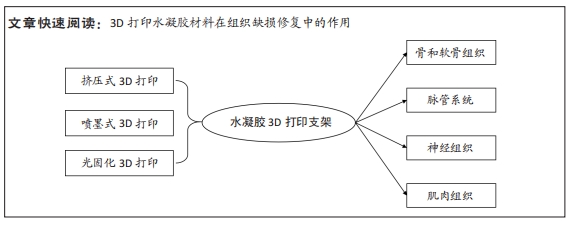
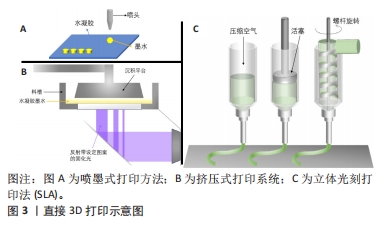
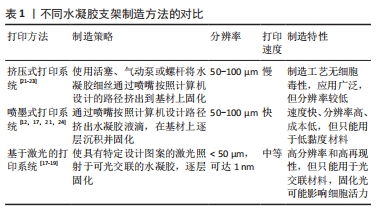
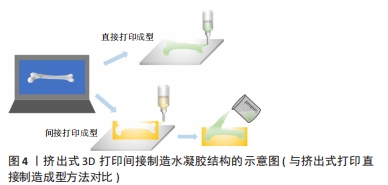
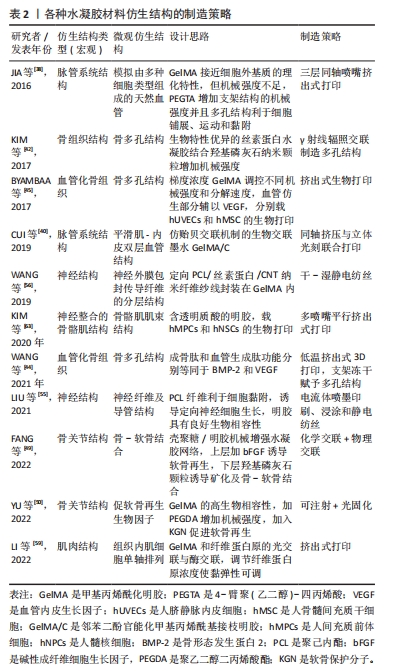
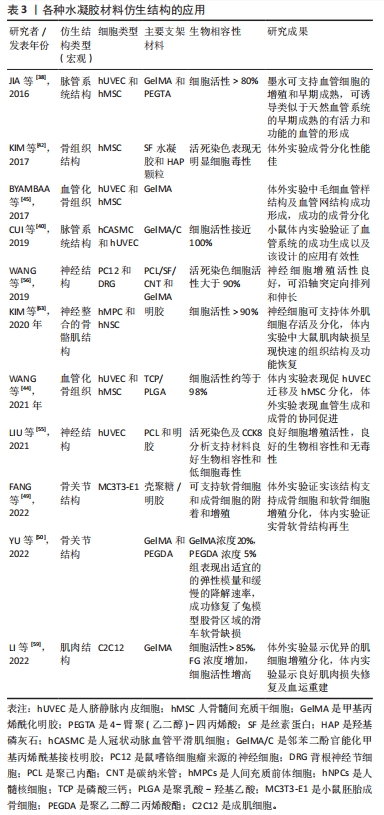
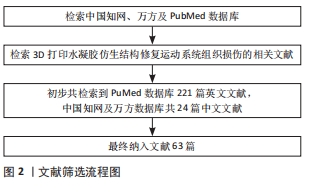
[1] 胡莹莹,李雷,张维,等.基于运动系统知识框架的解剖学教学认知体系构建[J].基础医学教育,2021,23(7):448-451.
[2] 张亮,韩泽奎,臧旖欣,等.仿生蛛网孔隙结构3D打印个性化钛网设计及三维有限元分析[J].中国组织工程研究,2023,27(30):4796-4801.
[3] NAIK RR, SINGAMANENI S. Introduction: bioinspired and biomimetic materials. Chem Rev. 2017;117(20):12581-12583.
[4] ZHANG Y, CHEN H, LI J. Recent advances on gelatin methacrylate hydrogels with controlled microstructures for tissue engineering. Int J Biol Macromol. 2022; 221:91-107.
[5] 王曙东,马倩,王可,等. 3D生物打印制备组织工程支架的研究进展[J].纺织学报,2023,44(3):210-220.
[6] WU Y. Electrohydrodynamic jet 3D printing in biomedical applications. Acta Biomater. 2021;128:21-41.
[7] SUN W, SCHAFFER S, DAI K, et al. 3D printing hydrogel-based soft and biohybrid actuators: a mini-review on fabrication techniques, applications, and challenges. Front Robot AI. 2021;8:673533.
[8] OSTROVIDOV S, SALEHI S, COSTANTINI M, et al. 3D bioprinting in skeletal muscle tissue engineering. Small. 2019;15(24):e1805530.
[9] LI J, WU C, CHU PK, et al. 3D printing of hydrogels: Rational design strategies and emerging biomedical applications. Mat Sci Eng R. 2020;140:100543.
[10] JANG TS, Jung HD, Pan HM, et al. 3D printing of hydrogel composite systems: recent advances in technology for tissue engineering. Int J Bioprint. 2018;4(1):126.
[11] KYLE S, JESSOP ZM, AL-SABAH A, et al. ‘Printability’ of candidate biomaterials for extrusion based 3D printing: state-of-the-art. Adv Healthc Mater. 2017;6(16): 1700264.
[12] LI X, LIU B, PEI B, et al. Inkjet bioprinting of biomaterials. Chem Rev. 2020;120(19): 10793-10833.
[13] AXPE E, OYEN ML. Applications of alginate-based bioinks in 3D bioprinting. Int J Mol Sci. 2016;17(12):1976.
[14] MANI MP, SADIA M, JAGANATHAN SK, et al. A review on 3D printing in tissue engineering applications. J Polym Eng, De Gruyter. 2022;42(3):243-265.
[15] ZHU W, MA X, GOU M, et al. 3D printing of functional biomaterials for tissue engineering. Curr Opin Biotechnol, 2016;40:103-112.
[16] BILLIET T, VANDENHAUTE M, SCHELFHOUT J, et al. A review of trends and limitations in hydrogel-rapid prototyping for tissue engineering. Biomaterials. 2012;33(26):6020-6041.
[17] MIRI AK, MIRZAEE I, HASSAN S, et al. Effective bioprinting resolution in tissue model fabrication. Lab Chip. 2019;19(11):2019-2037.
[18] LIN Q, TANG M, KE C. Thermo-responsive 3D-printed polyrotaxane monolith. Polymer Chemistry, The RSC. 2020;11(2):304-308.
[19] RASTOGI P, KANDASUBRAMANIAN B. Review of alginate-based hydrogel bioprinting for application in tissue engineering. Biofabrication. 2019;11(4):042001.
[20] SHINKAR K, RHODE K. Could 3D extrusion bioprinting serve to be a real alternative to organ transplantation in the future? Annals of 3D Printed Medicine. 2022;7:100066.
[21] BUDHARAJU H, SUBRAMANIAN A, SETHURAMAN S. Recent advancements in cardiovascular bioprinting and bioprinted cardiac constructs. Biomater Sci. 2021; 9(6):1974-1994.
[22] BUDHARAJU H, SUBRAMANIAN A, SETHURAMAN S. Efficient dual crosslinking of protein-in-polysaccharide bioink for biofabrication of cardiac tissue constructs. Biomater Adv. 2023;152:213486.
[23] RAMESH S, HARRYSSON OLA, RAO PK, et al. Extrusion bioprinting: recent progress, challenges, and future opportunities. Bioprinting. 2021;21:e00116.
[24] GOMES GAMA JF, DIAS EA, AGUIAR COELHO RMG, et al. Development and implementation of a significantly low-cost 3D bioprinter using recycled scrap material. Front Bioeng Biotechnol. 2023;11:1108396.
[25] CHIA HN, WU BM. Recent advances in 3D printing of biomaterials. J Biol Eng. 2015;9(1):4.
[26] HOUBEN A, VAN HOORICK J, VAN ERPS J, et al. Indirect rapid prototyping: opening up unprecedented opportunities in scaffold design and applications. Ann Biomed Eng. 2017;45(1):58-83.
[27] VAN HOORICK J, DECLERCQ H, DE MUYNCK A, et al. Indirect additive manufacturing as an elegant tool for the production of self-supporting low density gelatin scaffolds. J Mater Sci Mater Med. 2015;26(10):247.
[28] TABOAS JM, MADDOX RD, KREBSBACH PH, et al. Indirect solid free form fabrication of local and global porous, biomimetic and composite 3D polymer-ceramic scaffolds. Biomaterials. 2003;24(1):181-194.
[29] BILLIET T, GEVAERT E, DE SCHRYVER T, et al. The 3D printing of gelatin methacrylamide cell-laden tissue-engineered constructs with high cell viability. Biomaterials. 2014;35(1):49-62.
[30] LEE JM, YEONG WY. Design and printing strategies in 3D bioprinting of cell-hydrogels: a review. Adv Healthc Mater. 2016;5(22):2856-2865.
[31] KOLESKY DB, TRUBY RL, GLADMAN AS, et al. 3D bioprinting of vascularized, heterogeneous cell-laden tissue constructs. Adv Mater. 2014;26(19):3124-3130.
[32] HA DH, Ko DH, Kim JO, et al. Indirect fabrication of versatile 3D microfluidic device by a rotating plate combined 3D printing system. RSC Adv. 2018;8(66): 37693-37699.
[33] NAGHIEH S, SARKER MD, ABELSETH E, et al. Indirect 3D bioprinting and characterization of alginate scaffolds for potential nerve tissue engineering applications. J Mech Behav Biomed Mater. 2019;93:183-193.
[34] CIULLA MG, MASSIRONI A, SUGNU M, et al. Recent advances in the development of biomimetic materials. Gels. 2023;9(10):833.
[35] VINCENT JF. Biomimetics--a review. Proc Inst Mech Eng H. 2009;223(8):919-939.
[36] RADISIC M, YANG L, BOUBLIK J, et al. Medium perfusion enables engineering of compact and contractile cardiac tissue. Am J Physiol Heart Circ Physiol. 2004; 286(2):H507-H516.
[37] LIU Y, LIU X, ZHANG Y, et al. Interpenetrating polymer network HA/Alg-RGD hydrogel: an equilibrium of macroscopic stability and microscopic adaptability for 3D cell growth and vascularization. Biomacromolecules. 2023. doi: 10.1021/acs.biomac.3c01022.
[38] JIA W, GUNGOR-OZKERIM PS, Zhang YS, et al. Direct 3D bioprinting of perfusable vascular constructs using a blend bioink. Biomaterials. 2016;106:58-68.
[39] BARRS RW, JIA J, SILVER SE, et al. Biomaterials for bioprinting microvasculature. Chem Rev. 2020;120(19):10887-10949.
[40] CUI H, ZHU W, HUANG Y, et al. In vitro and in vivo evaluation of 3D bioprinted small-diameter vasculature with smooth muscle and endothelium. Biofabrication. 2019;12(1):015004.
[41] GUO J, YAO H, LI X, et al. Advanced hydrogel systems for mandibular reconstruction. Bioact Mater. 2023;21:175-193.
[42] KIM MH, KIM BS, LEE J, et al. Silk fibroin/hydroxyapatite composite hydrogel induced by gamma-ray irradiation for bone tissue engineering. Biomater Res. 2017;21:12.
[43] YAN Y, CHEN H, ZHANG H, et al. Vascularized 3D printed scaffolds for promoting bone regeneration. Biomaterials. 2019;190-191:97-110.
[44] WANG C, LAI J, LI K, et al. Cryogenic 3D printing of dual-delivery scaffolds for improved bone regeneration with enhanced vascularization. Bioact Mater. 2021; 6(1):137-145.
[45] BYAMBAA B, ANNABI N, YYE K, et al. Bioprinted osteogenic and vasculogenic patterns for engineering 3D bone tissue. Adv Healthc Mater. 2017;6(16):1700015.
[46] RIM YA, NAM Y, JU JH. Application of cord blood and cord blood-derived induced pluripotent stem cells for cartilage regeneration. Cell Transplant. 2019;28(5):529-537.
[47] GAO F , XU Z , LIANG Q, et al. Direct 3D printing of high strength biohybrid gradient hydrogel scaffolds for efficient repair of osteochondral defect. Adv Funct Mater. 2018;28(13):1706644.
[48] GAO Y, ZHANG X, ZHOU H. Biomimetic hydrogel applications and challenges in bone, cartilage, and nerve repair. Pharmaceutics. 2023;15(10):2405.
[49] FANG J, LIAO J, ZHONG C, et al. High-strength, biomimetic functional chitosan-based hydrogels for full-thickness osteochondral defect repair. ACS Biomater Sci Eng. 2022;8(10):4449-4461.
[50] YU H, FENG M, MAO G, et al. Implementation of photosensitive, injectable, interpenetrating, and kartogenin-modified GELMA/PEDGA biomimetic scaffolds to restore cartilage integrity in a full-thickness osteochondral defect model. ACS Biomater Sci Eng. 2022;8(10):4474-4485.
[51] JOHNSON K, ZHU S, TREMBLAY MS, et al. A stem cell-based approach to cartilage repair. Science. 2012;336(6082):717-721.
[52] ZHANG L, ZHANG H, WANG H, et al. Fabrication of multi-channel nerve guidance conduits containing schwann cells based on multi-material 3D bioprinting. 3D Print Addit Manuf. 2023;10(5):1046-1054.
[53] VIJAYAVENKATARAMAN S, ZHANG S, THAHARAH S, et al. Electrohydrodynamic jet 3D printed nerve guide conduits (NGCs) for peripheral nerve injury repair. Polymers. 2018;10(7):753.
[54] VIJAYAVENKATARAMAN S, THAHARAH S, ZHANG S, et al. Electrohydrodynamic jet 3D-printed PCL/PAA conductive scaffolds with tunable biodegradability as nerve guide conduits (NGCs) for peripheral nerve injury repair. Mater Design. 2019;162:171-184.
[55] LIU S, SUN L, ZHANG H, et al. High-resolution combinatorial 3D printing of gelatin-based biomimetic triple-layered conduits for nerve tissue engineering. Int J Biol Macromol. 2021;166:1280-1291.
[56] WANG L, WU Y, HU T, et al. Aligned conductive core-shell biomimetic scaffolds based on nanofiber yarns/hydrogel for enhanced 3D neurite outgrowth alignment and elongation. Acta Biomater. 2019;96:175-187.
[57] AGRAWAL G, AUNG A, VARGHESE S. Skeletal muscle-on-a-chip: an in vitro model to evaluate tissue formation and injury. Lab Chip. 2017;17(20):3447-3461.
[58] ZHANG Y, ZHANG Z, WANG Y, et al. 3D myotube guidance on hierarchically organized anisotropic and conductive fibers for skeletal muscle tissue engineering. Sci Eng C Mater Biol Appl. 2020;116:111070.
[59] LI T, HOU J, WANG L, et al. Bioprinted anisotropic scaffolds with fast stress relaxation bioink for engineering 3D skeletal muscle and repairing volumetric muscle loss. Acta Biomater. 2022;156:21-36.
[60] KO IK, LEE BK, LEE SJ, et al. The effect of in vitro formation of acetylcholine receptor (AChR) clusters in engineered muscle fibers on subsequent innervation of constructs in vivo. Biomaterials. 2013;34(13):3246-3255.
[61] VILMONT V, CADOT B, OUANOUNOU G, et al. A system for studying mechanisms of neuromuscular junction development and maintenance. Development. 2016; 143(13):2464-2477.
[62] ZHANG W, LIU Y, ZHANG H. Extracellular matrix: an important regulator of cell functions and skeletal muscle development. Cell Biosci. 2021;11:65.
[63] KIM JH, KIM I, SEOL YJ, et al. Neural cell integration into 3D bioprinted skeletal muscle constructs accelerates restoration of muscle function. Nat Commun. 2020;11:1025. |