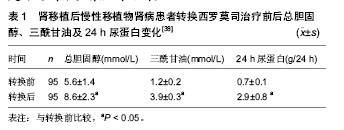
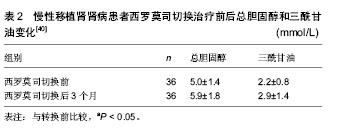
| [1] Ojo AO, Held PJ, Port FK, et al. Chronic renal failure after transplantation of a nonrenal organ. N Engl J Med.2003;349 (10): 931-940.[2] Glotz D,Charpentier B,Abramovicz D,et al.Thymoglobulin induction and sirolimus versus tacrolimus in kidney transplant recipients receiving mycophenolate mofetil and steroids. Transplantation.2010;89(12):1511-1517.[3] Hamdy AF, Bakr MA, Ghoneim MA. Long-term efficacy and safety of a calcineurin inhibitor-free regimen in live-donor renal transplant recipients. J Am Soc Nephrol.2008; 19(6): 1225-1232.[4] Flechner S M, Goldfarb D, Solez K, et al. Kidney transplantation with sirolimus and mycophenolate mofetil-based immunosuppression: 5-year results of a randomized prospective trial compared to calcineurin inhibitor drugs. Transplantation.2007;83(7):883-892.[5] Larson TS, Dean PG, Stegall MD, et al.Complete avoidance of calcineurin inhibitors in renal transplantation: a randomized trial comparing sirolimus and tacrolimus. Am J Transplant. 2006; 6(3):514-522.[6] Schaefer HM, Kizilisik AT, Feurer I, et al. Short-term results under three different immunosuppressive regimens at one center.Transplant Proc.2006;38(10):3466-3467.[7] Martinez-Mier G, Mendez-Lopez MT, Budar-Fernandez LF, et al. Living related kidney transplantation without calcineurin inhibitors: initial experience in a Mexican center. Transplantation. 2006;82(11):1533-1536.[8] Flechner SM, Kurian SM, Solez K, et al. De novo kidney transplantation without use of calcineurin inhibitors preserves renal structure and function at two years. Am J Transplant. 2004; 4(11):1776-1785.[9] Flechner SM, Goldfarb D, Modlin C, et al. Kidney transplantation without calcineurin inhibitor drugs: a prospective, randomized trial of sirolimus versus cyclosporine. Transplantation.2002;74(8):1070-1076.[10] Kotton CN, Kumar D, Caliendo AM, et al. International consensus guidelines on the management of cytomegalovirus in solid organ transplantation. Transplantation.2010;89(7):779-795.[11] Johnson RA, Wang X, Ma XL, et al. Human cytomegalovirus up-regulates the phosphatidylinositol 3-kinase (PI3-K) pathway: inhibition of PI3-K activity inhibits viral replication and virus-induced signaling.J Virol.2001;75(13):6022-6032.[12] Flechner SM. Sirolimus in kidney transplantation indications and practical guidelines: de novo sirolimus-based therapy without calcineurin inhibitors. Transplantation.2009;87 (8 Suppl): S1-S6.[13] Durrbach A, Rostaing L, Tricot L, et al. Prospective comparison of the use of sirolimus and cyclosporine in recipients of a kidney from an expanded criteria donor. Transplantation.2008;85(3):486-490.[14] Kreis H,Cisterne J M, Land W, et al. Sirolimus in association with mycophenolate mofetil induction for the prevention of acute graft rejection in renal allograft recipients. Transplantation. 2000;69(7):1252-1260.[15] Groth CG, Backman L, Morales JM, et al. Sirolimus (rapamycin)-based therapy in human renal transplantation: similar efficacy and different toxicity compared with cyclosporine. Sirolimus European Renal Transplant Study Group. Transplantation.1999;67(7):1036-1042.[16] Ekberg H,Bernasconi C,Tedesco-Silva H,et al.Calcineurin inhibitor minimization in the Symphony study: observational results 3 years after transplantation. Am J Transplant.2009; 9(8):1876-1885.[17] Flechner SM, Glyda M, Cockfield S, et al.The ORION study: comparison of two sirolimus-based regimens versus tacrolimus and mycophenolate mofetil in renal allograft recipients. Am J Transplant.2011;11(8):1633-1644.[18] Mills RE, Taylor KR, Podshivalova K, et al. Defects in skin gamma delta T cell function contribute to delayed wound repair in rapamycin-treated mice.J Immunol.2008;181(6): 3974-3983.[19] Tiong HY, Flechner SM, Zhou L, et al. A systematic approach to minimizing wound problems for de novo sirolimus-treated kidney transplant recipients. Transplantation.2009; 87(2): 296-302.[20] Schena FP, Pascoe MD, Alberu J, et al. Conversion from calcineurin inhibitors to sirolimus maintenance therapy in renal allograft recipients: 24-month efficacy and safety results from the CONVERT trial. Transplantation.2009 ; 87(2): 233-242.[21] Guba M, Pratschke J, Hugo C, et al. Early conversion to a sirolimus-based, calcineurin-inhibitor-free immunosuppression in the SMART trial: observational results at 24 and 36months after transplantation. Transpl Int.2012; 25(4):416-423.[22] Uslu A, Toz H, Sen S, et al. Late conversion from calcineurin inhibitor-based to sirolimus-based immunosuppression due to chronic toxicity: a prospective study with protocol biopsy amendment. Transplant Proc.2009;41(2):756-763.[23] Diekmann F, Budde K, Oppenheimer F, et al. Predictors of success in conversion from calcineurin inhibitor to sirolimus in chronic allograft dysfunction. Am J Transplant.2004; 4(11): 1869-1875.[24] Diekmann F, Budde K, Slowinski T, et al. Conversion to sirolimus for chronic allograft dysfunction: long-term results confirm predictive value of proteinuria. Transpl Int. 2008;21(2): 152-155.[25] Stallone G,Schena A,Infante B,et al.Sirolimus for Kaposi's sarcoma in renal-transplant recipients. N Engl J Med. 2005; 352 (13):1317-1323.[26] 韩澍,王亚伟,朱有华,等.肾移植术后并发卡波西肉瘤一例报告[J].第二军医大学学报,2007,28(7):708.[27] Fingar DC, Blenis J. Target of rapamycin (TOR): an integrator of nutrient and growth factor signals and coordinator of cell growth and cell cycle progression. Oncogene.2004; 23(18): 3151-3171.[28] Fingar DC, Richardson CJ, Tee AR, et al. mTOR controls cell cycle progression through its cell growth effectors S6K1 and 4E-BP1/eukaryotic translation initiation factor 4E. Mol Cell Biol.2004;24(1):200-216.[29] Schmelzle T, Hall MN. TOR, a central controller of cell growth. Cell.2000;103(2):253-262.[30] Hashemolhosseini S, Nagamine Y, Morley SJ, et al. Rapamycin inhibition of the G1 to S transition is mediated by effects on cyclin D1 mRNA and protein stability. J Biol Chem. 1998;273(23):14424-14429.[31] Abraham RT, Wiederrecht GJ. Immunopharmacology of rapamycin. Annu Rev Immunol.1996;14:483-510.[32] Huang S, Shu L, Dilling MB, et al. Sustained activation of the JNK cascade and rapamycin-induced apoptosis are suppressed by p53/p21(Cip1). Mol Cell.2003;11(6): 1491-1501.[33] Guba M, von Breitenbuch P, Steinbauer M, et al. Rapamycin inhibits primary and metastatic tumor growth by antiangiogenesis: involvement of vascular endothelial growth factor. Nat Med.2002;8(2):128-135.[34] Ai W, Bertram PG, Tsang CK, et al. Regulation of subtelomeric silencing during stress response. Mol Cell.2002; 10(6):1295-1305.[35] Letavernier E, Bruneval P, Vandermeersch S, et al. Sirolimus interacts with pathways essential for podocyte integrity.Nephrol Dial Transplant.2009;24(2):630-638.[36] Ko HT, Yin JL, Wyburn K, et al. Sirolimus reduces vasculopathy but exacerbates proteinuria in association with inhibition of VEGF and VEGFR in a rat kidney model of chronic allograft dysfunction. Nephrol Dial Transplant.2013; 28(2):327-336.[37] Ruiz JC, Diekmann F, Campistol JM, et al. Evolution of proteinuria after conversion from calcineurin inhibitors (CNI) to sirolimus (SRL) in renal transplant patients: a multicenter study. Transplant Proc.2005;37(9):3833-3835.[38] 田晓辉,薛武军,田普训,等.钙调磷酸酶抑制剂转换为西罗莫司治疗慢性移植物肾病的疗效研究[J].器官移植,2012,3(6):320-323.[39] Morrisett JD, Abdel-Fattah G, Kahan BD. Sirolimus changes lipid concentrations and lipoprotein metabolism in kidney transplant recipients.Transplant Proc,2003,35(3 Suppl): 143S-150S.[40] 黄洪锋,吴建永,韩飞,等.慢性移植肾肾病患者西罗莫司替代钙调磷酸酶抑制剂对肾功能的影响[J].中华医学杂志,2011,91(48): 3397-3400.[41] 徐志强,万有贵,陈博,等.西罗莫司在肾移植受者中的转换治疗[J].中国组织工程研究与临床康复,2011,15(5):810-812. |