中国组织工程研究 ›› 2024, Vol. 28 ›› Issue (7): 1083-1090.doi: 10.12307/2024.123
• 干细胞综述 stem cell review • 上一篇 下一篇
物理因子促进干细胞的成骨分化
王姗姗1,2,舒 晴2,田 峻1,2
- 1武汉体育学院运动医学院,湖北省武汉市 430079;2武汉大学中南医院康复医学科,湖北省武汉市 430071
-
收稿日期:2023-01-17接受日期:2023-03-14出版日期:2024-03-08发布日期:2023-07-17 -
通讯作者:田峻,华中科技大学同济医学院附属协和医院博士,主任医师,硕士生导师,武汉体育学院运动医学院,湖北省武汉市 430079;武汉大学中南医院康复医学科,湖北省武汉市 430071 -
作者简介:王姗姗,女,1999年生,湖南省古丈县人,土家族,武汉体育学院2022级在读硕士。 -
基金资助:国家自然科学基金面上项目(82174494),项目负责人:田峻
Physical factors promote osteogenic differentiation of stem cells
Wang Shanshan1, 2, Shu Qing2, Tian Jun1, 2
- 1School of Sports Medicine, Wuhan Sports University, Wuhan 430079, Hubei Province, China; 2Department of Rehabilitation Medicine, Zhongnan Hospital, Wuhan University, Wuhan 430071, Hubei Province, China
-
Received:2023-01-17Accepted:2023-03-14Online:2024-03-08Published:2023-07-17 -
Contact:Tian Jun, MD, Chief physician, Master’s supervisor, School of Sports Medicine, Wuhan Sports University, Wuhan 430079, Hubei Province, China; Department of Rehabilitation Medicine, Zhongnan Hospital, Wuhan University, Wuhan 430071, Hubei Province, China -
About author:Wang Shanshan, Master candidate, School of Sports Medicine, Wuhan Sports University, Wuhan 430079, Hubei Province, China; Department of Rehabilitation Medicine, Zhongnan Hospital, Wuhan University, Wuhan 430071, Hubei Province, China -
Supported by:National Natural Science Foundation of China, No. 82174494 (to TJ)
摘要:
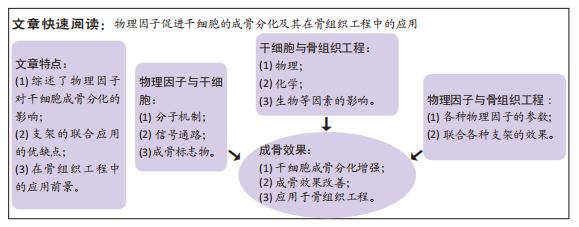
文题释义:
干细胞成骨分化:干细胞存在于多种成体组织中,包括皮肤、肌肉、骨髓和脂肪,在体外不同条件下来诱导分化成骨、软骨及其他结缔组织,因其来源广泛、分离无创伤、较低的免疫原性及生长周期短等特点,是组织工程种子细胞的重要来源。骨组织工程:是指用各种材料制备一种具有优良生物相容性、在人体内可逐步被降解吸收的细胞支架,这种仿生支架能像天然骨一样为细胞提供生存所需的三维空间,使细胞获得足够的营养物质,并进行气体交换,排除废物,细胞能够在支架上按预制形态生长,最终修复骨缺损。
目的:就物理因子如电磁场、超声等对干细胞成骨分化的分子机制以及信号通路的调控、在骨组织工程中应用的可行性等方面做一总结。
方法:应用计算机检索中国知网和PubMed数据库中近20年的相关文献,在标题和摘要中以“干细胞,骨缺损,成骨分化,电磁场,超声,冲击波,低强度激光,机械力,骨组织工程”或“stem cell,osteoporosis,osteogenic differentiation,Electromagnetic Fields,Ultrasound,Bone Tissue Engineering”为检索词进行检索,对相关文章94篇进行综述分析。
结果与结论:①物理因子作为一种无创、非接触的辅助疗法对骨组织工程有着显著影响,在调控干细胞的成骨分化、促进细胞的增殖以及在骨工程支架内的生存能力方面有着积极作用。②除了激活信号通路和成骨基因转录外,物理因子还可以改善血管化、增加支架中形成的骨的体积、面积和厚度,可以促进骨整合,提高骨支架再生健康骨组织的成功率。③然而,将物理因子用于骨组织工程的研究均采用不同的实验条件,如支架类型、细胞类型及干预条件等,无法直接进行比较以确定最佳参数设置,在临床应用中,这些不同干预对促进骨折愈合的有效性研究结果也缺乏一致性,因此未来还需要进一步确定物理因子用于骨组织工程的最佳参数。④总体而言,物理因子作为一种理想的辅助疗法,在与各种生物材料结合并应用于骨组织工程方面具有巨大的潜力。
https://orcid.org/0000-0006-5453-0607 (王姗姗);https://orcid.org/0000-0002-3053-5001 (田峻)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
王姗姗, 舒 晴, 田 峻. 物理因子促进干细胞的成骨分化[J]. 中国组织工程研究, 2024, 28(7): 1083-1090.
Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090.
细胞的成骨分化是一个复杂的过程,受物理、化学及生物等因素的影响,由多种调控因子和蛋白质共同调控。一些相关的信号通路如Wnt/β-catenin信号通路、Notch信号通路、骨形态发生蛋白2/Smads信号通路等均在成骨分化过程中起着重要作用[7-9]。研究表明,人胎盘间充质干细胞与多孔羟基磷灰石支架构成的组织工程骨,在辛伐他汀脂质体纳米微球的作用下,使骨形态发生蛋白4、骨形态发生蛋白2以及生长因子高表达,表明辛伐他汀缓释微球可促进组织工程的成骨性能表达[10]。还有研究表明生姜和大蒜提取物能诱导成骨细胞增殖、降低破骨细胞活性。在大鼠股骨远端模型中,天然来源的生姜和大蒜提取物在患者特异性3DP支架生物医学装置中促进成骨、血管生成和增强的Ⅰ型胶原形成,因此常用于低承重骨组织工程和牙科应用[11]。有研究表明在3D打印磷酸钙支架中维生素D3的释放促进成骨细胞增殖并抑制破骨细胞的吸收,从而应用于骨缺损[12]。当牙周膜干细胞接种在新型纳米羟基磷灰石包被的染料木-壳聚糖结合支架时,此支架上调细胞活力和碱性磷酸酶活性,同时诱导成骨分化标志物骨涎蛋白、骨桥蛋白和骨钙素的mRNA表达,结果表明牙周膜干细胞植入该支架时促进了骨修复[13]。有研究开发了一种新型可注射磷酸钙水泥-水凝胶纤维-干细胞构建物,该可注射支架成型快、孔隙率高,该构建物中的人诱导多能干细胞衍生的间充质干细胞、牙髓干细胞和骨髓间充质干细胞均表现出良好的增殖和成骨分化能力,碱性磷酸酶、Runt相关转录因子2、α1-1型胶原基因和骨钙素均显著升高以及干细胞在支架中的矿化量显著增加,结果表明该构建物中的干细胞均是有前景的骨组织工程细胞来源[14]。
在大段骨缺损的治疗中,要么植入具有特定性能的材料以诱导骨生长,要么经过干细胞处理以实现受损组织的再生。在临床应用方面,最佳的骨移植替代物必须是对间充质干细胞具有良好黏附性能的可生物降解、抗菌、生物相容性和骨诱导性材料。而自体间充质干细胞是最有前途的候选者。在植入之前先将自体间充质干细胞种植在种植体上,可以显著增加诱导骨再生的初始干细胞和成骨细胞的数量。通过减少细胞增殖和迁移到植入部位所需的时间,可以减少治疗时间,提高植入率。当种植体与干细胞治疗相结合时,骨诱导行为尤为重要。物理、化学以及生物等诱导因子具有很多优点,来源丰富、结构复杂多样以及广泛而可以高效、可逆、定向和准确地诱导间充质干细胞向成骨细胞分化以及促进血管长入,因此在干细胞治疗骨缺损中有重要应用前景,但是如何选择一种稳定并可在临床推广使用的方式还需进一步研究。近年来,研究者们发现物理因子作为一种新的干预手段,在干细胞的调控中发挥了作用,并可联合干细胞应用于骨组织工程中,这有望在再生医学中开发出新的领域。
2.2 物理因子可以促进干细胞成骨分化参与的骨组织工程修复效果 骨组织工程侧重于利用生物反应器对种子细胞、生长因子和生物活性支架进行体外操作[15]。骨组织工程的成功临床转化需要克服一系列重大挑战。支架是组织工程骨的中心环节,理想的支架需模拟细胞外基质的三维结构,并具备许多优势包括拥有支持细胞黏附、生长和迁移的多孔结构;以促进细胞-支架材料的相互作用;具备足够的弹性和机械性能;可控制的降解速率;新骨的形成均匀分布以避免骨坏死;体内最小程度的炎症和毒性反应等。此外理想的支架需要在植入的几周内在植入体内部或周围形成血管,以积极支持营养、氧气和废物运输等[16]。文章总结了目前应用于骨组织工程的生物支架材料以及优缺点[17-31],见表1,主要包括高分子材料、无机陶瓷和金属等。
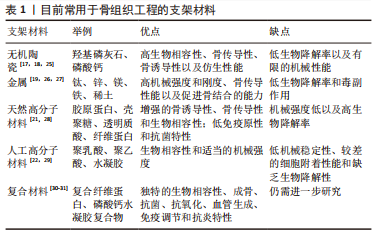
磷酸钙陶瓷是一类可调控的生物活性材料,已被广泛运用于骨组织的修复[17],它们具有支持成骨细胞黏附、增殖和刺激新骨形成的表面特性[18],在骨修复方面具有优秀的前景。但磷酸钙自身也存在诸多不足包括有限的机械强度和较高的脆性使其难以在承重部位使用。与天然来源的生物材料相比,金属材料比如钛及其合金具有良好的防腐性能和生物相容性而广泛地应用于骨组织工程支架,如全髋关节置换和全膝关节置换假体、脊柱融合器和接骨板等[19]。天然高分子材料具备良好的生物降解性和生物相容性,并可促进细胞黏附和生长,如壳聚糖、胶原蛋白、明胶、透明质酸、藻酸盐和丝素蛋白等[20]。丝素蛋白作为一种具有优异机械性能、生物可降解性、生物相容性和生物可吸收性的天然蛋白质,引起了研究者们的极大关注[21]。
尽管人工高分子材料,生物相容性相对较差,亲水性和细胞黏附能力相对较弱,并有可能引起无菌性炎症反应,但力学性能相对优于天然高分子材料的其固定的成分比例和易加工性同样是不可忽视的优势,其中应用最广泛的是聚乳酸、聚乙酸和聚乳酸聚乙酸[22]。除了传统的支架材料,3D生物打印支架似乎成为了更好的选择。与原始骨缺损填充支架不同的是其在机械强度和微观空间结构上可与人体骨组织相似,使种子细胞骨髓间充质干细胞分化成骨后可与缺损骨组织完全吻合,是在骨缺损领域具有广泛前景的新型治疗技术[23],同样类似的还有可注射生物支架,在骨组织工程方面具有很好的应用潜力[24]。
这些材料各有优缺点,同时也在不断地发展。目前用于骨缺损修复的骨组织工程支架可由多种材料、多种方法制成,这些复合支架在移植后能否长入正常的新鲜骨组织并由此代替原来的骨组织发挥作用成为其治疗成功的关键。通过混合不同的活性成分可以赋予支架新的生物活性,这为临床医生治疗骨缺损提供新的选择。在骨组织工程的应用中,生长因子可以调节细胞的增殖分化和细胞外基质的合成,这种作用主要通过早期自分泌和旁分泌的方式,提高成骨细胞的增殖速度和活性,显著增强骨组织的再生能力。
目前常见的生长因子包括胰岛素样生长因子、血小板衍生生长因子和骨形态发生蛋白等,其中以骨形态发生蛋白的研究和应用最广泛[32]。同样还有很多外源因素对骨组织工程的成功与否起着关键作用,目前的研究发现中药如淫羊藿苷等[33]。物理治疗技术的形成和发展过程,见图3。
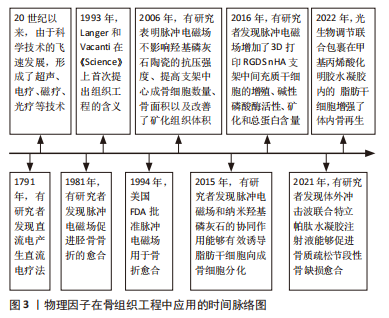
早在《黄帝内经》中已有药熨及按跷等物理因子治疗疾病的记载,随着现代科技的不断发展,不仅促进现代医学的发展,也使古老的物理疗法得以不断地形成和完善,形成了光疗、感应电、超声波及磁场等诸多物理因子疗法,很快应用于各种疾病。而在整个世界范围内,大段骨缺损的治疗仍未完善,自从1993年骨组织工程概念的提出,给患者带来了新的期盼,研究者们致力于提升骨组织工程在临床中的发展。近年来,国内外利用物理因子干预干细胞的调控促进其成骨分化以及在骨组织工程中的应用有了一定的成果。但其如何影响干细胞的分化则需要进一步的探讨,目前应用较多的物理因子主要有脉冲电磁场、光、冲击波、超声波以及力学刺激等。
2.2.1 超声 超声在临床上是一种常见的物理诊断和治疗手段。目前很多学者将目光聚集其在再生医学中的应用,一些体内外的实验都有涉及到低强度脉冲超声在干细胞定向分化中的作用[34]。低强度脉冲超声已被证明具有多种生物效应,包括促进组织愈合、血管生成和组织再生;抑制炎症和缓解疼痛;以及刺激细胞增殖和分化等[35],低强度脉冲超声已被报道可以促进间充质干细胞分化和骨折愈合[36],但生物物理机制尚不清楚,其机械刺激如何转化为化学刺激以及如何对干细胞进行调控需要进一步深入研究。
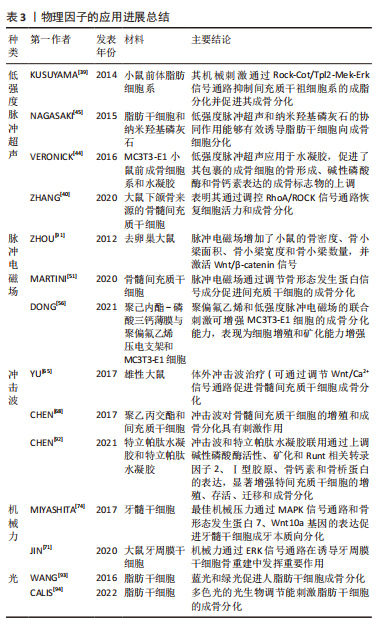
有研究显示,细胞通过整合素介导的机械感受器将组织中超声的机械信号转化为骨中的生化反应,导致环氧合酶2的产生,从而刺激骨折修复。在低强度脉冲超声的作用下,纳米机械力发生器通过调节机械敏感性瞬时受体电位melastatin 7、肌动蛋白细胞骨架和细胞内钙振荡,促进骨髓间充质干细胞的成骨分化和骨形成[37];相关研究表明,60和90 mW/cm2、30 min/d强度的超声剂量能够促进基质细胞衍生因子1(stromal cell-derived factor-1,SDF-1)表达,通过SDF-1/CXCR4信号通路促进牙周膜干细胞迁移,而Twist家族b HLH转录因子1可能是机械转导过程中的机械应力传感器[38];此外,在干细胞成骨分化的机制中,有关研究表明,低强度脉冲超声通过ROCK-Cot/Tpl2-MEK-ERK信号通路、RhoA/ROCK信号通路以及经典的Wnt/β-catenin信号通路[39-41],促进干细胞的成骨分化和骨组织再生以及新骨形成和种植体骨融合,但上述机制仍需进一步研究证实。此外,除了时间强度,占空比相对来说更为重要,关于超声不同占空比处理对间充质干细胞的生长和分化的影响,目前尚未有足够的报道。
低强度脉冲超声已被证明对骨折修复有效而广泛应用于临床诊断和治疗。相关研究将低强度脉冲超声与先进的3D打印技术相结合,其工作参数为1.5 MHz,20%占空比,150 mW/cm2,结果表明,3D打印支架在低强度脉冲超声作用下可显著促进间充质干细胞增殖、碱性磷酸酶活性、钙沉积和总蛋白含量的增高,从而使其在未来再生医学的应用中具有良好前景[42];采用1 MHz,占空比分别为20%和50%,强度为50 mW/cm2,辐照时间为10 min/d的低强度脉冲超声处理人肺泡骨源间充质干细胞,结果显示碱性磷酸酶显著增强,干细胞中CD29及CD44等的基因表达水平显著增高以及矿化结节数明显增加。这些结果表明,低强度脉冲超声可以增强人肺泡骨源间充质干细胞的细胞活力和成骨分化能力,可以作为临床应用的有效治疗方法[43]。
此外,超声产生的物理力可以用于物理变形胶原蛋白水凝胶,而封装在这些水凝胶中的活细胞则通过上调骨细胞分化关键标志物的基因表达来响应超声产生的力,结果显示碱性磷酸酶和骨钙素这两种公认的成骨细胞分化标志物的基因表达在包裹的成骨细胞中显著升高,同时其还能通过改变这些水凝胶的机械刚度来调节细胞对超声波的反应,这为骨缺损治疗提供了一个新的工程策略[44]。有研究表明,低强度脉冲超声刺激和纳米羟基磷灰石作为支架的组合显著增加了脂肪来源的干细胞的成骨分化,以及碱性磷酸酶活性、钙沉积及Runt相关转录因子2的表达大幅增加;将脂肪干细胞移植到小鼠颅骨缺损中,组合效应又促进了缺陷边缘的新骨形成。以上研究表明,低强度脉冲超声和纳米羟基磷灰石的协同作用能够有效诱导脂肪干细胞向成骨细胞分化,为脂肪干细胞的自体移植以及促进骨再生提供了新的治疗策略[45]。
就目前来看,低强度脉冲超声在骨科应用中常被用作肌肉骨骼组织修复的生物物理刺激,以增强组织再生。针对骨折的修复,它能够促进干细胞向损伤部位的募集,并在不同阶段利用其分化潜能引导其成骨或成软骨细胞谱系。然而,在骨质疏松的应用中存在争议,临床结果与临床前结果存在差异,这提示可能与穿透能量不足有关。另一方面,在应用于大段骨缺损中,很多研究者都证明了低强度脉冲超声促进干细胞成骨分化的积极作用以及在组织工程骨中促进干细胞的迁移、黏附和新骨的形成,但是其在临床上的治疗效果尚未有足够的报道。总之,超声是一种很有前途的机械刺激骨组织工程细胞的方法,应开展更多的研究,以确定最佳的超声参数,并为低强度脉冲超声与其他类型的支架材料和细胞类型的积极作用提供更多的证据。
2.2.2 脉冲电磁场 脉冲电磁场在骨科领域的应用已有至少三十年的历史,在骨不连、骨折和相关骨愈合问题的骨修复中发挥了重要作用[46-47]。脉冲电磁场目前被广泛应用于骨科领域以促进修复性成骨,大量研究证明其可以促进成骨[48-49]。骨形态发生蛋白属于转化生长因子β家族,被称为成骨分化的强诱导剂[50]。相关研究表明骨形态发生蛋白信号成分参与脉冲电磁场诱导成骨分化,以及增强了人间充质干细胞中骨形态发生蛋白2的活性[51];在有关体外研究中,脉冲电磁场通过上调AKT、MAPP激酶和RRAGA蛋白来促进mTOR通路的激活,刺激成骨分化[52];还有研究发现脉冲电磁场可通过激活转化生长因子β信号通路,刺激人骨髓间充质干细胞中miR21-5p的表达,介导其对骨代谢的影响,通过miR21-5p抑制重组蛋白Smad7的表达,从而激活年轻女性骨髓间充质干细胞中的该通路,进而控制Runt相关转录因子2的功能,促进其成骨[53]。
脉冲电磁场被认为是临床中促进骨再生的一种非侵入治疗方法[54]。相关研究利用3D打印的钛植入体和脉冲电磁场联合治疗(50 Hz,1 mT)可有效促进多孔钛表面骨质疏松衍生的骨髓间充质干细胞的活性和成骨分化,显著增强了骨再生和骨整合,这种新型的3D打印多孔钛和脉冲电磁场的联合疗法具有促进骨质疏松患者牙种植体或人工假体的骨再生和骨整合的潜力,在骨组织工程中具有巨大的应用前景[55]。有关研究表明,聚偏二氟乙烯涂层包被的聚己内酯-磷酸三钙支架与脉冲电磁场(50 Hz,0.6 mT,30 min/d)两者联合刺激可增强MC3T3-E1细胞的成骨分化能力,表现为细胞增殖和矿化能力增强,RUNT相关转录因子2、骨唾液酸蛋白、骨桥蛋白和骨钙素表达水平升高,这为电增强骨诱导提供了一种强有力的策略[56]。还有相关研究成功制备了基于化学和物理多重交联和相互作用的Alg-DA/Ac-β-CD/明胶水凝胶,证实脉冲电磁场不仅可以促进间充质干细胞的软骨分化,而且可以抑制软骨诱导过程中肥大标志物的表达,从而提高组织工程构建的质量,但是剂量的关系还需进一步研究[57]。
有体外实验利用电磁场(15 Hz,1 mT,4 h/d)联合水凝胶-羟基磷灰石-单体Ⅰ型胶原支架组织工程可促进兔骨软骨缺损修复,其分别通过激活PI3K/AKT/mTOR和Wnt1/LRP6/β-catenin通路促进骨髓间充质干细胞增殖和成软骨分化[58];有关研究表明,在施加的电磁场(5 V-1 Hz,0.62 mT)下,3D水凝胶负载牙根尖乳头干细胞通过增加牙本质涎磷蛋白、牙特异性细胞外基质蛋白、碱性磷酸酶和Ⅰ型胶原蛋白的活性来增强根尖乳头干细胞的活性,提高其成骨和成牙本质潜能,这种基于电磁场的细胞刺激方法为牙科组织工程提供了一种有前途的策略[59]。这些实验结果均显示了电磁场具有增强骨软骨修复、骨再生的巨大潜力。
脉冲电磁场可以促进早期和晚期成骨,增强骨髓间充质干细胞矿化,提高细胞活力,增强成骨细胞碱性磷酸酶的活性,并通过抑制破骨细胞的形成和成熟抑制骨吸收等,见图4。在与各种新型支架的联合疗法中,脉冲电磁场具有促进骨质疏松患者牙种植体和人工假体的骨再生和骨整合的潜力,并能有效促进构建体表面骨髓间充质干细胞的活性和成骨分化,这种效应归因于电磁场和支架之间的协同作用,可能刺激了机械转导途径,进而激活细胞间的生化信号,使其分化或增殖。这可能为大段骨缺损的治疗提供一种安全有效的选择,为患者带来了福音,在骨组织工程中具有巨大的应用前景。

2.2.3 冲击波 体外冲击波是一种特殊的声波,其作用于人体组织可产生空化、成骨以及镇痛效应,促进转化生长因子β、胰岛素生长因子1和血管内皮生长因子等生长因子的表达,诱导骨髓间充质干细胞向骨祖细胞分化,修复损伤组织[60]。此外,当其从皮肤向骨折部位传播时,冲击波的物理刺激导致骨折端微骨折,从而诱导微小骨痂形成,促进骨折愈合,因此被广泛运用于骨科[61]。体外冲击波疗法是指利用压缩气体产生机械性脉冲波,以脉冲方式冲击治疗部位的一种对组织损伤疗效显著的无创方法。近年来,冲击波疗法开始应用于骨不连的治疗,有效率为60%-90%[62]。目前,冲击波疗法常用于治疗许多骨科疾病,如足底筋膜炎、肘关节外上髁炎、肩部钙化性肌腱病和长骨骨折不愈合[63]。
有研究发现,冲击波可通过调节Wnt/Ca2+信号通路促进骨髓间充质干细胞成骨分化,从而改善骨关节炎大鼠的临床症状[64]; 有研究表明60 kPa、6 Hz、1 000 s、单次30 min的剂量能够诱导Wnt5a/Ca2+信号通路下游分子蛋白激酶C、磷脂酶C和CaMKⅡ蛋白表达水平的变化,表明适宜强度的冲击波通过激活Wnt5a/Ca2+信号通路诱导骨髓间充质干细胞的分化[65];此外,在骨化软骨交界处募集间充质干细胞是其增强骨修复的关键步骤。而转化生长因子β1和血管内皮生长因子积极参与间充质干细胞的募集和分化,它们的产生是由冲击波提供的物理刺激直接或间接促进的[66];另一项研究表明,冲击波疗法诱导三磷酸腺苷释放,并通过表达P2X7受体来诱导间充质干细胞的成骨分化[67]。
随着骨组织工程的不断发展,冲击波展现了其独特的优点。相关研究将冲击波(500 个脉冲,0.1 mJ/mm2,4 Hz)与局部注射特立帕肽水凝胶相结合,研究结果显示此组合通过上调碱性磷酸酶活性、矿化和Runt相关转录因子2、Ⅰ型胶原、骨钙素和骨桥蛋白的表达,在体外显著促进了骨质疏松症来源的骨髓间充质干细胞的增殖、活力、迁移和成骨分化,以及在体内显著改善节段性骨缺损的愈合;并可调节骨质疏松性骨折中不良的成骨微环境,具有促进脆性骨折愈合的潜力[68]。研究表明,冲击波(10 kV,500个脉冲)处理的骨髓间充质干细胞接种在生物可降解材料聚乙丙交酯支架上,结果显示成骨分化增强,结节形成增多,同时促进骨髓间充质干细胞向成骨前体细胞增殖和分化,从而促进骨再生[69]。由此可见,冲击波能够触发、诱导和放大细胞活化和迁移,联合支架能够显著改善骨修复,这将为骨组织工程提供新的研究思路。尽管如此,但仍需要进一步的随机试验来完善此领域的证据。
2.2.4 机械力 由细胞外基质、其他局部细胞类型和生化刺激组成的复杂微环境会影响干细胞自我更新和分化。除生化因素外,机械因素越来越被认为是干细胞行为和功能的关键调节因子,机械力在调节干细胞的命运和行为中发挥着重要作用[70],主要应用于牙齿正畸、牙缺损和牙槽骨改建等相关牙医学中。有关研究表明,力能够诱导牙周膜干细胞中TRPV4的激活,其通过ERK信号影响RANKL/骨保护素系统调控破骨细胞的分化、骨重塑和牙正畸[71];有研究表明在没有其他成骨诱导剂的情况下,模拟正畸力的机械刺激,如静态等轴应变,增加了成骨标志物骨桥蛋白和碱性磷酸酶的表达水平,诱导牙髓干细胞向成骨细胞分化[72]。
为了更好地了解机械应力对牙髓干细胞分化的影响,有研究发现牙髓干细胞在压应力下增加了碱性磷酸酶、牙本质基质蛋白1、骨形态发生蛋白2、牙本质涎磷蛋白和Ⅰ型胶原蛋白等成牙相关转录基因的表达,因此推断机械刺激可用于启动牙本质-牙髓复合体内部的修复机制[73];此外,机械压力还能通过MAPK、ERK1/2和p38信号通路以及骨形态发生蛋白7的表达促进牙髓干细胞的分化[74];研究还发现周期性张力应变的机械刺激诱导成牙细胞标志物牙本质涎磷蛋白、牙本质基质蛋白1、骨桥蛋白和骨唾液酸蛋白的表达。并证明牙髓干细胞的成牙分化是由NF-E2相关转录因子2(Nrf2)/HO-1通路介导[75]。尽管已知物理和机械因素在调控干细胞命运中发挥关键作用,但仍需要进一步研究来阐明干细胞对各种类型力的响应所涉及的详细分子机制和信号通路。
目前,机械力被认为是最容易操纵和控制的。相关研究将机械刺激引入掺有硅酸钙的鱼明胶甲基丙烯酸酯水凝胶中,结果表明负重支架施加生物力学刺激,能够使人牙周韧带细胞的黏附和增殖能力进一步提升至少5%,并且能够增强碱性磷酸酶、骨桥蛋白和骨钙素等成骨相关标志物的分泌[76]。相关研究将浸入边界法用于骨组织工程的祖细胞(人骨膜衍生细胞)上的流体诱导机械刺激,这些祖细胞附着在灌注生物反应器装置内的常规孔钛支架上,剪切流动条件下产生的机械性应力定位于细胞与其环境界面的特定区域,已被证明参与骨细胞对动态剪切流动条件的成骨反应,以及细胞外基质的重塑[77]。相关研究表明,从人下颌牙槽骨中收获的人肺泡骨来源的间充质干细胞用低流体动态剪切应力刺激10-60 min/d后的细胞的增殖能力和活力增加。
此外,低流体动态剪切应力条件还增加了骨涎蛋白、Runt相关转录因子2、Ⅰ型胶原蛋白、碱性磷酸酶、骨钙素和骨桥蛋白的基因表达。表明在适当的强度和时间下,低流体动态剪切应力可以有效和积极地调节人肺泡骨来源的间充质干细胞的增殖和分化[78]。上述研究结果表明,力学刺激在骨组织再生中起着至关重要的作用,包括动态静水压力、循环拉伸应变、机械压缩和循环单轴压缩应变等力学刺激可以促进干细胞的成牙本质向分化和成骨分化。它通过机械转导促进干细胞的黏附,增殖以及促进成骨分化。同时还可以通过影响支架的刚度、降解速率和溶胀能力,促进支架成骨和血管的生成能力,将骨组织工程带入新的阶段。因此这提示研究者们可以通过结合科学的不同方法来制备新型支架,使组织工程更接近临床应用。
2.2.5 光刺激 在过去的几年中,利用光生物调节来增强干细胞的增殖和诱导其分化已经引起越来越多的关注。光生物调节是利用低功率相干或非相干光源在可见光和近红外范围内实现光的应用。大量文献表明光生物调节疗法对干细胞的成骨分化有着积极作用,被广泛应用于再生医学和牙科医学中[79-81]。干细胞行为产生积极影响的最合适和有效的波长、光束类型、能量密度(通量)、照射模式和照射时间仍在不断地探索之中。有研究表明,在不同的光处理中,采用能量密度为0.4 J/cm2的635 nm半导体激光能够通过Akt信号的激活正向影响间充质干细胞的行为[82];相关研究通过使用660 nm波长的激光,促进间充质干细胞的增殖和成骨分化[37];此外,使用0.5和1.0 J/cm2的激光照射牙髓干细胞[83]、功率为40 mW、能量为0.56、1.96和5.04 J 的660 nm激光处理脂肪间充质干细胞[84],都表明了光生物调节疗法促进间充质干细胞的增殖和分化。虽然使用的参数有所不同,但均一致表明在与光生物调节疗法相关的成骨培养基中,促进了细胞的增殖和成骨分化,这可能代表了干细胞应用中的一种创新预处理。
此外,低强度激光治疗已被报道可改善细胞增殖和分化,具有缓解疼痛、舒张血管、促进组织愈合、细胞增殖、矿化和成骨诱导等优点[85]。大量的实验旨在研究不同能量强度的激光治疗对骨髓间充质干细胞增殖和成骨分化的影响[86-90],见表2。
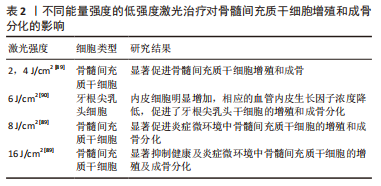
在低强度的激光治疗下,可以显著促进健康及炎症微环境中骨髓间充质干细胞增殖和成骨,而在高强度的激光治疗下显著抑制了健康及炎症微环境中骨髓间充质干细胞的增殖及成骨分化,提示研究者们这可能是因为这种高能量密度可以通过加热效应损伤细胞,这需要进一步的研究来证明;研究发现光刺激也具有一定的累积效应,多色光源(从接种后第3天开始,以92 mW/cm2的功率从距细胞10 cm处开始,每隔2 d施加5 min ,持续至第28天)光刺激可促进间充质干细胞的成骨分化[86]。以上的研究结果均证明了光疗在促进干细胞增殖分化中的积极影响。然而,就其细胞和分子效应而言,治疗性激光的机制仍不清楚,这提示研究者们需要更进一步的研究。
目前,有一些基础的体内外实验探讨了光在组织工程中的作用。基于支架的组织工程构建物和光生物调节相辅相成。在光生物调节疗法(660 nm,功率输出20 mW,光束面积0.028 cm2,能量密度5 J/cm2)的作用下,壳聚糖支架内生长的牙根尖乳头干细胞表现出增强的增殖和迁移能力,出现健康的牙髓组织,这表明光生物调节应用于壳聚糖水凝胶可以在未来通过细胞归巢途径改善牙髓再生[87];有研究表明,光刺激在不影响细胞增殖的同时,促进了间充质干细胞在丝素支架上成骨基因的表达,诱导其分化为成熟的成骨细胞,碱性磷酸酶的活性和支架的胶原和钙含量证实了这些结果。总之,包括生物有效波长的多色光在内的等离子弧光源可以作为一种有效的工具来诱导3D支架中的成骨分化和骨再生[86]。
有相关研究将GEL-MA水凝胶溶液(pH 7.0)以50 μL的体积缓慢注射到颅骨缺损处,结果表明载有脂肪干细胞的水凝胶通过UV-A(300- 500 nm,200 mW/cm2)的干预,能刺激脂肪干细胞的成骨分化,以及对骨再生具有良好的影响[88]。大多数研究均表明光对细胞增殖和分化有积极作用,不同参数的具体影响不尽相同。光生物调节通过可见光或近红外光,引起细胞内的物理或化学变化。尽管发现了一些激光生物刺激的机制,但还不完全清楚,可能通过能量转换将光能转换为代谢能来改善细胞的活性,这些结果取决于能量密度、功率输出、辐射频率、光源类型、细胞或培养基类型等因素。在当前的研究中不乏相反的结果,因此,还需大量的研究确定合适的参数,以获得具有对比性和更加准确的结果。光生物调节仍然是具有生物刺激骨组织以及增加细胞增殖和分化一种有用的再生治疗工具。

| [1] LEE FH, SHEN PC, JOU IM, et al. A Population-based 16-year study on the risk factors of surgical site infection in patients after bone grafting. Medicine. 2015;94(47):e2034. [2] QI J, YU T, HU B, et al. Current biomaterial-based bone tissue engineering and translational medicine. Int J Mol Sci. 2021;22(19):10233. [3] PINA S, OLIVEIRA JM, REIS RL. Natural-based nanocomposites for bone tissue engineering and regenerative medicine: a review. Adv Mater. 2015; 27(7):1143-1169. [4] LOU S, DUAN Y, NIE H, et al. Mesenchymal stem cells: Biological characteristics and application in disease therapy. Biochimie. 2021;185:9-21. [5] ZAKRZEWSKI W, DOBRZYŃSKI M, SZYMONOWICZ M, et al. Stem cells: past, present, and future. Stem Cell Res Ther. 2019;10(1):68. [6] MAZINI L, ROCHETTE L, AMINE M, et al. Regenerative capacity of adipose derived stem cells (ADSCs), comparison with mesenchymal stem cells (MSCs). Int J Mol Sci. 2019;20(10):2523. [7] MAZINI L, ROCHETTE L, AMINE M, et al. ZEB1 mediates bone marrow mesenchymal stem cell osteogenic differentiation partly via Wnt/β-catenin signaling. Front Mol Biosci. 2021;8:682728. [8] 周春梅,李淑慧,温齐古丽·乃库力,等.Notch和Wnt信号通路在自体骨髓间充质干细胞复合富血小板纤维蛋白修复兔牙槽骨缺损中的表达[J].华西口腔医学杂志,2016,34(2):130-135. [9] 张伟丽,罗敏,彭江,等. BMP-2/Smads信号通路促进骨代谢失衡大鼠骨髓间充质干细胞的成骨分化[J].中华老年骨科与康复电子杂志, 2021,7(2):65-72. [10] 青薇,代彦君,黄丽娟,等.辛伐他汀缓释微球影响骨组织工程成骨性能的体外实验[J].中国组织工程研究,2019,23(2):239-244. [11] BOSE S, BANERJEE D, VU AA. Ginger and garlic extracts enhance osteogenesis in 3d printed calcium phosphate bone scaffolds with bimodal pore distribution. ACS Appl Mater Interfaces. 2022;14(11): 12964-12975. [12] VU AA, BOSE S. Effects of vitamin D3 release from 3D printed calcium phosphate scaffolds on osteoblast and osteoclast cell proliferation for bone tissue engineering. RSC Adv. 2019;9(60):34847-34853. [13] GE S, ZHAO N, WANG L, et al. Bone repair by periodontal ligament stem cell-seeded nanohydroxyapatite-chitosan scaffold. Int J Nanomedicine. 2012;7: 5405-5414. [14] WANG L, ZHANG C, LI C, et al. Injectable calcium phosphate with hydrogel fibers encapsulating induced pluripotent, dental pulp and bone marrow stem cells for bone repair. Mater Sci Eng C Mater Biol Appl. 2016;69: 1125-1136. [15] TSIKLIN IL, SHABUNIN AV, KOLSANOV AV, et al. In vivo bone tissue engineering strategies: advances and prospects. Polymers (Basel). 2022; 14(15):3222. [16] XU C, DAI G, HONG Y. Recent advances in high-strength and elastic hydrogels for 3D printing in biomedical applications. Acta Biomater. 2019;95:50-59. [17] DOROZHKIN SV, EPPLE M. Biological and medical significance of calcium phosphates. Angew Chem Int Ed Engl. 2002;41(17):3130-3146. [18] SONG G, HABIBOVIC P, BAO C, et al. The homing of bone marrow MSCs to non-osseous sites for ectopic bone formation induced by osteoinductive calcium phosphate. Biomaterials. 2013;34(9):2167-2176. [19] GHASSEMI T, SHAHROODI A, EBRAHIMZADEH M H, et al. Current concepts in scaffolding for bone tissue engineering. Arch Bone Jt Surg. 2018;6(2):90-99. [20] JAHAN K, TABRIZIAN M. Composite biopolymers for bone regeneration enhancement in bony defects. Biomater Sci. 2016;4(1):25-39. [21] SUN W, GREGORY DA, TOMEH MA, et al. Silk fibroin as a functional biomaterial for tissue engineering. Int J Mol Sci. 2021;22(3):1499. [22] 张文贤,周红,刘涛,等.健骨益痹方含药血清对骨髓间充质干细胞体外增殖及成骨分化的影响[J].时珍国医国药,2019,30(7):1584-1586. [23] WANG Y, GAO M, WANG D, et al. Nanoscale 3d bioprinting for osseous tissue manufacturing. Int J Nanomedicine. 2020;15:215-226. [24] 黄波,陈明学,彭礼庆,等.可注射性甲基丙烯酸酐改性明胶/软骨源性基质微粒复合水凝胶支架的制备及生物相容性[J].中国组织工程研究,2022,26(16):2480-2486. [25] SAMAVEDI S, WHITTINGTON AR, GOLDSTEIN AS. Calcium phosphate ceramics in bone tissue engineering: a review of properties and their influence on cell behavior. Acta Biomater. 2013;9(9):8037-8045. [26] COCKERILL I, SU Y, SINHA S, et al. Porous zinc scaffolds for bone tissue engineering applications: a novel additive manufacturing and casting approach. Mater Sci Eng C Mater Biol Appl. 2020;110:110738. [27] NATARAJAN D, YE Z, WANG L, et al. Rare earth smart nanomaterials for bone tissue engineering and implantology: advances, challenges, and prospects. Bioeng Transl Med. 2021;7(1):e10262. [28] 丁鑫鑫,周延民,相星辰,等.壳聚糖复合材料在骨组织工程中的研究进展[J].华西口腔医学杂志,2018,36(4):441-446. [29] HAO Z, LI H, WANG Y, et al. Supramolecular peptide nanofiber hydrogels for bone tissue engineering: from multihierarchical fabrications to comprehensive applications. Adv Sci (Weinh). 2022;9(11):e2103820. [30] WANG L, ZHANG C, LI C, et al. Injectable calcium phosphate with hydrogel fibers encapsulating induced pluripotent, dental pulp and bone marrow stem cells for bone repair. Mater Sci Eng C Mater Biol Appl. 2016;69: 1125-1136. [31] NOORI A, ASHRAFI SJ, VAEZ-GHAEMI R, et al. A review of fibrin and fibrin composites for bone tissue engineering. Int J Nanomedicine. 2017;12: 4937-4961. [32] 汪鎏,宋东哲,黄定明.骨形态发生蛋白9调控干细胞分化与骨再生[J].中国组织工程研究,2021,25(19):3064-3070. [33] 陈震东,高辉,徐房添.淫羊藿苷促进间充质干细胞成骨分化:为治疗骨缺损提供一个良好的方向[J].中国组织工程研究,2016,20(50):7594-7600. [34] TAN Y, GUO Y, REED-MALDONADO A, et al. Low-intensity pulsed ultrasound stimulates proliferation of stem/progenitor cells: what we need to know to translate basic science research into clinical applications. Asian J Androl. 2021;23(6):602-610. [35] LIANG Q, DU L, ZHANG R, et al. Stromal cell-derived factor-1/Exendin-4 cotherapy facilitates the proliferation, migration and osteogenic differentiation of human periodontal ligament stem cells in vitro and promotes periodontal bone regeneration in vivo. Cell Prolif. 2021;54(3): e12997. [36] IWASHINA T, MOCHIDA J, MIYAZAKI T, et al. Low-intensity pulsed ultrasound stimulates cell proliferation and proteoglycan production in rabbit intervertebral disc cells cultured in alginate. Biomaterials. 2006;27(3): 354-361. [37] YAO H, ZHANG L, YAN S, et al. Low-intensity pulsed ultrasound/nanomechanical force generators enhance osteogenesis of BMSCs through microfilaments and TRPM7. J Nanobiotechnol. 2022;20(1):378. [38] WANG Y, LI J, QIU Y, et al. Low‑intensity pulsed ultrasound promotes periodontal ligament stem cell migration through TWIST1‑mediated SDF‑1 expression. Int J Mol Med. 2018;42(1):322-330. [39] KUSUYAMA J, BANDOW K, SHAMOTO M, et al. Low intensity pulsed ultrasound (LIPUS) influences the multilineage differentiation of mesenchymal stem and progenitor cell lines through ROCK-Cot/Tpl2-MEK-ERK signaling pathway. J Biol Chem. 2014;289(15):10330-10344. [40] ZHANG R, WANG Z, ZHU G, et al. Low-intensity pulsed ultrasound modulates rhoa/rock signaling of rat mandibular bone marrow mesenchymal stem cells to rescue their damaged cytoskeletal organization and cell biological function induced by radiation. Stem Cells Int. 2020; 2020:8863577. [41] HUA Z, LI S, LIU Q, et al. Low-Intensity pulsed ultrasound promotes osteogenic potential of iPSC-derived MSCs but fails to simplify the iPSC-EB-MSC differentiation process. Front Bioeng Biotechnol. 2022;10:841778. [42] ZHOU X, CASTRO N J, ZHU W, et al. Improved human bone marrow mesenchymal stem cell osteogenesis in 3D bioprinted tissue scaffolds with low intensity pulsed ultrasound stimulation. Sci Rep. 2016;6:32876. [43] LIM K, KIM J, SEONWOO H, et al. In vitro effects of low-intensity pulsed ultrasound stimulation on the osteogenic differentiation of human alveolar bone-derived mesenchymal stem cells for tooth tissue engineering. Biomed Res Int. 2013;2013:269724. [44] VERONICK J, ASSANAH F, NAIR LS, et al. The effect of acoustic radiation force on osteoblasts in cell/hydrogel constructs for bone repair. Exp Biol Med (Maywood). 2016;241(10):1149-1156. [45] NAGASAKI R, MUKUDAI Y, YOSHIZAWA Y, et al. A combination of low-intensity pulsed ultrasound and nanohydroxyapatite concordantly enhances osteogenesis of adipose-derived stem cells from buccal fat pad. Cell Med. 2015;7(3):123-131. [46] WALKER NA, DENEGAR CR, PREISCHE J. Low-intensity pulsed ultrasound and pulsed electromagnetic field in the treatment of tibial fractures: a systematic review. J Athl Train. 2007;42(4):530-535. [47] MIDURA RJ, IBIWOYE MO, POWELL KA, et al. Pulsed electromagnetic field treatments enhance the healing of fibular osteotomies. J Orthop Res. 2005;23(5):1035-1046. [48] HANNEMANN PFW, ESSERS BAB, SCHOTS JPM, et al. Functional outcome and cost-effectiveness of pulsed electromagnetic fields in the treatment of acute scaphoid fractures: a cost-utility analysis. BMC Musculoskelet Disord. 2015;16:84. [49] SHI HF, XIONG J, CHEN YX, et al. Early application of pulsed electromagnetic field in the treatment of postoperative delayed union of long-bone fractures: a prospective randomized controlled study. BMC Musculoskelet Disord. 2013;14:35. [50] RAHMAN MS, AKHTAR N, JAMIL HM, et al. TGF-β/BMP signaling and other molecular events: regulation of osteoblastogenesis and bone formation. Bone Res. 2015;3:15005. [51] MARTINI F, PELLATI A, MAZZONI E, et al. Bone morphogenetic protein-2 signaling in the osteogenic differentiation of human bone marrow mesenchymal stem cells induced by pulsed electromagnetic fields. Int J Mol Sci. 2020;21(6):2104. [52] FERRONI L, GARDIN C, DOLKART O, et al. Pulsed electromagnetic fields increase osteogenetic commitment of MSCs via the mTOR pathway in TNF-α mediated inflammatory conditions: an in-vitro study. Sci Rep. 2018; 8(1):5108. [53] SELVAMURUGAN N, HE Z, RIFKIN D, et al. Pulsed electromagnetic field regulates microRNA 21 expression to activate TGF- β signaling in human bone marrow stromal cells to enhance osteoblast differentiation. Stem Cells Int. 2017;2017:2450327. [54] LIANG H, LIU X, PI Y, et al. 3D-Printed β-tricalcium phosphate scaffold combined with a pulse electromagnetic field promotes the repair of skull defects in rats. ACS Biomater Sci Eng. 2019;5(10):5359-5367. [55] YE M, LIU W, YAN L, et al. 3D‑printed Ti6Al4V scaffolds combined with pulse electromagnetic fields enhance osseointegration in osteoporosis. Mol Med Rep. 2021;23(6):410. [56] DONG Y, SURYANI L, ZHOU X, et al. Synergistic effect of pvdf-coated pcl-tcp scaffolds and pulsed electromagnetic field on osteogenesis. Int J Mol Sci. 2021;22(12):6438. [57] LI Y, LI L, LI Y, et al. Enhancing cartilage repair with optimized supramolecular hydrogel-based scaffold and pulsed electromagnetic field. Bioact Mater. 2022;22:312-324. [58] YAN J, LIU C, TU C, et al. Hydrogel-hydroxyapatite-monomeric collagen type-I scaffold with low-frequency electromagnetic field treatment enhances osteochondral repair in rabbits. Stem Cell Res Ther. 2021;12(1):572. [59] DUTTA SD, BIN J, GANGULY K, et al. Electromagnetic field-assisted cell-laden 3D printed poloxamer-407 hydrogel for enhanced osteogenesis. RSC Adv. 2021;11(33):20342-20354. [60] ZHANG C, HUANG H, YANG L, et al. Extracorporeal shock wave therapy for pain relief after arthroscopic treatment of osteochondral lesions of talus. J Foot Ankle Surg. 2020;59(1):190-194. [61] AUERSPERG V, TRIEB K. Extracorporeal shock wave therapy: an update. EFORT Open Rev. 2020;5(10):584-592. [62] SANSONE V, RAVIER D, PASCALE V, et al. Extracorporeal shockwave therapy in the treatment of nonunion in long bones: a systematic review and meta-analysis. J Clin Med. 2022;11(7):1977. [63] ROMEO P, LAVANGA V, PAGANI D, et al. Extracorporeal shock wave therapy in musculoskeletal disorders: a review. Med Princ Pract. 2014;23(1):7-13. [64] ZHOU J, HE H, YANG L, et al. Effects of pulsed electromagnetic fields on bone mass and Wnt/β-catenin signaling pathway in ovariectomized rats. Arch Med Res. 2012;43(4):274-282. [65] YU L, LIU S, ZHAO Z, et al. Extracorporeal Shock Wave Rebuilt Subchondral Bone In Vivo and Activated Wnt5a/Ca2+ Signaling In Vitro. Biomed Res Int. 2017;2017:1404650. [66] CHEN YJ, WURTZ T, WANG C J, et al. Recruitment of mesenchymal stem cells and expression of TGF-beta 1 and VEGF in the early stage of shock wave-promoted bone regeneration of segmental defect in rats. J Orthop Res. 2004;22(3):526-534. [67] SUN D, JUNGER WG, YUAN C, et al. Shockwaves induce osteogenic differentiation of human mesenchymal stem cells through ATP release and activation of P2X7 receptors. Stem Cells. 2013;31(6):1170-1180. [68] CHEN Q, XIA C, SHI B, et al. Extracorporeal shock wave combined with teriparatide-loaded hydrogel injection promotes segmental bone defects healing in osteoporosis. Tissue Eng Regen Med. 2021;18(6):1021-1033. [69] LEE JJ, NG HY, LIN YH, et al. The synergistic effect of cyclic tensile force and periodontal ligament cell-laden calcium silicate/gelatin methacrylate auxetic hydrogel scaffolds for bone regeneration. Cells. 2022;11(13):2069. [70] VINING KH, MOONEY DJ. Mechanical forces direct stem cell behaviour in development and regeneration. Nature Reviews. Nat Rev Mol Cell Biol. 2017;18(12):728-742. [71] JIN S, HE D, WANG Y, et al. Mechanical force modulates periodontal ligament stem cell characteristics during bone remodelling via TRPV4. Cell Prolif. 2020;53(10):e12912. [72] TABATABAEI FS, JAZAYERI M, GHAHARI P, et al. Effects of equiaxial strain on the differentiation of dental pulp stem cells without using biochemical reagents. Mol Cell Biomech. 2014;11(3):209-220. [73] YANG H, SHU YX, WANG LY, et al. Effect of cyclic uniaxial compressive stress on human dental pulp cells in vitro. Connect Tissue Res. 2018;59(3): 255-262. [74] MIYASHITA S, AHMED NEMB, MURAKAMI M, et al. Mechanical forces induce odontoblastic differentiation of mesenchymal stem cells on three-dimensional biomimetic scaffolds. J Tissue Eng Regen Med. 2017;11(2): 434-446. [75] LI G, FU N, YANG X, et al. Mechanical compressive force inhibits adipogenesis of adipose stem cells. Cell Prolif. 2013;46(5):586-594. [76] LEE J J, NG H Y, LIN Y H, et al. The synergistic effect of cyclic tensile force and periodontal ligament cell-laden calcium silicate/gelatin methacrylate auxetic hydrogel scaffolds for bone regeneration. Cells. 2022;11(13):2069. [77] GUYOT Y, SMEETS B, ODENTHAL T, et al. Immersed Boundary Models for Quantifying Flow-Induced Mechanical Stimuli on Stem Cells Seeded on 3D scaffolds in perfusion bioreactors. PLoS Comput Biol. 2016;12(9):e1005108. [78] LIM KT, KIM J, SEONWOO H, et al. Enhanced osteogenesis of human alveolar bone-derived mesenchymal stem cells for tooth tissue engineering using fluid shear stress in a rocking culture method. Tissue Eng Part C Methods. 2013;19(2):128-145. [79] HAMBLIN MR. Photobiomodulation or low-level laser therapy. J Biophotonics. 2016;9(11-12):1122-1124. [80] ETEMADI A, FAGHIH A, CHINIFORUSH N. Effects of Photobiomodulation therapy with various laser wavelengths on proliferation of human periodontal ligament mesenchymal stem cells. Photochem Photobiol. 2022;98(5):1182-1189. [81] REN C, MCGRATH C, JIN L, et al. The effectiveness of low-level laser therapy as an adjunct to non-surgical periodontal treatment: a meta-analysis. J Periodontal Res. 2017;52(1):8-20. [82] TANI A, CHELLINI F, GIANNELLI M, et al. Red (635 nm), near-infrared (808 nm) and violet-blue (405 nm) photobiomodulation potentiality on human osteoblasts and mesenchymal stromal cells: a morphological and molecular in vitro study. Int J Mol Sci. 2018;19(7):1946. [83] ZACCARA IM, GINANI F, MOTA-FILHO HG, et al. Effect of low-level laser irradiation on proliferation and viability of human dental pulp stem cells. Lasers Med Sci. 2015;30(9):2259-2264. [84] DE ANDRADE ALM, LUNA GF, BRASSOLATTI P, et al. Photobiomodulation effect on the proliferation of adipose tissue mesenchymal stem cells. Lasers Med Sci. 2019;34(4):677-683. [85] CARROLL JD, MILWARD MR, COOPER PR, et al. Developments in low level light therapy (LLLT) for dentistry. Dent Mater. 2014;30(5):465-475. [86] ÇAKMAK AS, ÇAKMAK S, VATANSEVER HS, et al. Photostimulation of osteogenic differentiation on silk scaffolds by plasma arc light source. Lasers Med Sci. 2018;33(4):785-794. [87] MOREIRA MS, SARRA G, CARVALHO GL, et al. Physical and biological properties of a chitosan hydrogel scaffold associated to photobiomodulation therapy for dental pulp regeneration: an in vitro and in vivo study. Biomed Res Int. 2021;2021:6684667. [88] FEKRAZAD R, ASEFI S, ALLAHDADI M, et al. Effect of photobiomodulation on mesenchymal stem cells. Photomed Laser Surg. 2016;34(11):533-542. [89] WANG L, WU F, LIU C, et al. Low-level laser irradiation modulates the proliferation and the osteogenic differentiation of bone marrow mesenchymal stem cells under healthy and inflammatory condition. Lasers Med Sci. 2019;34(1):169-178. [90] GUTIÉRREZ D, ROUABHIA M, ORTIZ J, et al. Low-level laser irradiation promotes proliferation and differentiation on apical papilla stem cells. J Lasers Med Sci. 2021;12:e75. [91] ZHOU J, HE H, YANG L, et al. Effects of pulsed electromagnetic fields on bone mass and Wnt/β-catenin signaling pathway in ovariectomized rats. Arch Med Res. 2012;43(4):274-282. [92] CHEN Q, XIA C, SHI B, et al. Extracorporeal shock wave combined with teriparatide-loaded hydrogel injection promotes segmental bone defects healing in osteoporosis. Tissue Eng Regen Med. 2021;18(6):1021-1033. [93] WANG Y, HUANG YY, WANG Y, et al. Photobiomodulation (blue and green light) encourages osteoblastic-differentiation of human adipose-derived stem cells: role of intracellular calcium and light-gated ion channels. Sci Rep. 2016;6:33719. [94] CALIS M, IRMAK G, DEMIRTAS TT, et al. Photobiomodulation combined with adipose-derived stem cells encapsulated in methacrylated gelatin hydrogels enhances in vivo bone regeneration. Lasers Med Sci. 2022;37(1):595-606. |
| [1] | 潘小龙, 樊飞燕, 应春苗, 刘飞祥, 张运克. 中药抑制间充质干细胞衰老的作用及机制[J]. 中国组织工程研究, 2024, 28(7): 1091-1098. |
| [2] | 徐灿丽, 何文星, 汪 磊, 吴芳婷, 王佳慧, 段雪琳, 赵铁建, 赵 斌, 郑 洋. 肝脏类器官研究的文献计量学分析[J]. 中国组织工程研究, 2024, 28(7): 1099-1104. |
| [3] | 刘瀚峰, 王晶晶, 余云生. 人造外泌体治疗心肌梗死:应用现状及前景[J]. 中国组织工程研究, 2024, 28(7): 1118-1123. |
| [4] | 马树微, 何 生, 韩 冰, 张缭云. 间充质干细胞来源外泌体治疗动物急性肝衰竭的Meta分析[J]. 中国组织工程研究, 2024, 28(7): 1137-1142. |
| [5] | 孙宇康, 宋丽娟, 温春丽, 丁智斌, 田 昊, 马 东, 马存根, 翟晓艳. 基于Web of Science近十年干细胞治疗心肌梗死的可视化分析[J]. 中国组织工程研究, 2024, 28(7): 1143-1148. |
| [6] | 冯睿钦, 韩 娜, 张 蒙, 谷馨怡, 张丰识. 1%富血小板血浆联合骨髓间充质干细胞促进周围神经损伤的修复[J]. 中国组织工程研究, 2024, 28(7): 985-992. |
| [7] | 王 雯, 郑芃芃, 孟浩浩, 刘 浩, 袁长永. 过表达Sema3A促进牙髓干细胞和MC3T3-E1的成骨分化[J]. 中国组织工程研究, 2024, 28(7): 993-999. |
| [8] | 邱晓燕, 李碧欣, 黎敬弟, 范垂钦, 马 廉, 王鸿武. MAFA-PDX1过表达慢病毒感染人脐带间充质干细胞向胰岛素分泌细胞的分化[J]. 中国组织工程研究, 2024, 28(7): 1000-1006. |
| [9] | 刘麒薇, 张俊辉, 杨 袁, 王金娟. 脐带间充质干细胞治疗多囊卵巢综合征的作用及机制[J]. 中国组织工程研究, 2024, 28(7): 1015-1020. |
| [10] | 张克凡, 石 辉. 细胞因子治疗骨关节炎的研究现状及应用前景[J]. 中国组织工程研究, 2024, 28(6): 961-967. |
| [11] | 韦沅汛, 陈 锋, 林宗汉, 张 驰, 潘成镇, 韦宗波. Notch信号通路与骨质疏松症及中医药防治[J]. 中国组织工程研究, 2024, 28(4): 587-593. |
| [12] | 杨雨晴, 陈志宇. 早期短暂M1巨噬细胞在骨组织工程中的作用及应用[J]. 中国组织工程研究, 2024, 28(4): 594-601. |
| [13] | 周世博, 关健斌, 俞 兴, 赵 赫, 杨永栋, 刘 涛. 股骨骨缺损动物模型制备现状及特点[J]. 中国组织工程研究, 2024, 28(4): 633-638. |
| [14] | 吴 天, 赵 越, 胡 蓉. 携双抗体纳米微泡对卵巢癌细胞增殖活性的影响[J]. 中国组织工程研究, 2024, 28(3): 341-346. |
| [15] | 戴 京, 刘沙沙, 沈明敬. 负载外泌体的可注射水凝胶修复种植体周围骨缺损[J]. 中国组织工程研究, 2024, 28(3): 347-354. |
骨组织工程中的核心三要素是生物支架、活性因子和种子细胞。而其中种子细胞的选择在骨组织工程的应用至关重要。间充质干细胞是从骨髓、脂肪组织、牙体组织、羊膜、胎盘、脐带和脐血等多种组织中分离得到的具有自我更新和多向分化潜能的干细胞,在一定条件下,间充质干细胞可以分化为多种器官或组织,如骨、脂肪、肌肉、神经元、心肌细胞及肝细胞等[4]。干细胞的成骨分化是一个复杂的过程,受物理、化学及生物等因素的影响,由多种调控因子和蛋白质共同调控,因此,如何促进、诱导干细胞的增殖和分化以达到最佳的骨缺损修复是不断深入研究的热点。
物理因子疗法简称理疗,在现代医学中是指应用自然界和人工各种物理因子如声、光、电、磁、冷、热、水、机械和放射能等因素作用于人体,以预防和治疗疾病。理疗的常用方法包括:声疗(超声波等)、光疗(红外线、紫外线、激光等)、电疗(直流电、低频、中频、高频电疗法等)、水疗(水浴、中药熏蒸等)、冷热疗(热敷、蜡疗等)、磁疗和压力疗法等,具有诸多作用包括镇痛、消炎、促进骨折愈合、软化瘢痕及刺激神经肌肉等。近年来,物理因子疗法广泛运用于人们的生活里,同时也在不断地开拓新的领域,国内外许多研究表明物理因子对干细胞的分化、调控具有一定的优势,通过与骨组织工程的结合应用能够进一步促进骨缺损的修复,文章将对此方面的研究进展进行综述。 中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1.1 检索人及检索时间 第一作者在2022年11月进行检索。
1.1.2 检索文献时限 检索时间起止时间2003年1月至2022年11月。
1.1.3 检索数据库 中国知网、PubMed和Web of Science数据库。
1.1.4 检索词 英文检索词为“stem cell,osteoporosis,osteogenic differentiation,Electromagnetic Fields,Ultrasound,shock wave,low-level laser therapy,Mechanical force,Bone Tissue Engineering”;中文检索词为“干细胞,骨缺损,成骨分化,电磁场,超声,冲击波,低强度激光,机械力,骨组织工程”。
1.1.5 检索文献类型 研究原著、综述、述评、病例报告和荟萃分析。
1.1.6 手工检索情况 无。
1.1.7 检索策略 以PubMed数据库检索策略为例,见图1。

1.2 入选标准
1.2.1 纳入标准 与研究主题相关的文献。
1.2.2 排除标准 观点陈旧、研究内容重复的文献。
1.3 文献质量评估和数据的提取 初步检索得到文献453篇,其中英文文献428篇,中文文献25篇,排除与研究相关性低、研究内容陈旧及重复性文献,最终纳入94篇符合标准的文献,其中来自中国知网数据库的文献8篇,来自PubMed 数据库的文献64篇,来自Web of Science 数据库的文献22篇;中文文献8篇,英文文献86篇。见图2。
 作为一种非侵入和非接触式治疗,物理因子在促进干细胞成骨分化中有着积极的影响:促进细胞的黏附、干细胞的成骨分化以及血管的生成。以上的种种研究均表明物理因子作为一种辅助疗法为骨组织工程的发展开辟了新的道路。但目前物理因子与干细胞的结合仅仅在起步阶段,剂量时间强度的运用还不够精准,调控机制通路未完全阐明,无法准确控制其分化以及对人体暴露的安全性等。尽管骨组织工程的本质在以往得到了深入研究,但它们的理想组合仍然存在争议。因此,理想的支架材料在当前仍是一个值得探讨的问题。目前,流行的趋势是通过组合使用各种具有生物相容性的支架材料来达到疗效的最大化。
作为一种非侵入和非接触式治疗,物理因子在促进干细胞成骨分化中有着积极的影响:促进细胞的黏附、干细胞的成骨分化以及血管的生成。以上的种种研究均表明物理因子作为一种辅助疗法为骨组织工程的发展开辟了新的道路。但目前物理因子与干细胞的结合仅仅在起步阶段,剂量时间强度的运用还不够精准,调控机制通路未完全阐明,无法准确控制其分化以及对人体暴露的安全性等。尽管骨组织工程的本质在以往得到了深入研究,但它们的理想组合仍然存在争议。因此,理想的支架材料在当前仍是一个值得探讨的问题。目前,流行的趋势是通过组合使用各种具有生物相容性的支架材料来达到疗效的最大化。3.2 作者综述区别于他人他篇的特点 物理因子应用于骨组织工程中已经引起了越来越多的学者关注。但大多数的综述都将重点放在间充质干细胞的成骨分化上,未能联合骨组织工程。研究者们在作用机制以及联合新型支架的合适剂量方面总结了最新的研究进展。并且骨骼是一种高度血管化的组织,其发育、成熟、重塑和再生取决于血管供应的严格调节。在运用骨组织工程创造骨骼替代物时,也必须考虑到血管的再生情况。除了以往血管生成生长因子的使用,在这篇综述中,研究者们还介绍了各种物理因子作为辅助疗法在促进血管生成中的积极作用。
3.3 综述的局限性 文章简述了物理因子对干细胞的调控以及在骨组织工程中的应用现状,着重总结了各类物理因子如超声、脉冲电磁场、光生物调节以及机械力等促进干细胞成骨分化的作用及机制,并联合了骨组织工程,为骨缺损的治疗提供了新的思路和方法。然而现阶段研究中将物理因子应用于组织工程中的研究较少,故于此方面的综述较为局限,具体的剂量仍是未知数。
3.4 综述的重要意义 在骨缺损的治疗中,常常难以达到人们期望的治疗效果。由于肌肉骨骼系统疾病的医疗费用负担日益增加,以及药物和生长因子的不良反应,人们越来越需要安全、经济和简单的治疗方法,物理因子作为一种无创、非接触的辅助疗法应运而生。在骨组织工程的应用里,支架的机械性能、生物相容性以及血管化在骨组织再生中具有重要作用。物理因子可以通过上调碱性磷酸酶活性、骨钙素和骨桥蛋白的表达,显著增强特间充质干细胞的增殖、存活、迁移、成骨分化以及血管的生成而在骨科以及口腔医学中大放异彩。此外,它在骨关节炎和骨质疏松的骨缺损模型中,可以改善骨的质量、增加小鼠的骨密度、骨小梁面积、骨小梁宽度和骨小梁数量。所以,将物理因子与骨组织工程结合到一起,是一种具有潜力的新策略。
3.5 课题专家组对未来的建议 目前,研究者们要做的就是最佳参数的设置、相关的分子和细胞机制以及阐明人体暴露的安全性。总之,物理因子作为一种理想的辅助疗法,在与各种生物材料结合在用于骨组织工程方面具有巨大的潜力,利用各种物理因子来提高生物材料的性能将是骨缺损治疗的有效策略。因此,在未来的研究中,学者们如何把物理因子和骨组织工程结合的疗效最大化有待更进一步的努力,而这也为骨缺损的综合治疗提供了新的思路和挑战。 中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
 #br#
#br#
文题释义:
干细胞成骨分化:干细胞存在于多种成体组织中,包括皮肤、肌肉、骨髓和脂肪,在体外不同条件下来诱导分化成骨、软骨及其他结缔组织,因其来源广泛、分离无创伤、较低的免疫原性及生长周期短等特点,是组织工程种子细胞的重要来源。骨组织工程:是指用各种材料制备一种具有优良生物相容性、在人体内可逐步被降解吸收的细胞支架,这种仿生支架能像天然骨一样为细胞提供生存所需的三维空间,使细胞获得足够的营养物质,并进行气体交换,排除废物,细胞能够在支架上按预制形态生长,最终修复骨缺损。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
大段骨缺损一直是骨科临床上未能解决的难题。虽然骨移植可以成为一种合适有效的治疗选择,但骨供体并不广泛,且移植的成功率与高感染率或疾病传播率有关[1]。骨组织工程是由生物工程、细胞移植和材料科学组成的21世纪生命科学领域的前沿交叉学科,旨在构建修复损伤骨的生物替代物[2]。骨组织工程作为一种新型的引导骨组织再生的方法成为当前的研究热点。近年来,骨组织工程为治疗骨缺损的相关研究方兴未艾,除了在天然骨的基础上对其进行优化外,也开发了各式各样的骨支架,呈现出良好的生物相容性、骨传导率以及更优力学性能的特点[3]。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||