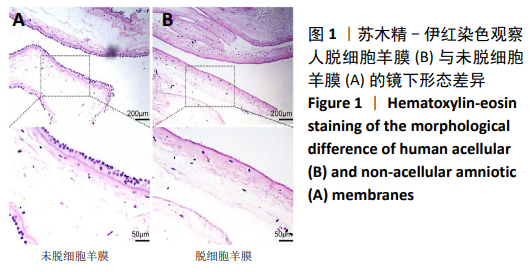
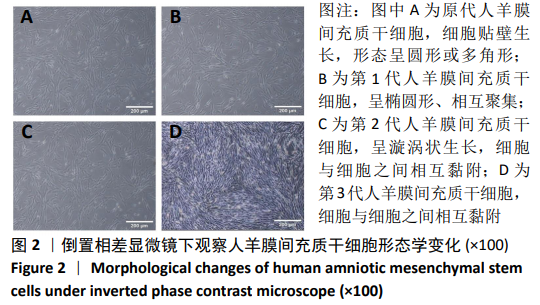
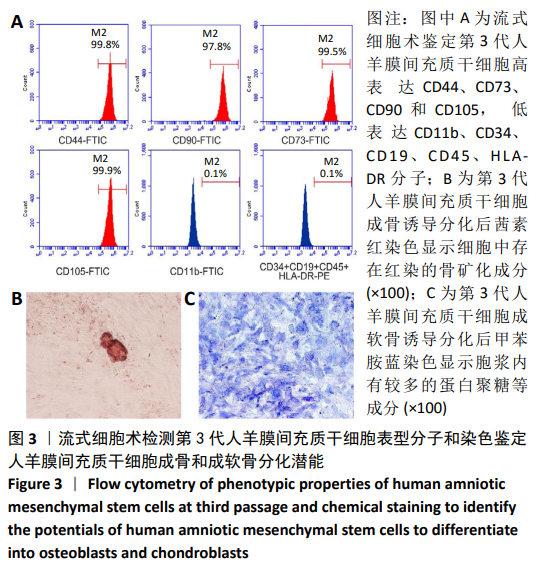
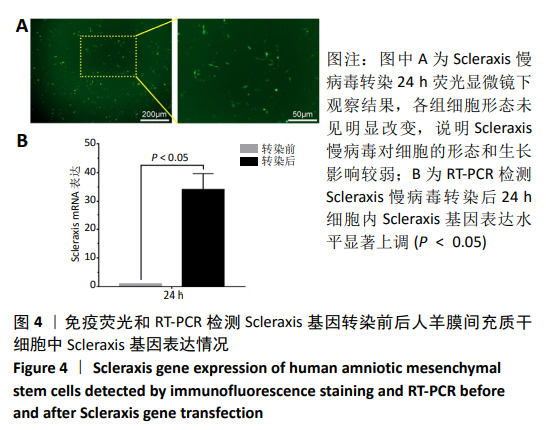
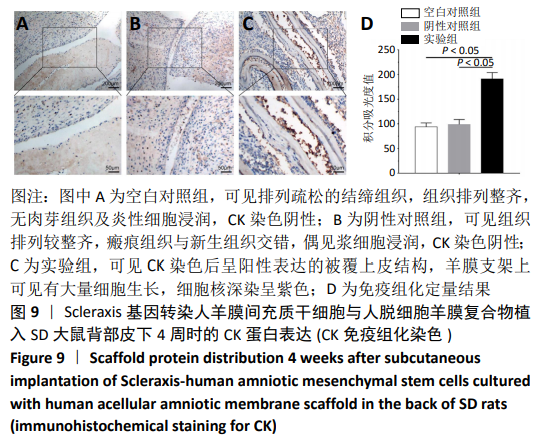
[1] ŚMIGIELSKI R, ZDANOWICZ U, DRWIĘGA M, et al. The anatomy of the anterior cruciate ligament and its relevance to the technique of reconstruction. Bone Joint J. 2016;98-B(8):1020-1026.
[2] SOMMERFELDT M, GOODINE T, RAHEEM A, et al. Relationship Between Time to ACL Reconstruction and Presence of Adverse Changes in the Knee at the Time of Reconstruction. Orthop J Sports Med. 2018;6(12):2325967118813917.
[3] AGA C, RISBERG MA, FAGERLAND MW, et al. No Difference in the KOOS Quality of Life Subscore Between Anatomic Double-Bundle and Anatomic Single-Bundle Anterior Cruciate Ligament Reconstruction of the Knee: A Prospective Randomized Controlled Trial With 2 Years’ Follow-up. Am J Sports Med. 2018; 46(10):2341-2354.
[4] RAN J, HU Y, LE H, et al. Ectopic tissue engineered ligament with silk collagen scaffold for ACL regeneration: A preliminary study. Acta Biomater. 2017;53: 307-317.
[5] ZHANG PX, HAN N, KOU YH, et al. Tissue engineering for the repair of peripheral nerve injury. Neural Regen Res. 2019;14(1):51-58.
[6] ZHOU H, WANG L, ZHANG C, et al. Feasibility of repairing full-thickness skin defects by iPSC-derived epithelial stem cells seeded on a human acellular amniotic membrane. Stem Cell Res Ther. 2019;10(1):155.
[7] XUE SL, LIU K, PAROLINI O, et al. Human acellular amniotic membrane implantation for lower third nasal reconstruction: a promising therapy to promote wound healing. Burns Trauma. 2018;6:34.
[8] 杨继彬,朱喜忠,熊华章,等.人羊膜间充质干细胞与人脱细胞羊膜支架的生物相容性[J].中国组织工程研究杂志,2018,22(5):742-747.
[9] LIU Z, LI Y, REN Y, et al. Enhancement of in vitro proliferation and bioactivity of human anterior cruciate ligament fibroblasts using an in situ tissue isolation method and basic fibroblast growth factor culture conditions: A pilot analysis. Medicine (Baltimore). 2019;98(22):e15907.
[10] LARSON BL, YU SN, PARK H, et al. Chondrogenic, hypertrophic, and osteochondral differentiation of human mesenchymal stem cells on three-dimensionally woven scaffolds. J Tissue Eng Regen Med. 2019;13(8):1453-1465.
[11] HASSAN G, KASEM I, SOUKKARIEH C, et al. A Simple Method to Isolate and Expand Human Umbilical Cord Derived Mesenchymal Stem Cells: Using Explant Method and Umbilical Cord Blood Serum. Int J Stem Cells. 2017;10(2):184-192.
[12] HE A, LIU L, LUO X, et al. Repair of osteochondral defects with in vitro engineered cartilage based on autologous bone marrow stromal cells in a swine model. Sci Rep. 2017;7:40489.
[13] LIU R, ZHANG X, FAN Z, et al. Human amniotic mesenchymal stem cells improve the follicular microenvironment to recover ovarian function in premature ovarian failure mice. Stem Cell Res Ther. 2019;10(1):299.
[14] ABBASI-KANGEVARI M, GHAMARI SH, SAFAEINEJAD F, et al. Potential Therapeutic Features of Human Amniotic Mesenchymal Stem Cells in Multiple Sclerosis: Immunomodulation, Inflammation Suppression, Angiogenesis Promotion, Oxidative Stress Inhibition, Neurogenesis Induction, MMPs Regulation, and Remyelination Stimulation. Front Immunol. 2019;10:238.
[15] YUAN Z, BIAN Y, MA X, et al. LncRNA H19 Knockdown in Human Amniotic Mesenchymal Stem Cells Suppresses Angiogenesis by Associating with EZH2 and Activating Vasohibin-1. Stem Cells Dev. 2019;28(12):781-790.
[16] GOHI BFCA, LIU XY, ZENG HY, et al. Enhanced efficiency in isolation and expansion of hAMSCs via dual enzyme digestion and micro-carrier. Cell Biosci. 2020;10:2.
[17] BIAN Y, DU Y, WANG R, et al. A comparative study of HAMSCs/HBMSCs transwell and mixed coculture systems. IUBMB Life. 2019;71(7):1048-1055.
[18] ZHANG C, DU Y, YUAN H, et al. HAMSCs/HBMSCs coculture system ameliorates osteogenesis and angiogenesis against glucolipotoxicity. Biochimie. 2018;152: 121-133.
[19] CHEN W, XIAO S, WEI Z, et al. Schwann Cell-Like Cells Derived from Human Amniotic Mesenchymal Stem Cells Promote Peripheral Nerve Regeneration through a MicroRNA-214/c-Jun Pathway. Stem Cells Int. 2019;2019:2490761.
[20] LI Y, LIU Z, TANG Y, et al. Schnurri-3 regulates BMP9-induced osteogenic differentiation and angiogenesis of human amniotic mesenchymal stem cells through Runx2 and VEGF. Cell Death Dis. 2020;11(1):72.
[21] BIAN Y, MA X, WANG R, et al. Human amnion-derived mesenchymal stem cells promote osteogenesis of human bone marrow mesenchymal stem cells against glucolipotoxicity. FEBS Open Bio. 2018;9(1):74-81.
[22] TOPOLUK N, HAWKINS R, TOKISH J, et al. Amniotic Mesenchymal Stromal Cells Exhibit Preferential Osteogenic and Chondrogenic Differentiation and Enhanced Matrix Production Compared With Adipose Mesenchymal Stromal Cells. Am J Sports Med. 2017;45(11):2637-2646.
[23] CRANE JL, XIAN L, CAO X. Role of TGF-β Signaling in Coupling Bone Remodeling. Methods Mol Biol. 2016;1344:287-300.
[24] 邹刚,李豫皖,金瑛,等.TGF-β1联合VEGF对人羊膜间充质干细胞向韧带成纤维细胞体外分化作用的研究[J].中国修复重建外科杂志,2017,31(5): 582-593.
[25] KAGUE E, HUGHES SM, LAWRENCE EA, et al. Scleraxis genes are required for normal musculoskeletal development and for rib growth and mineralization in zebrafish. FASEB J. 2019;33(8):9116-9130.
[26] HSIEH CF, YAN Z, SCHUMANN RG, et al. In Vitro Comparison of 2D-Cell Culture and 3D-Cell Sheets of Scleraxis-Programmed Bone Marrow Derived Mesenchymal Stem Cells to Primary Tendon Stem/Progenitor Cells for Tendon Repair. Int J Mol Sci. 2018;19(8). pii: E2272.
[27] SAKABE T, SAKAI K, MAEDA T, et al. Transcription factor scleraxis vitally contributes to progenitor lineage direction in wound healing of adult tendon in mice. J Biol Chem. 2018;293(16):5766-5780.
[28] ALBERTON P, POPOV C, PRÄGERT M, et al. Conversion of human bone marrow-derived mesenchymal stem cells into tendon progenitor cells by ectopic expression of scleraxis. Stem Cells Dev. 2012;21(6):846-858.
[29] NICHOLS AEC, WERRE SR, DAHLGREN LA. Transient Scleraxis Overexpression Combined with Cyclic Strain Enhances Ligament Cell Differentiation. Tissue Eng Part A. 2018;24(19-20):1444-1455.
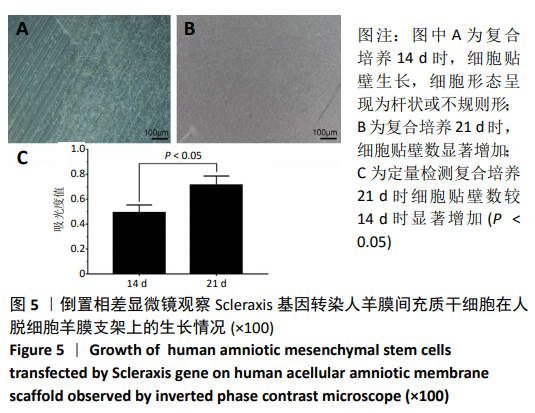
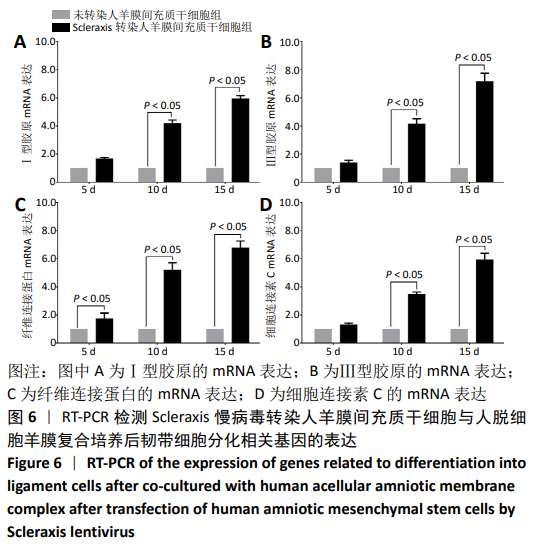
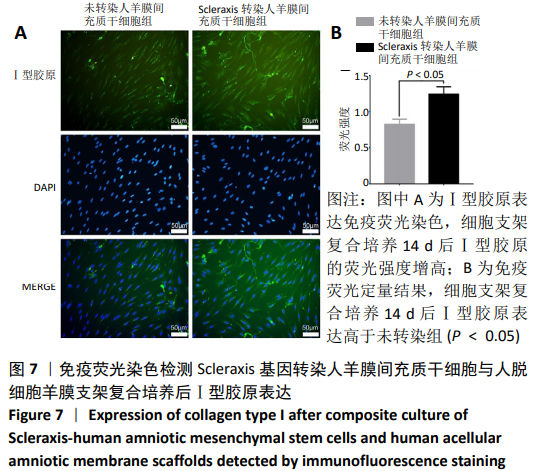
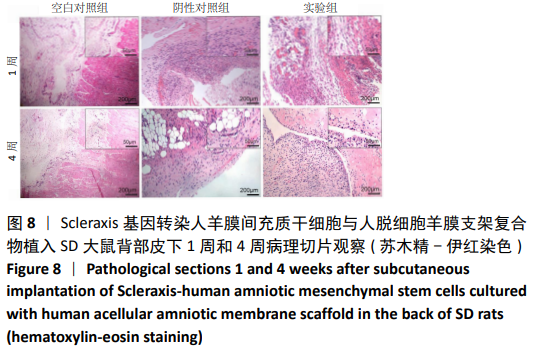
[30] 朱喜忠,刘子铭,吴术红.Scleraxis慢病毒基因感染人羊膜间充质干细胞向肌腱细胞的定向分化[J].中国组织工程研究,2017,21(33):5382-5387.
[31] NOVAKOVA SS, MAHALINGAM VD, FLORIDA SE, et al. Tissue-engineered tendon constructs for rotator cuff repair in sheep. J Orthop Res. 2018;36(1):289-299.
[32] CHEN C, LIU F, TANG Y, et al. Book-Shaped Acellular Fibrocartilage Scaffold with Cell-loading Capability and Chondrogenic Inducibility for Tissue-Engineered Fibrocartilage and Bone-Tendon Healing. ACS Appl Mater Interfaces. 2019;11(3):2891-2907.
[33] SANAEI P, CUMMINGS LJ, WATERS SL, et al. Curvature- and fluid-stress-driven tissue growth in a tissue-engineering scaffold pore. Biomech Model Mechanobiol. 2019;18(3):589-605.
[34] ZHANG Z, ZENG L, YANG J, et al. Amniotic membrane-derived stem cells help repair osteochondral defect in a weight-bearing area in rabbits. Exp Ther Med. 2017;14(1):187-192.
[35] CHEHELCHERAGHI F, EIMANI H, HOMAYOONSADRAIE S, et al. Effects of Acellular Amniotic Membrane Matrix and Bone Marrow-Derived Mesenchymal Stem Cells in Improving Random Skin Flap Survival in Rats. Iran Red Crescent Med J. 2016;18(6):e25588.
[36] ZHANG L, ZOU D, LI S, et al. An Ultra-thin Amniotic Membrane as Carrier in Corneal Epithelium Tissue-Engineering. Sci Rep. 2016;6:21021.
[37] TANG K, WU J, XIONG Z, et al. Human acellular amniotic membrane: A potential osteoinductive biomaterial for bone regeneration. J Biomater Appl. 2018;32(6):754-764.
[38] JORGE LF, FRANCISCO JC, BERGONSE N, et al. Tracheal repair with acellular human amniotic membrane in a rabbit model. J Tissue Eng Regen Med. 2018; 12(3):e1525-e1530.
[39] ZHENG Y, JI S, WU H, et al. Topical administration of cryopreserved living micronized amnion accelerates wound healing in diabetic mice by modulating local microenvironment. Biomaterials. 2017;113:56-67.
[40] DESPEYROUX A, DURET C, GONDEAU C, et al. Mesenchymal stem cells seeded on a human amniotic membrane improve liver regeneration and mouse survival after extended hepatectomy. J Tissue Eng Regen Med. 2018;12(4):1062-1073. |