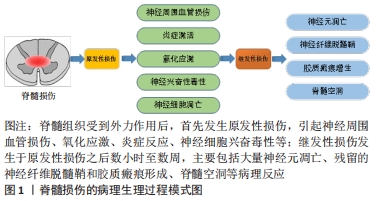
[1] LI HL, XU H, LI YL, et al. Epidemiology of traumatic spinal cord injury in Tianjin, China: An 18-year retrospective study of 735 cases. J Spinal Cord Med. 2019;42(6):778-785.
[2] AHUJA CS, NORI S, TETREAULT L, et al. Traumatic Spinal Cord Injury-Repair and Regeneration. Neurosurgery. 2017;80(3S):S9-S22.
[3] MCRAE J, SMITH C, EMMANUEL A, et al. The experiences of individuals with cervical spinal cord injury and their family during post-injury care in non-specialised and specialised units in UK. BMC Health Serv Res. 2020;20(1):783.
[4] HACHEM LD, MOTHE AJ, TATOR CH. Unlocking the paradoxical endogenous stem cell response after spinal cord injury. Stem Cells. 2020;38(2):187-194.
[5] O’SHEA TM, BURDA JE, SOFRONIEW MV. Cell biology of spinal cord injury and repair. J Clin Invest. 2017;127(9):3259-3270.
[6] KIMURA T, HORIKOSHI Y, KURIYAGAWA C, et al. Rho/ROCK Pathway and Noncoding RNAs: Implications in Ischemic Stroke and Spinal Cord Injury. Int J Mol Sci. 2021;22(21):11573.
[7] CHENG P, LIAO HY, ZHANG HH. The role of Wnt/mTOR signaling in spinal cord injury. J Clin Orthop Trauma. 2022;25:101760.
[8] ZAVVARIAN MM, ZHOU C, KAHNEMUYIPOUR S, et al. The MAPK Signaling Pathway Presents Novel Molecular Targets for Therapeutic Intervention after Traumatic Spinal Cord Injury: A Comparative Cross-Species Transcriptional Analysis. Int J Mol Sci. 2021;22(23):12934.
[9] BUTTITTA LA, EDGAR BA. How size is controlled: from Hippos to Yorkies. Nat Cell Biol. 2007;9(11):1225-1227.
[10] YU FX, ZHAO B, GUAN KL. Hippo Pathway in Organ Size Control, Tissue Homeostasis, and Cancer. Cell. 2015;163(4):811-828.
[11] ZHAO B, WEI X, LI W, et al. Inactivation of YAP oncoprotein by the Hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev. 2007;21(21):2747-2761.
[12] DEY A, VARELAS X, GUAN KL. Targeting the Hippo pathway in cancer, fibrosis, wound healing and regenerative medicine. Nat Rev Drug Discov. 2020;19(7):480-494.
[13] GONZÁLEZ-ALONSO P, ZAZO S, MARTÍN-APARICIO E, et al. The Hippo Pathway Transducers YAP1/TEAD Induce Acquired Resistance to Trastuzumab in HER2-Positive Breast Cancer. Cancers (Basel). 2020; 12(5):1108.
[14] HUANG Z, ZHOU JK, WANG K, et al. PDLIM1 Inhibits Tumor Metastasis Through Activating Hippo Signaling in Hepatocellular Carcinoma. Hepatology. 2020; 71(5): 1643-1659.
[15] YIN F, YU JZ, ZHENG YG, et al. Spatial organization of Hippo signaling at the plasma membrane mediated by the tumor suppressor Merlin/NF2. Cell. 2013;154(6):1342-1355.
[16] DONG JX, FELDMANN G, HUANG JB, et al. Elucidation of a universal size-control mechanism in Drosophila and mammals. Cell. 2007; 130(6):1120-1133.
[17] ZHAO B, LI L, TUMANENG K, et al. A coordinated phosphorylation by Lats and CK1 regulates YAP stability through SCFβ-TRCP. Genes Dev. 2010;24(1):72-85.
[18] MENG Z, MOROISHI T, MOTTIER-PAVIE V, et al. MAP4K family kinases act in parallel to MST1/2 to activate LATS1/2 in the Hippo pathway. Nat Commun. 2015;6:8357.
[19] HERGOVICH A. The Roles of NDR Protein Kinases in Hippo Signalling. Genes (Basel). 2016;7(5):21.
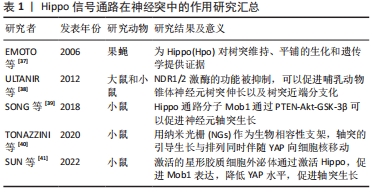
[20] EMOTO K. The growing role of the Hippo--NDR kinase signalling in neuronal development and disease. J Biochem. 2011;150(2):133-141.
[21] CHENG J, WANG S, DONG Y, et al. The Role and Regulatory Mechanism of Hippo Signaling Components in the Neuronal System. Front Immunol. 2020;11:281.
[22] ZHANG Y, CHEN K, SLOAN SA, et al. An RNA-sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex. J Neurosci. 2014;34(36):11929-11947.
[23] POON CL, MITCHELL KA, KONDO S, et al. The Hippo Pathway Regulates Neuroblasts and Brain Size in Drosophila melanogaster. Curr Biol. 2016; 26(8):1034-1042.
[24] SHACKELFORD DB, SHAW RJ. The LKB1-AMPK pathway: metabolism and growth control in tumour suppression. Nat Rev Cancer. 2009;9(8): 563-575.
[25] NGUYEN HB, BABCOCK JT, WELLS CD, et al. LKB1 tumor suppressor regulates AMP kinase/mTOR-independent cell growth and proliferation via the phosphorylation of Yap. Oncogene. 2013;32(35):4100-4109.
[26] MO JS, MENG Z, KIM YC, et al. Cellular energy stress induces AMPK-mediated regulation of YAP and the Hippo pathway. Nat Cell Biol. 2015;17(4):500-510.
[27] GAILITE I, AERNE BL, TAPON N. Differential control of Yorkie activity by LKB1/AMPK and the Hippo/Warts cascade in the central nervous system. Proc Natl Acad Sci U S A. 2015;112(37):E5169-5178.
[28] LAVADO A, PARK JY, PARÉ J, et al. The Hippo Pathway Prevents YAP/TAZ-Driven Hypertranscription and Controls Neural Progenitor Number. Dev Cell. 2018;47(5):576-591.e8.
[29] THOMPSON R, CHAN C. Signal transduction of the physical environment in the neural differentiation of stem cells. Technology (Singap World Sci). 2016;4(1):1-8.
[30] DING R, WEYNANS K, BOSSING T, et al. The Hippo signalling pathway maintains quiescence in Drosophila neural stem cells. Nat Commun. 2016;7:10510.
[31] KUMAR D, NITZAN E, KALCHEIM C. YAP promotes neural crest emigration through interactions with BMP and Wnt activities. Cell Commun Signal. 2019;17(1):69.
[32] ZHAO X, LE TP, ERHARDT S, et al. Hippo-Yap Pathway Orchestrates Neural Crest Ontogenesis. Front Cell Dev Biol. 2021;9:706623.
[33] JUKAM D, XIE B, RISTER J, et al. Opposite feedbacks in the Hippo pathway for growth control and neural fate. Science. 2013;342(6155): 1238016.
[34] HUANG Z, XIONG WC. Neogenin-YAP signaling in neocortical astrocytic differentiation. Neurogenesis (Austin). 2016;3(1):e1248735.
[35] PETROVA V, EVA R. The Virtuous Cycle of Axon Growth: Axonal Transport of Growth-Promoting Machinery as an Intrinsic Determinant of Axon Regeneration. Dev Neurobiol. 2018;78(10):898-925.
[36] O’DONOVAN KJ. Intrinsic Axonal Growth and the Drive for Regeneration. Front Neurosci. 2016;10:486.
[37] EMOTO K, PARRISH JZ, JAN LY, et al. The tumour suppressor Hippo acts with the NDR kinases in dendritic tiling and maintenance. Nature. 2006;443(7108):210-213.
[38] ULTANIR SK, HERTZ NT, LI G, et al. Chemical genetic identification of NDR1/2 kinase substrates AAK1 and Rabin8 Uncovers their roles in dendrite arborization and spine development. Neuron. 2012;73(6): 1127-1142.
[39] SONG Z, HAN X, ZOU H, et al. PTEN-GSK3β-MOB1 axis controls neurite outgrowth in vitro and in vivo. Cell Mol Life Sci. 2018;75(23):4445-4464.
[40] TONAZZINI I, MASCIULLO C, SAVI E, et al. Neuronal contact guidance and YAP signaling on ultra-small nanogratings. Sci Rep. 2020;10(1): 3742.
[41] SUN H, CAO X, GONG A, et al. Extracellular vesicles derived from astrocytes facilitated neurite elongation by activating the Hippo pathway. Exp Cell Res. 2022;411(1):112937.
[42] POPLAWSKI GHD, LIE R, HUNT M, et al. Adult Rat Myelin Enhances Axonal Outgrowth From Neural Stem Cells. Sci Transl Med. 2018; 10(442):eaal2563.
[43] BARNABÉ-HEIDER F, GÖRITZ C, SABELSTRÖM H, et al. Origin of new glial cells in intact and injured adult spinal cord. Cell Stem Cell. 2010; 7:470-482.
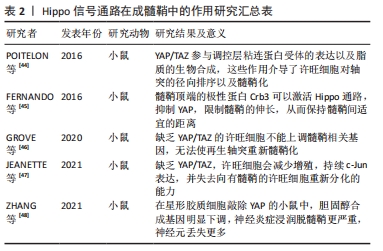
[44] POITELON Y, LOPEZ-ANIDO C, CATIGNAS K, et al. YAP and TAZ control peripheral myelination and the expression of laminin receptors in Schwann cells. Nat Neurosci. 2016;19(7):879-887.
[45] FERNANDO RN, COTTER L, PERRIN-TRICAUD C, et al. Optimal myelin elongation relies on YAP activation by axonal growth and inhibition by Crb3/Hippo pathway. Nat Commun. 2016;7:12186.
[46] GROVE M, LEE H, ZHAO H, et al. Axon-dependent expression of YAP/TAZ mediates Schwann cell remyelination but not proliferation after nerve injury. Elife. 2020;9:e50138.
[47] JEANETTE H, MARZIALI LN, BHATIA U, et al. YAP and TAZ regulate Schwann cell proliferation and differentiation during peripheral nerve regeneration. Glia. 2021;69(4):1061-1074.
[48] ZHANG J, XU X, LIU H, et al. Astrocytic YAP prevents the demyelination through promoting expression of cholesterol synthesis genes in experimental autoimmune encephalomyelitis. Cell Death Dis. 2021; 12(10):907.
[49] GADANI SP, WALSH JT, LUKENS JR, et al. Dealing with Danger in the CNS: The Response of the Immune System to Injury. Neuron. 2015;87: 47-62.
[50] ZHANG M, TAO W, YUAN Z, et al. Mst-1 deficiency promotes post-traumatic spinal motor neuron survival via enhancement of autophagy flux. Neurochem. 2017;143(2):244-256.
[51] ZHOU L, ZHANG Q, ZHANG P, et al. c-Abl-mediated Drp1 phosphorylation promotes oxidative stress-induced mitochondrial fragmentation and neuronal cell death. Cell Death Dis. 2017;8(10): e3117.
[52] ZHANG P, WANG T, ZHANG D, et al. Exploration of MST1-Mediated Secondary Brain Injury Induced by Intracerebral Hemorrhage in Rats via Hippo Signaling Pathway. Transl Stroke Res. 2019;10(6):729-743.
[53] HUANG L, LI S, DAI Q, et al. Astrocytic Yes-associated protein attenuates cerebral ischemia-induced brain injury by regulating signal transducer and activator of transcription 3 signaling. Exp Neurol. 2020;333:113431.
[54] YU H, CAO X, LI W, et al. Targeting connexin 43 provides anti-inflammatory effects after intracerebral hemorrhage injury by regulating YAP signaling. J Neuroinflammation. 2020;17(1):322.
[55] QING J, LIU X, WU Q, et al. Hippo/YAP Pathway Plays a Critical Role in Effect of GDNF Against Aβ-Induced Inflammation in Microglial Cells. DNA Cell Biol. 2020;39(6):1064-1071.
[56] BRADBURY EJ, BURNSIDE ER. Moving beyond the glial scar for spinal cord repair. Nat Commun. 2019;10(1):3879.
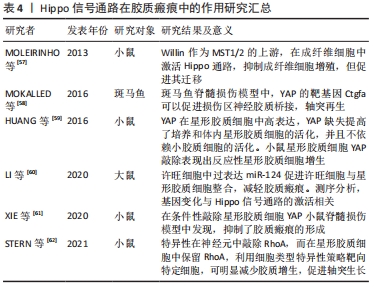
[57] MOLEIRINHO S, PATRICK C, TILSTON-LÜNEL AM, et al. Willin, an upstream component of the hippo signaling pathway, orchestrates mammalian peripheral nerve fibroblasts. PLoS One. 2013;8(4):e60028.
[58] MOKALLED MH, PATRA C, DICKSON AL, et al. Injury-induced ctgfa directs glial bridging and spinal cord regeneration in zebrafish. Science. 2016;354(6312):630-634.
[59] HUANG Z, WANG Y, HU G, et al. YAP Is a Critical Inducer of SOCS3, Preventing Reactive Astrogliosis. Cereb Cortex. 2016;26(5):2299-2310.
[60] LI Z, YU Y, KANG J, et al. MicroRNA-124 Overexpression in Schwann Cells Promotes Schwann Cell-Astrocyte Integration and Inhibits Glial Scar Formation Ability. Front Cell Neurosci. 2020;14:144.
[61] XIE C, SHEN X, XU X, et al. Astrocytic YAP Promotes the Formation of Glia Scars and Neural Regeneration after Spinal Cord Injury. J Neurosci. 2020;40(13):2644-2662.
[62] STERN S, HILTON BJ, BURNSIDE ER, et al. RhoA drives actin compaction to restrict axon regeneration and astrocyte reactivity after CNS injury. Neuron. 2021;109(21):3436-3455.e9.
|