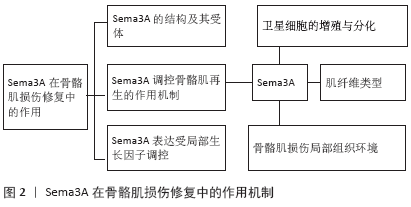 Sema3A有望成为改善骨骼肌损伤后修复的新治疗靶点,当前针对Sema3A的临床药物设计正受到许多研究者的密切关注[63]。进一步明确Sema3A及其受体在骨骼肌再生过程中的表达特征和对骨骼肌再生的调控机制,可为促进骨骼肌损伤后修复的药物研究及干细胞治疗等手段提供新的视角。值得警惕的是,Sema3A在许多肿瘤疾病的发生和侵袭中发挥复杂且重要的功能 [64],因此进一步明确Sema3A与骨骼肌组织恶性疾病的发生及侵袭之间是否存在联系,将为未来的药物安全性研究提供重要线索。
Sema3A有望成为改善骨骼肌损伤后修复的新治疗靶点,当前针对Sema3A的临床药物设计正受到许多研究者的密切关注[63]。进一步明确Sema3A及其受体在骨骼肌再生过程中的表达特征和对骨骼肌再生的调控机制,可为促进骨骼肌损伤后修复的药物研究及干细胞治疗等手段提供新的视角。值得警惕的是,Sema3A在许多肿瘤疾病的发生和侵袭中发挥复杂且重要的功能 [64],因此进一步明确Sema3A与骨骼肌组织恶性疾病的发生及侵袭之间是否存在联系,将为未来的药物安全性研究提供重要线索。