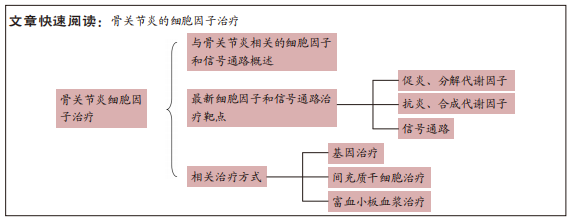
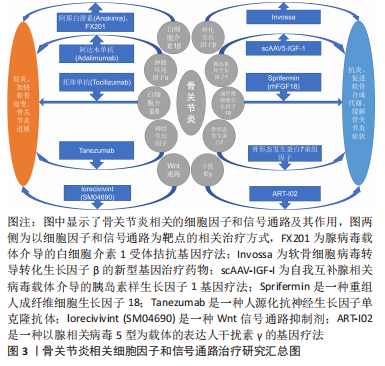
[1] HUNTER DJ, MARCH L, CHEW M. Osteoarthritis in 2020 and beyond: a Lancet Commission. Lancet. 2020;396(10264):1711-1712.
[2] BERENBAUM F, WALLACE IJ, LIEBERMAN DE, et al. Modern-day environmental factors in the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2018;14(11):674-681.
[3] LOESER RF, COLLINS JA, DIEKMAN BO. Ageing and the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2016;12(7):412-420.
[4] SANCHEZ-LOPEZ E, CORAS R, TORRES A, et al. Synovial inflammation in osteoarthritis progression. Nat Rev Rheumatol. 2022;18(5):258-275.
[5] KRAUS VB, BLANCO FJ, ENGLUND M, et al. Call for standardized definitions of osteoarthritis and risk stratification for clinical trials and clinical use. Osteoarthritis Cartilage. 2015;23(8):1233-1241.
[6] 汪国翔,章晓云.骨关节炎病变过程中炎症细胞因子及相关信号通路的作用机制[J].中国组织工程研究,2021,25(14):2266-2273.
[7] CHEVALIER X, EYMARD F, RICHETTE P. Biologic agents in osteoarthritis: hopes and disappointments. Nat Rev Rheumatol. 2013;9(7):400-410.
[8] NA HS, PARK JS, CHO KH, et al. Interleukin-1-interleukin-17 signaling axis induces cartilage destruction and promotes experimental osteoarthritis. Front Immunol. 2020;11:730.
[9] NIXON AJ, GROL MW, LANG H M, et al. Disease-modifying osteoarthritis treatment with interleukin-1 receptor antagonist gene therapy in small and large animal models. Arthritis Rheumatol. 2018;70(11):1757-1768.
[10] MALEMUD CJ. Anticytokine therapy for osteoarthritis evidence to date. Drugs Aging. 2010;27(2):95-115.
[11] SENTER R, BOYCE R, REPIC M, et al. Efficacy and safety of FX201, a novel intra-articular IL-1Ra gene therapy for osteoarthritis treatment, in a rat model. HUM GENE THER. 2022;33(9-10):541-549.
[12] FLEXION THERAPEUTICS, INC. An open-label, single ascending dose study to assess the safety and tolerability of FX201 in patients with osteoarthritis of the knee: NCT04119687. clinicaltrials.gov, 2021[2022-04-14].
[13] VERBRUGGEN G, WITTOEK R, CRUYSSEN BV, et al. Tumour necrosis factor blockade for the treatment of erosive osteoarthritis of the interphalangeal finger joints: a double blind, randomised trial on structure modification. Ann Rheum Dis. 2012;71(6):891-898.
[14] ZHAO YP, LIU B, TIAN QY, et al. Progranulin protects against osteoarthritis through interacting with TNF-α and β-Catenin signalling. Ann Rheum Dis. 2015;74(12):2244-2253.
[15] CANADIAN RESEARCH EDUCATION IN ARTHRITIS. Osteoarthritis of the knee, inflammation, and the effect of adalimumab (OKINADA): a randomized placebo-controlled Trial: NCT02471118. clinicaltrials.gov, 2021[2022-11-30].
[16] RYU JH, YANG S, SHIN Y, et al. Interleukin-6 plays an essential role in hypoxia-inducible factor 2α-induced experimental osteoarthritic cartilage destruction in mice. Arthritis Rheum. 2011;63(9):2732-2743.
[17] TSUCHIDA AI, BEEKHUIZEN M, `T HART MC, et al. Cytokine profiles in the joint depend on pathology, but are different between synovial fluid, cartilage tissue and cultured chondrocytes. Arthritis Res Ther. 2014; 16(5):441.
[18] NASI S, SO A, COMBES C, et al. Interleukin-6 and chondrocyte mineralisation act in tandem to promote experimental osteoarthritis. Ann Rheum Dis. 2016;75(7):1372-1379.
[19] LATOURTE A, CHERIFI C, MAILLET J, et al. Systemic inhibition of IL-6/Stat3 signalling protects against experimental osteoarthritis. Ann Rheum Dis. 2017;76(4):748-755.
[20] RICHETTE P, LATOURTE A, SELLAM J, et al. Efficacy of tocilizumab in patients with hand osteoarthritis: double blind, randomised, placebo-controlled, multicentre trial. Ann Rheum Dis. 2021;80(3):349-355.
[21] YU X, QI Y, ZHAO T, et al. NGF increases FGF2 expression and promotes endothelial cell migration and tube formation through PI3K/Akt and ERK/MAPK pathways in human chondrocytes. Osteoarthritis Cartilage. 2019;27(3):526-534.
[22] GONDAL FR, BILAL J, KENT KWOH C. Tanezumab for the treatment of osteoarthritis pain. Drugs Today (Barc). 2022;58(4):187-200.
[23] COLLISON J. Anti-NGF therapy improves osteoarthritis pain. Nat Rev Rheumatol. 2019;15(8):450.
[24] MOBASHERI A, CHOI H, MARTÍN-VASALLO P. Over-production of therapeutic growth factors for articular cartilage regeneration by protein production platforms and protein packaging cell lines. Biology (Basel). 2020;9(10):330.
[25] LEE H, KIM H, SEO J, et al. Tissue Gene-C promotes an anti-inflammatory micro-environment in a rat monoiodoacetate model of osteoarthritis via polarization of M2 macrophages leading to pain relief and structural improvement. Inflammopharmacology. 2020;28(5):1237-1252.
[26] ORTVED KF, BEGUM L, MOHAMMED HO, et al. Implantation of rAAV5-IGF-I transduced autologous chondrocytes improves cartilage repair in full-thickness defects in the equine model. Mol Ther. 2015;23(2):363-273.
[27] HEMPHILL DD, MCILWRAITH CW, SLAYDEN RA, et al. Adeno-associated virus gene therapy vector scAAVIGF-I for transduction of equine articular chondrocytes and RNA-seq analysis. Osteoarthritis Cartilage. 2016;24(5): 902-911.
[28] MAIHÖFER J, MADRY H, REY‐RICO A, et al. Hydrogel‐guided, rAAV‐mediated IGF‐I overexpression enables long‐term cartilage repair and protection against perifocal osteoarthritis in a large‐animal full‐thickness chondral defect model at one year in vivo. Adv Mater. 2021;33(16):e2008451.
[29] MÜLLER S, LINDEMANN S, GIGOUT A. Effects of sprifermin, IGF1, IGF2, BMP7, or CNP on bovine chondrocytes in monolayer and 3D culture. J Orthop Res. 2020;38(3):653-662.
[30] ROEMER FW, KRAINES J, AYDEMIR A, et al. Evaluating the structural effects of intra-articular sprifermin on cartilage and non-cartilaginous tissue alterations, based on sqMRI assessment over 2 years. Osteoarthritis Cartilage. 2020;28(9):1229-1234.
[31] EMD SERONO RESEARCH DEVELOPMENT INSTITUTE, INC. A multicenter, randomized, double-blind, placebo controlled, parallel-group trial to investigate the efficacy and safety of different intra articular (i.a.) dosages of sprifermin in subjects with primary osteoarthritis of the knee: results/NCT01919164. clinicaltrials.gov, 2020[2022-05-19].
[32] HOCHBERG MC, GUERMAZI A, GUEHRING H, et al. Effect of intra-articular sprifermin vs. placebo on femorotibial joint cartilage thickness in patients with osteoarthritis: the FORWARD randomized clinical trial. JAMA. 2019; 322(14):1360-1370.
[33] ZENG N, CHEN XY, YAN ZP, et al. Efficacy and safety of sprifermin injection for knee osteoarthritis treatment: a meta-analysis. Arthritis Res Ther. 2021; 23(1):107.
[34] BOON MR, VAN DER HORST G, VAN DER PLUIJM G, et al. Bone morphogenetic protein 7: a broad-spectrum growth factor with multiple target therapeutic potency. Cytokine Growth Factor Rev. 2011;22(4):221-229.
[35] HAYASHI M, MUNETA T, TAKAHASHI T, et al. Intra-articular injections of bone morphogenetic protein-7 retard progression of existing cartilage degeneration: progression of existing cartilage degeneration. J Orthop Res. 2010;28(11):1502-1506.
[36] HUNTER DJ, PIKE MC, JONAS BL, et al. Phase 1 safety and tolerability study of BMP-7 in symptomatic knee osteoarthritis. BMC Musculoskelet Disord. 2010;11:232.
[37] CARON MMJ, RIPMEESTER EGJ, VAN DEN AKKER G, et al. Discovery of bone morphogenetic protein 7-derived peptide sequences that attenuate the human osteoarthritic chondrocyte phenotype. Mol Ther Methods Clin Dev. 2021;21:247-261.
[38] ZHAO R, CHEN NN, ZHOU XW, et al. Exogenous IFN-beta regulates the RANKL-c-Fos-IFN-beta signaling pathway in the collagen antibody-induced arthritis model. J Transl Med. 2014;12:330.
[39] BEVAART L, AALBERS CJ, VIERBOOM MPM, et al. Safety, Biodistribution, and efficacy of an AAV-5 vector encoding human interferon-beta (ART-I02) delivered via intra-articular injection in rhesus monkeys with collagen-induced arthritis. Hum Gene Ther Clin Dev. 2015;26(2):103-112.
[40] ARTHROGEN. A single dose clinical trial to study the safety of ART-I02 in patients with arthritis: NCT02727764. clinicaltrials.gov, 2018[2022-05-01].
[41] YAZICI Y, MCALINDON TE, FLEISCHMANN R, et al. A novel Wnt pathway inhibitor, SM04690, for the treatment of moderate to severe osteoarthritis of the knee: results of a 24-week, randomized, controlled, phase 1 study. Osteoarthritis Cartilage. 2017;25(10):1598-1606.
[42] DESHMUKH V, HU H, BARROGA C, et al. A small-molecule inhibitor of the Wnt pathway (SM04690) as a potential disease modifying agent for the treatment of osteoarthritis of the knee. Osteoarthritis Cartilage. 2018; 26(1):18-27.
[43] YAZICI Y, MCALINDON TE, GIBOFSKY A, et al. A Phase 2b randomized trial of lorecivivint, a novel intra-articular CLK2/DYRK1A inhibitor and Wnt pathway modulator for knee osteoarthritis. Osteoarthritis Cartilage. 2021;29(5): 654-666.
[44] BIOSPLICE THERAPEUTICS, INC. A phase 3, 28-week, multicenter, randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of a single injection of SM04690 injected in the target knee joint of moderately to severely symptomatic osteoarthritis subjects: NCT04385303. clinicaltrials.gov, 2022[2022-05-19].
[45] NAILWAL NP, DOSHI GM. Role of intracellular signaling pathways and their inhibitors in the treatment of inflammation. Inflammopharmacology. 2021;29(3):617-640.
[46] MINGUZZI M, PANICHI V, D’ADAMO S, et al. Pleiotropic roles of NOTCH1 signaling in the loss of maturational arrest of human osteoarthritic chondrocytes. Int J Mol Sci. 2021;22(21):12012.
[47] CHOI MC, JO J, PARK J, et al. NF-κB signaling pathways in osteoarthritic cartilage destruction. Cells. 2019;8(7):734.
[48] ZHOU C, CUI Y, YANG Y, et al. Runx1 protects against the pathological progression of osteoarthritis. Bone Res. 2021;9(1):50.
[49] LAWSON TB, MÄKELÄ JTA, KLEIN T, et al. Nanotechnology and osteoarthritis. part 1: clinical landscape and opportunities for advanced diagnostics. J Orthop Res. 2021;39(3):465-472.
[50] LAWSON TB, MÄKELÄ JTA, KLEIN T, et al. Nanotechnology and Osteoarthritis. Part 2: Opportunities for advanced devices and therapeutics. J Orthop Res. 2021;39(3):473-484.
[51] WEI P, BAO R. Intra-articular mesenchymal stem cell injection for knee osteoarthritis: mechanisms and clinical evidence. Int J Mol Sci. 2022; 24(1):59.
[52] HUANG J, LIU Q, XIA J, et al. Modification of mesenchymal stem cells for cartilage-targeted therapy. J Transl Med. 2022;20(1):515.
[53] WU H, PENG Z, XU Y, et al. Engineered adipose-derived stem cells with IGF-1-modified mRNA ameliorates osteoarthritis development. Stem Cell Res Ther. 2022;13(1):19.
[54] EVERTS P, ONISHI K, JAYARAM P, et al. Platelet-rich plasma: new performance understandings and therapeutic considerations in 2020. Int J Mol Sci. 2020; 21(20):7794.
[55] RIEWRUJA K, PHAKHAM S, SOMPOLPONG P, et al. Cytokine profiling and intra-articular injection of autologous platelet-rich plasma in knee osteoarthritis. Int J Mol Sci. 2022;23(2):890.
[56] NIEMIEC P, SZYLUK K, BALCERZYK A, et al. Why PRP works only on certain patients with tennis elbow? Is PDGFB gene a key for PRP therapy effectiveness? A prospective cohort study. BMC Musculoskelet Disord. 2021;22(1):710.
[57] NOWACZYK A, SZWEDOWSKI D, DALLO I, et al. Overview of first-line and second-line pharmacotherapies for osteoarthritis with special focus on intra-articular treatment. Int J Mol Sci. 2022;23(3):1566.
[58] OUDELAAR BW, PEERBOOMS JC, HUIS IN T VELD R, et al. Concentrations of blood components in commercial platelet-rich plasma separation systems: a review of the literature. Am J Sports Med. 2019;47(2):479-487.
[59] EYMARD F, ORNETTI P, MAILLET J, et al. Correction to: Intra‑articular injections of platelet‑rich plasma in symptomatic knee osteoarthritis: a consensus statement from French‑speaking experts. Knee Surg Sports Traumatol Arthrosc. 2021;29(10):3211-3212.
[60] LANA JF, MACEDO A, INGRAO ILG, et al. Leukocyte-rich PRP for knee osteoarthritis: current concepts. J Clin Orthop Trauma. 2019;10(Suppl 1): S179-S182.
|