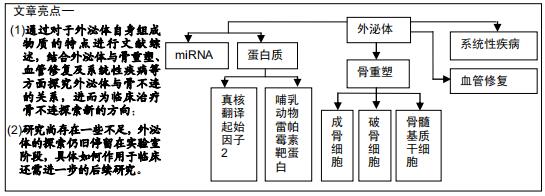
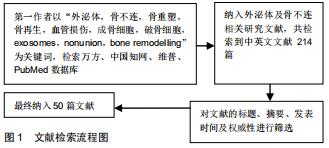
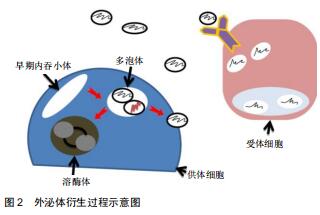
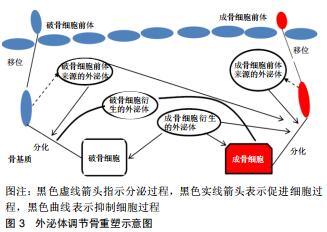
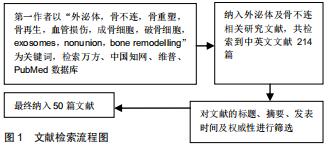
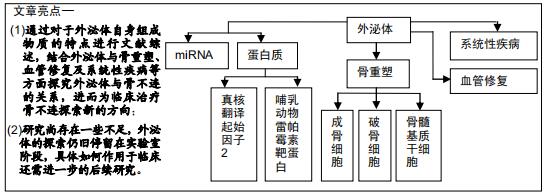
|
[1] CALORI GM. Non-unions. Clinical Cases in Mineral & Bone Metabolism the Official Journal of the Italian Society of Osteoporosis Mineral Metabolism & Skeletal Diseases, 2017;14(2):186-188.
[2] 骆永锋,龚劲纯,吴俊,等.带锁髓内钉治疗四肢创伤骨折后骨不连的临床研究[J]. 齐齐哈尔医学院学报,2017,38(2):193-195.
[3] WU KJ, LI SH, YEH KT, et al. The risk factors of nonunion after intramedullary nailing fixation of femur shaft fracture in middle age patients. Medicine(Baltimore). 2019;98:e16559.
[4] YANEZ-MO M, SIJANDER PR, ANDREU Z, et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015; 4(1):27066.
[5] 李晓晔,周潇逸,张子程,等.外泌体中miRNA对骨重建过程影响研究进展[J].中国矫形外科杂志,2019,27(18):1683-1686.
[6] 赵濛,刘志红,李金泉.外泌体组成特征及其作为细胞通讯和分子标记的生物学作用[J].中国生物化学与分子生物学报, 2016, 32(6):612-619.
[7] LU J,WANG QY,SHENG JG, et al. Exosomes in the repair of bone defects: next-generation therapeutic tools for the treatment of nonunion. Biomed Res Int. 2019;2019: 1983131.
[8] 寿崟,马宇航,虎力,等.外泌体在糖尿病及其并发症的发生、发展和诊治中的作用[J].生理学报,2019,71(6):917-934.
[9] LU Z, ZUO B, JING R, et al. Dendritic cell-derived exosomes elicit tumor regression in autochthonous hepatocellular carcinoma mouse models. J Hepatol. 2017;67: 739-748.
[10] EL-SAGHIR J, NASSAR F, TAWIL N, et al. ATL-derived exosomes modulate mesenchymal stem cells: potential role in leukemia progression. Retrovirology. 2016; 13: 73.
[11] BRAICU C, TOMULEASA C, MONROIG P, et al. Exosomes as divine messengers: are they the Hermes of modern molecular oncology? Cell Death Differ. 2015;22: 34-45.
[12] POURAKBARI R, KHODADADI M, AGHEBATI-MALEKI A, et al. The potential of exosomes in the therapy of the cartilage and bone complications; emphasis on osteoarthritis. Life Sci. 2019;236: 116861.
[13] YAN Z, GUO Y, WANG Y, et al. MicroRNA profiles of BMSCs induced into osteoblasts with osteoinductive medium. Exp Ther Med. 2018;15: 2589-2596.
[14] HAMMOND SM. Dicing and slicing: the core machinery of the RNA interference pathway. FEBS Lett. 2005;579: 5822-5829.
[15] HUTVAGNER G. Small RNA asymmetry in RNAi: Function in RISC assembly and gene regulation. Febs Lett. 2005;579(26): 5850-5857.
[16] WEILNER S, SCHRAML E, REDL H, et al. Secretion of microvesicular miRNAs in cellular and organismal aging. Exp Gerontol. 2013;48(7):626-633.
[17] ZHANG Y, XIE RL, CROCE CM, et al. A program of microRNAs controls osteogenic lineage progression by targeting transcription factor Runx2. Proc Natl Acad Sci U S A. 2011;108(24):9863-9868.
[18] LI Z, HASSAN MQ, VOLINIA S, et al. A microRNA signature for a BMP2-induced osteoblast lineage commitment program. Proc Natl Acad Sci U S A. 2008;105(37): 13906-13911.
[19] LIU Y, MA Y, ZHANG J, et al. Exosomes: a novel therapeutic agent for cartilage and bone tissue regeneration. Dose Response. 2019;17: 1559325819892702.
[20] XU JF, YANG GH, PAN XH, et al. Altered microrna expression profile in exosomes during osteogenic differentiation of human bone marrow-derived mesenchymal stem cells. PLoS ONE. 2014;9(12):e114627.
[21] GE M, KE R, CAI T, et al. Identification and proteomic analysis of osteoblast-derived exosomes. Biochem Biophys Res Commun. 2015;467(1):27-32.
[22] SHRESTHA N, BAHNAN W, WILEY DJ, et al. Eukaryotic Initiation Factor 2 (eIF2) signaling regulates proinflammatory cytokine expression and bacterial invasion. J Biol Chem. 2012; 287(34):28738-28744.
[23] AKTAS BH, BORDELOIS P, PEKER S, et al. Depletion of eIF2·GTP·Met-tRNAi translation initiation complex up-regulates BRCA1 expression in vitro and in vivo. Oncotarget. 2015;6:6902-6914.
[24] JOHNSON ME, DELIARD S, ZHU F, et al. A ChIP-seq-defined genome-wide map of MEF2C binding reveals inflammatory pathways associated with its role in bone density determination. Calcif Tissue Int. 2014;94(4):396-402.
[25] TANAKA KI, KAJI H, YAMAGUCHI T, et al. Involvement of the Osteoinductive Factors, Tmem119 and BMP-2, and the ER Stress Response PERK–eIF2α–ATF4 Pathway in the Commitment of Myoblastic into Osteoblastic Cells. Calcif Tissue Int. 2014;94(4):454-464.
[26] YANG X, MATSUDA K, BIALEK P, et al. ATF4 is a substrate of RSK2 and an essential regulator of osteoblast biology; implication for Coffin-Lowry Syndrome. Cell. 2015;117(3): 387-398.
[27] NOJIMA H, TOKUNAGA C, EGUCHI S, et al. The Mammalian Target of Rapamycin (mTOR) Partner, Raptor, Binds the mTOR Substrates p70 S6 Kinase and 4E-BP1 through Their TOR Signaling (TOS) Motif. J Biol Chem. 2003;278(18): 15461-15464.
[28] JACINTO E, LOEWITH R, SCHMIDT A, et al. Mammalian TOR complex 2 controls the actin cytoskeleton and is rapamycin insensitive. Nat Cell Biol. 2004;6(11):1122-1128.
[29] LI X, CHANG B, WANG B, et al. Rapamycin promotes osteogenesis under inflammatory conditions. Mol Med Rep. 2017; 16: 8923-8929.
[30] SUN H, KIM JK, MORTENSEN RM, et al. Osteoblast-targeted Suppression of PPARγ Increases Osteogenesis through Activation of mTOR Signaling. Stem Cells. 2013;31: 2183-2192.
[31] LONG H, ZHU Y, LIN Z, et al. miR-381 modulates human bone mesenchymal stromal cells (BMSCs) osteogenesis via suppressing Wnt signaling pathway during atrophic nonunion development. Cell Death Dis. 2019;10: 470.
[32] XIE Y, CHEN Y, ZHANG L, et al. The roles of bone-derived exosomes and exosomal microRNAs in regulating bone remodelling. J Cell Mol Med. 2017;21: 1033-1041.
[33] MOHAMMED MK,SHAO C,WANG J,et al. Wnt/β-catenin signaling plays an ever-expanding role in stem cell self-renewal, tumorigenesis and cancer chemoresistance. Genes Dis.2016;3(1):11-40.
[34] CUI Y, LUAN J, LI H, et al. Exosomes derived from mineralizing osteoblasts promote ST2 cell osteogenic differentiation by alteration of microRNA expression. FEBS Lett. 2016; 590(1):185-192.
[35] HASSAN MQ, MAEDA Y, TAIPALEENMAKI H, et al. miR-218 directs a Wnt signaling circuit to promote differentiation of osteoblasts and osteomimicry of metastatic cancer cells. J Biol Chem. 2012;287(50):42084-42092.
[36] NEGISHI-KOGA T, SHINOHARA M, KOMATSU N, et al. Suppression of bone formation by osteoclastic expression of semaphorin 4D. Nature Med. 2011;17(11):1473-1480.
[37] SUN W, ZHAO C, LI Y, et al. Osteoclast-derived microRNA-containing exosomes selectively inhibit osteoblast activity. Cell Discov. 2016; 2:16015.
[38] LI D, LIU J, GUO B, et al. Osteoclast-derived exosomal miR-214-3p inhibits osteoblastic bone formation. Nature Commun. 2016;7:10872.
[39] DI LEVA G, CHEUNG DG, BUZZETTI M. miRNAs in bone metastasis. Exp Rev Endocrinol Metabol. 2017: 17446651.2017.1383893.
[40] HUYNH N, VONMOSS L, SMITH D, et al. Characterization of regulatory extracellular vesicles from osteoclasts. J Dent Res. 2016:0022034516633189.
[41] MARTINS M, RIBEIRO D, MARTINS A, et al. Extracellular vesicles derived from osteogenically induced human bone marrow mesenchymal stem cells can modulate lineage commitment. Stem Cell Rep. 2016; 6(3):284-291.
[42] QIN Y, WANG L, GAO Z, et al. Bone marrow stromal/stem cell-derived extracellular vesicles regulate osteoblast activity and differentiation in vitro and promote bone regeneration in vivo. Sci Rep. 2016; 6:21961.
[43] 陈燕,姜胜军,彭友俭.骨髓间充质干细胞来源的外泌体对成骨细胞增殖和分化的影响[J].口腔医学研究,2019,35(4):401-404.
[44] 刘一飞,王宇辰,朱昱,等.尿源性干细胞的外泌体修复骨不连的作用研究[J].上海医学,2019,42(7):411-417.
[45] LIANG X, ZHANG L, WANG S, et al. Exosomes secreted by mesenchymal stem cells promote endothelial cell angiogenesis by transferring miR-125a. J Cell Sci. 2016; 129(11):2182-2189.
[46] KUCHARZEWSKA P, CHRISTIANSON HC, WELCH JE, et al. Exosomes reflect the hypoxic status of glioma cells and mediate hypoxia-dependent activation of vascular cells during tumor development. Proc Natl Acad Sci U S A. 2013;110(18): 7312-7317.
[47] LI XC, CHEN CY, WEI LM, et al. Exosomes derived from endothelial progenitor cells attenuate vascular repair and accelerate reendothelialization by enhancing endothelial function. Cytotherapy. 2016;18(2):253-262.
[48] ABDELALI A, PIERRE-HENRI D, TAREK B, et al. Microparticles from patients with metabolic syndrome induce vascular hypo-reactivity via fas/fas-ligand pathway in mice. PLoS ONE. 2011; 6(11):e27809.
[49] SU T, XIAO Y, XIAO Y, et al. Bone marrow mesenchymal stem cells-derived exosomal mir-29b-3p regulates aging-associated insulin resistance. ACS Nano. 2019;13: 2450-2462.
[50] FURUTA T, MIYAKI S, ISHITOBI H, et al. Mesenchymal stem cell-derived exosomes promote fracture healing in a mouse model. Stem Cells Transl Med. 2016;5(12):1620-1630.
|