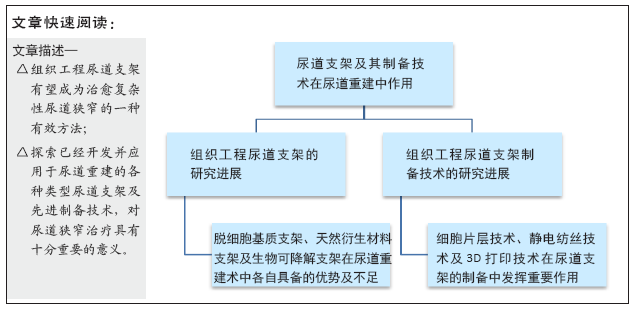
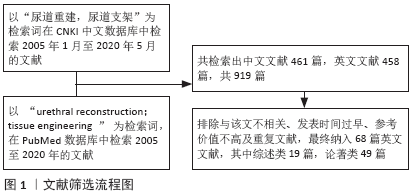
[1] ZAID UB, LAVIEN G, PETERSON AC. Management of the Recurrent Male Urethral Stricture. Curr Urol Rep. 2016;17(4):33.
[2] SIMSEK A, ALDAMANHORI R, CHAPPLE CR, et al. Overcoming scarring in the urethra: Challenges for tissue engineering. Asian J Urol. 2018;5(2):69-77.
[3] HAMPSON LA, MCANINCH JW, BREYER BN. Male urethral strictures and their management. Nat Rev Urol. 2014;11(1):43-50.
[4] DING J, LI Q, LI S, et al. Ten years’ experience for hypospadias repair: combined buccal mucosa graft and local flap for urethral reconstruction. Urol Int. 2014; 93(4):454-459.
[5] MARKIEWICZ MR, DESANTIS JL, MARGARONE JE 3RD, et al. Morbidity associated with oral mucosa harvest for urological reconstruction: an overview. J Oral Maxillofac Surg. 2008;66(4):739-744.
[6] HARRIS CR, OSTERBERG EC, SANFORD T, et al. National Variation in Urethroplasty Cost and Predictors of Extreme Cost: A Cost Analysis With Policy Implications. Urology. 2016;94:246-254.
[7] XUE JD, GAO J, FU Q, et al. Seeding cell approach for tissue-engineered urethral reconstruction in animal study: A systematic review and meta-analysis. Exp Biol Med (Maywood). 2016;241(13):1416-1428.
[8] ATALA A. Engineering organs. Curr Opin Biotechnol. 2009;20(5):575-592.
[9] CHAPPLE C. Tissue engineering of the urethra: where are we in 2019? World J Urol. 2020;8(9):2101-2105.
[10] DORIN RP, POHL HG, DE FILIPPO RE, et al. Tubularized urethral replacement with unseeded matrices: what is the maximum distance for normal tissue regeneration? World J Urol. 2008;26(4):323-326.
[11] DE GRAAF P, RAMADAN R, LINSSEN EC, et al. The multilayered structure of the human corpus spongiosum. Histol Histopathol. 2018;33(12):1335-1345.
[12] COENEN AMJ, BERNAERTS KV, HARINGS JAW, et al. Elastic materials for tissue engineering applications: Natural, synthetic, and hybrid polymers. Acta Biomater. 2018;79:60-82.
[13] BIRDER L, ANDERSSON KE. Urothelial signaling. Physiol Rev. 2013;93(2):653-680.
[14] LALLA M, GREGERSEN H, OLSEN LH, et al. In vivo biomechanical assessment of anterior rabbit urethra after repair of surgically created hypospadias. J Urol. 2010;184(2):675-680.
[15] MIJAILOVICH SM, SULLIVAN MP, YALLA SV, et al. Effect of urethral compliance on the steady state p-Q relationships assessed with a mechanical analog of the male lower urinary tract. Neurourol Urodyn. 2007;26(2):234-246.
[16] ABBAS TO, YALCIN HC, PENNISI CP. From Acellular Matrices to Smart Polymers: Degradable Scaffolds that are Transforming the Shape of Urethral Tissue Engineering. Int J Mol Sci. 2019;20(7):1763.
[17] RAJASEKARAN M, STEIN P, PARSONS CL. Toxic factors in human urine that injure urothelium. Int J Urol. 2006;13(4):409‐414.
[18] SHALUMON KT, KUO CY, WONG CB, et al. Gelatin/Nanohyroxyapatite Cryogel Embedded Poly(lactic-co-glycolic Acid)/Nanohydroxyapatite Microsphere Hybrid Scaffolds for Simultaneous Bone Regeneration and Load-Bearing. Polymers (Basel). 2018;10(6):620.
[19] MARGOLIS G, POLYAK B, COHEN S. Magnetic Induction of Multiscale Anisotropy in Macroporous Alginate Scaffolds. Nano Lett. 2018;18(11):7314-7322.
[20] ULERY BD, NAIR LS, LAURENCIN CT. Biomedical Applications of Biodegradable Polymers. J Polym Sci B Polym Phys. 2011;49(12):832-864.
[21] STACHEWICZ U, SZEWCZYK PK, KRUK A, et al. Pore shape and size dependence on cell growth into electrospun fiber scaffolds for tissue engineering: 2D and 3D analyses using SEM and FIB-SEM tomography. Mater Sci Eng C Mater Biol Appl. 2019;95:397-408.
[22] FU WJ, WANG ZX, LI G, et al. A surface-modified biodegradable urethral scaffold seeded with urethral epithelial cells. Chin Med J (Engl). 2011; 124(19):3087-3092.
[23] FIGALLO E, FLAIBANI M, ZAVAN B, et al. Micropatterned biopolymer 3D scaffold for static and dynamic culture of human fibroblasts. Biotechnol Prog. 2007;23(1):210-216.
[24] RAYA-RIVERA A, ESQUILIANO DR, YOO JJ, et al. Tissue-engineered autologous urethras for patients who need reconstruction: an observational study. Lancet. 2011;377(9772):1175-1182.
[25] DAVIS NF, CUNNANE EM, O’BRIEN FJ, et al. Tissue engineered extracellular matrices (ECMs) in urology: Evolution and future directions. Surgeon. 2018; 16(1):55-65.
[26] ENGEL O, SOAVE A, RINK M, et al.Tissue engineering in reconstructive urology. Urologe A. 2015;54(5):690-695.
[27] RIDER P, KAČAREVIĆ ŽP, ALKILDANI S, et al. Bioprinting of tissue engineering scaffolds. J Tissue Eng. 2018;9:2041731418802090.
[28] SIMÕES IN, VALE P, SOKER S, et al. Acellular Urethra Bioscaffold: Decellularization of Whole Urethras for Tissue Engineering Applications. Sci Rep. 2017;7:41934.
[29] ZHANG K, GUO X, ZHAO W, et al. Application of Wnt Pathway Inhibitor Delivering Scaffold for Inhibiting Fibrosis in Urethra Strictures: In Vitro and in Vivo Study. Int J Mol Sci. 2015;16(11):27659-27676.
[30] SAYEG K, FREITAS-FILHO LG, WAITZBERG ÂF, et al. Integration of collagen matrices into the urethra when implanted as onlay graft. Int Braz J Urol. 2013;39(3):414-423.
[31] DE FILIPPO RE, KORNITZER BS, YOO JJ, et al. Penile urethra replacement with autologous cell-seeded tubularized collagen matrices. J Tissue Eng Regen Med. 2015;9(3):257-264.
[32] ORABI H, ABOUSHWAREB T, ZHANG Y, et al. Cell-seeded tubularized scaffolds for reconstruction of long urethral defects: a preclinical study. Eur Urol. 2013;63(3):531-538.
[33] LIU Y, MA W, LIU B, et al. Urethral reconstruction with autologous urine-derived stem cells seeded in three-dimensional porous small intestinal submucosa in a rabbit model. Stem Cell Res Ther. 2017;8(1):63.
[34] DAVIS NF, CALLANAN A, MCGUIRE BB, et al. Evaluation of viability and proliferative activity of human urothelial cells cultured onto xenogenic tissue-engineered extracellular matrices. Urology. 2011;77(4):1007.e1-1007.e10077.
[35] WANG F, LIU T, YANG L, et al. Urethral reconstruction with tissue-engineered human amniotic scaffold in rabbit urethral injury models. Med Sci Monit. 2014;20:2430-2438.
[36] RAMUTA TŽ, KREFT ME. Human Amniotic Membrane and Amniotic Membrane-Derived Cells: How Far Are We from Their Use in Regenerative and Reconstructive Urology? Cell Transplant. 2018;27(1):77-92.
[37] ORABI H, BOUHOUT S, MORISSETTE A, et al. Tissue engineering of urinary bladder and urethra: advances from bench to patients. ScientificWorldJournal. 2013;2013:154564.
[38] LIU Y, HUANG L, YUAN W, et al. Sustained release of stromal cell-derived factor-1 alpha from silk fibroin microfiber promotes urethral reconstruction in rabbits. J Biomed Mater Res A. 2020;108(8):1760-1773.
[39] XIE M, SONG L, WANG J, Et al. Evaluation of stretched electrospun silk fibroin matrices seeded with urothelial cells for urethra reconstruction. J Surg Res. 2013;184(2):774-781.
[40] LV X, LI Z, CHEN S, et al. Structural and functional evaluation of oxygenating keratin/silk fibroin scaffold and initial assessment of their potential for urethral tissue engineering. Biomaterials. 2016;84:99-110.
[41] CHUNG YG, TU D, FRANCK D, et al. Acellular bi-layer silk fibroin scaffolds support tissue regeneration in a rabbit model of onlay urethroplasty. PLoS One. 2014;9(3):e91592.
[42] ALGARRAHI K, AFFAS S, SACK BS, et al. Repair of injured urethras with silk fibroin scaffolds in a rabbit model of onlay urethroplasty. J Surg Res. 2018;229:192-199.
[43] TIAN B, SONG L, LIANG T, et al. Repair of urethral defects by an adipose mesenchymal stem cell porous silk fibroin material. Mol Med Rep. 2018;18(1):209-215.
[44] NAJI M, RASOULI J, SHAKHSSALIM N, et al. Supportive features of a new hybrid scaffold for urothelium engineering. Arch Med Sci. 2015;11(2): 438-445.
[45] DORATI R, COLONNA C, TOMASI C, et al. Design of 3D scaffolds for tissue engineering testing a tough polylactide-based graft copolymer. Mater Sci Eng C Mater Biol Appl. 2014;34:130-139.
[46] SARTONEVA R, HAIMI S, MIETTINEN S, et al. Comparison of a poly-L-lactide-co-ε-caprolactone and human amniotic membrane for urothelium tissue engineering applications. J R Soc Interface. 2011;8(58):671-677.
[47] HICKS BG, LOPEZ EA, EASTMAN R JR, et al. Differential affinity of vitronectin versus collagen for synthetic biodegradable scaffolds for urethroplastic applications. Biomaterials. 2011;32(3):797-807.
[48] GUELCHER SA. Biodegradable polyurethanes: synthesis and applications in regenerative medicine. Tissue Eng Part B Rev. 2008;14(1):3-17.
[49] LV X, GUO Q, HAN F, et al. Electrospun Poly(l-lactide)/Poly(ethylene glycol) Scaffolds Seeded with Human Amniotic Mesenchymal Stem Cells for Urethral Epithelium Repair. Int J Mol Sci. 2016;17(8):1262.
[50] PATEL S, PATEL G. A Review and Analysis on Recent Advancements in Bubble Electrospinning Technology for Nanofiber Production. Recent Pat Nanotechnol. 2019;13(2):80-91.
[51] VILLARREAL-GÓMEZ LJ, CORNEJO-BRAVO JM, VERA-GRAZIANO R, et al. Electrospinning as a powerful technique for biomedical applications: a critically selected survey. J Biomater Sci Polym Ed. 2016;27(2):157-176.
[52] KISHAN AP, COSGRIFF-HERNANDEZ EM. Recent advancements in electrospinning design for tissue engineering applications: A review. J Biomed Mater Res A. 2017;105(10):2892-2905.
[53] NIU Y, LIU G, FU M, et al. Designing a multifaceted bio-interface nanofiber tissue-engineered tubular scaffold graft to promote neo-vascularization for urethral regeneration. J Mater Chem B. 2020;8(8):1748-1758.
[54] WEI G, LI C, FU Q, et al. Preparation of PCL/silk fibroin/collagen electrospun fiber for urethral reconstruction. Int Urol Nephrol. 2015;47(1):95‐99.
[55] KOBAYASHI J, KIKUCHI A, AOYAGI T, et al. Cell sheet tissue engineering: Cell sheet preparation, harvesting/manipulation, and transplantation. J Biomed Mater Res A. 2019;107(5):955-967.
[56] ZHOU S, YANG R, ZOU Q, et al. Fabrication of Tissue-Engineered Bionic Urethra Using Cell Sheet Technology and Labeling By Ultrasmall Superparamagnetic Iron Oxide for Full-Thickness Urethral Reconstruction. Theranostics. 2017;7(9):2509-2523.
[57] VERSTEEGDEN LR, VAN KAMPEN KA, JANKE HP, et al. Tubular collagen scaffolds with radial elasticity for hollow organ regeneration. Acta Biomater. 2017;52:1-8.
[58] CATTAN V, BERNARD G, ROUSSEAU A, et al. Mechanical stimuli-induced urothelial differentiation in a human tissue-engineered tubular genitourinary graft. Eur Urol. 2011;60(6):1291-1298.
[59] LAL H, PATRALEKH MK. 3D printing and its applications in orthopaedic trauma: A technological marvel. J Clin Orthop Trauma. 2018;9(3):260-268.
[60] KAČAREVIĆ ŽP, RIDER PM, ALKILDANI S, et al. An Introduction to 3D Bioprinting: Possibilities, Challenges and Future Aspects. Materials (Basel). 2018;11(11):2199.
[61] WANG N, LU JG, HE XL, et al. Effects of ischemic postconditioning on reperfusion injury in rat liver grafts after orthotopic liver transplantation. Hepatol Res. 2009;39(4):382-390.
[62] GU BK, CHOI DJ, PARK SJ, et al. 3D Bioprinting Technologies for Tissue Engineering Applications. Adv Exp Med Biol. 2018;1078:15-28.
[63] KANG HW, LEE SJ, KO IK, et al. A 3D bioprinting system to produce human-scale tissue constructs with structural integrity. Nat Biotechnol. 2016;34(3):312-319.
[64] HASSAN S, PRAKASH G, OZTURK A, et al. Evolution and Clinical Translation of Drug Delivery Nanomaterials. Nano Today. 2017;15:91-106.
[65] CASTILHO M, DE RUIJTER M, BEIRNE S, et al. Multitechnology Biofabrication: A New Approach for the Manufacturing of Functional Tissue Structures? Trends Biotechnol. 2020;S0167-7799(20)30119-0. doi: 10.1016/j.tibtech.2020.04.014.
[66] HÖLZL K, LIN S, TYTGAT L, et al. Bioink properties before, during and after 3D bioprinting. Biofabrication. 2016;8(3):032002.
[67] BALOGOVÁ AF, HUDÁK R, TÓTH T, et al. Determination of geometrical and viscoelastic properties of PLA/PHB samples made by additive manufacturing for urethral substitution. J Biotechnol. 2018;284:123-130.
[68] ZHANG K, FU Q, YOO J, et al. 3D bioprinting of urethra with PCL/PLCL blend and dual autologous cells in fibrin hydrogel: An in vitro evaluation of biomimetic mechanical property and cell growth environment. Acta Biomater. 2017;50:154-164.
|