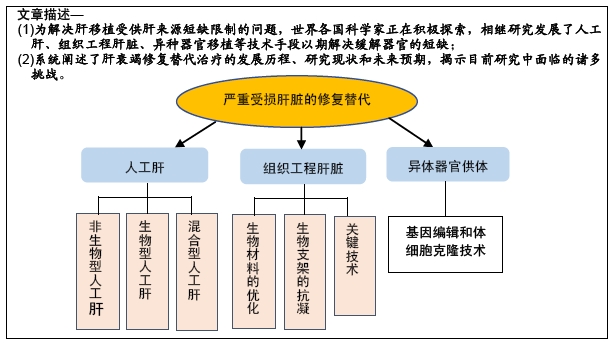
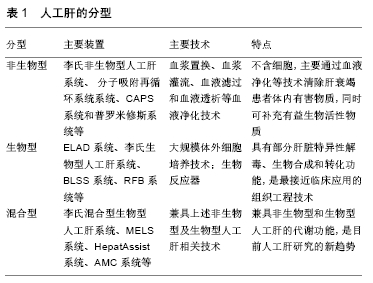
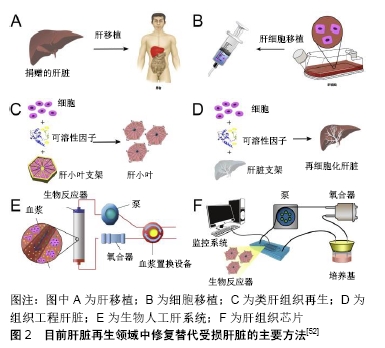
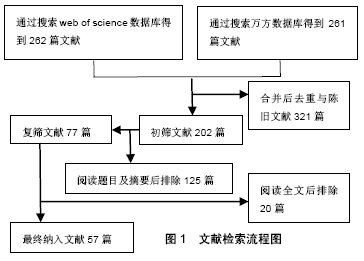
[1] 沈中阳,陆伟.中国肝移植乙型肝炎防治指南(2016版)[J].临床肝胆病杂志,2017,33(2):213-220.
[2] 刘昶荣.卫健委:2017年我国器官移植手术量世界第二. https://baijiahao.baidu.com/s?id=1607589663010107836,中国青年报中青在线.2018-08-01.
[3] 中华人民共和国国家卫生健康委员会.2019年最新公布:169所器官移植医疗机构名单. http://www.nhc.gov.cn/wjw/qgyzjg/ 201902/f2f2bb84e2ac4cf5993e9c7c5d30def3.shtml2019-02-25.
[4] 朱继业,高鹏骥.我国公民逝世后器官捐献肝移植现状[J].中华肝脏外科手术学电子杂志,2016,5(5):273-276.
[5] 范晓礼,叶啟发,王彦峰,等.比较心脏死亡器官捐赠与脑死亡器官捐赠供肝肝移植预后的荟萃分析[J].中华器官移植杂志, 2014, 35(2):86-93.
[6] 谢文勇,李涛. 活体肝移植的研究进展[J].器官移植,2018,9(4): 250-254.
[7] EUROPEAN ASSOCIATION FOR THE STUDY OF THE LIVER. EASL Clinical Practice Guidelines: Liver transplantation. J Hepatol.2016;64(2):433-485.
[8] ZHANG J, ZHAO X, LIANG L, et al.A decade of progress in liver regenerative medicine Biomaterials. 2018;157: 161-176.
[9] STRAIN AJ, NEUBERGER JM. A bioartificial liver--state of the art. Science. 2002; 295(5557): 1005-1009.
[10] MEDICINE POR.Principles of Regenerative Medicine. Elsevier Inc. 2008:928-945.
[11] ENOSAWA S, MIYASHITA T, TANAKA H, et al.Prolongation of survival of pigs with ischemic liver failure by treatment with a bioartificial liver using glutamine synthetase transfected recombinant HepG2. Transplant Proc. 2001;33(1-2): 1945-1947.
[12] NIU D, WEI HJ, LIN L, et al.Inactivation of porcine endogenous retrovirus in pigs using CRISPR-Cas9. Science. 2017;357(6357):1303-1307.
[13] LIU Q, LIU Z, DALAKAS E. Prevalence of porcine endogenous retrovirus in Chinese pig breeds and in patients treated with a porcine liver cell-based bioreactor. World J Gastroenterol. 2005;11(30):4727-4730.
[14] DUAN Y, MA X, MA X, et al. Differentiation and characterization of metabolically functioning hepatocytes from human embryonic stem cells.Stem Cells. 2010;28(4): 674-686.
[15] SHI XL, GAO Y, YAN Y, et al.Improved survival of porcine acute liver failure by a bioartificial liver device implanted with induced human functional hepatocytes. Cell Res. 2016;26(2): 206-216.
[16] ZHANG K, ZHANG L, LIU W, et al.In Vitro Expansion of Primary Human Hepatocytes with Efficient Liver Repopulation Capacity. Cell Stem Cell.2018;23(6):806-819.e4.
[17] FU GB, HUANG WJ, ZENG M, et al.Expansion and differentiation of human hepatocyte-derived liver progenitor-like cells and their use for the study of hepatotropic pathogens. Cell Res. 2019;29(1):8-22.
[18] LI J, LI LJ, CAO HC, et al.Establishment of highly differentiated immortalized human hepatocyte line with simian virus 40 large tumor antigen for liver based cell therapy. ASAIO J.2005;51(3):262-268.
[19] LEE SY, KIM HJ, CHOI D.Cell sources, liver support systems and liver tissue engineering: alternatives to liver transplantation. Int J Stem Cells.2015;8(1):36-47.
[20] MILLIS JM, CRONIN DC, JOHNSON R, et al. Initial experience with the modified extracorporeal liver-assist device for patients with fulminant hepatic failure: system modifications and clinical impact. Transplantation.2002; 74(12):1735-1746.
[21] LV G, ZHAO L, ZHANG A, et al.Bioartificial liver system based on choanoid fluidized bed bioreactor improve the survival time of fulminant hepatic failure pigs. Biotechnol Bioeng. 2011; 108(9):2229-2236.
[22] MAZARIEGOS GV, PATZER JF, LOPEZ RC, et al.First clinical use of a novel bioartificial liver support system (BLSS).Am J Transplant.2002;2(3):260-266.
[23] QIAN Y, LANJUAN L, JIANRONG H, et al.Study of severe hepatitis treated with a hybrid artificial liver support system.Int J Artif Organs.2003;26(6):507-513.
[24] DEMETRIOU AA, BROWN RS, BUSUTTIL RW, et al. Prospective, randomized, multicenter, controlled trial of a bioartificial liver in treating acute liver failure. Ann Surg. 2004;239(5):660-667; discussion 667-670.
[25] SAUER IM, KARDASSIS D, ZEILLINGER K, et al.Clinical extracorporeal hybrid liver support--phase I study with primary porcine liver cells.Xenotransplantation.2003;10(5): 460-469.
[26] HOLLISTER SJ, MURPHY WL.Scafold translation:barriers between concept and clinic. Tissue Eng Part B Rev. 2011;17(6): 459-474.
[27] YANNAS IV, BURKE JF, GORDON PL, et al. Design of an artificial skin. II. Control of chemical composition.J Biomed Mater Res.1980;14(2):107-132.
[28] VACANTI JP, MORSE MA, SALTZMAN WM, et al. Selective cell transplantation using bioabsorbable artificial polymers as matrices.J Pediatr Surg.1988;23(1 Pt 2):3-9.
[29] SOTO-GUTIERREZ A, ZHANG L, MEDBERRY C, et al.A whole-organ regenerative medicine approach for liver replacement. Tissue Eng Part C Methods. 2011;17(6): 677-686.
[30] LI J, LI L, YU H, et al.Growth and metabolism of human hepatocytes on biomodified collagen poly(lactic-co-glycolic acid) three-dimensional scaffold.ASAIO J. 2006;52(3): 321-327.
[31] JANORKAR AV, RAJAGOPALAN P, YARMUSH ML, et al. The use of elastin-like polypeptide-polyelectrolyte complexes to control hepatocyte morphology and function in vitro. Biomaterials.2008;29(6):625-632.
[32] KIM IY, SEO SJ, MOON HS, et al.Chitosan and its derivatives for tissue engineering applications. Biotechnol Adv. 2008; 26(1):1-21.
[33] UYGUN BE, SOTO-GUTIERREZ A, YAGI H, et al. Organ reengineering through development of a transplantable recellularized liver graft using decellularized liver matrix. Nat Med.2010;16(7): 814-820.
[34] BAO J, SHI Y, SUN H, et al. Construction of a portal implantable functional tissue-engineered liver using perfusion-decellularized matrix and hepatocytes in rats. Cell Transplant.2011;20(5): 753-766.
[35] GUTHRIE R. Review and management of side effects associated with antiplatelet therapy for prevention of recurrent cerebrovascular events. Adv Ther.2011;28(6):473-482.
[36] ROJAS-HERNANDEZ CM, GARCIA DA.The novel oral anticoagulants. Semin Thromb Hemost.2013; 39(2):117-126.
[37] DUQUE SL, BRACK N, POSTMA A, et al. Surface modification of electrospun fibres for biomedical applications: A focus on radical polymerization methods. Biomaterials. 2016;106:24-45.
[38] ZHANG J, SENGER B, VAUTIER D, et al. Natural polyelectrolyte films based on layer-by layer deposition of collagen and hyaluronic acid. Biomaterials. 2005;26(16): 3353-3361.
[39] SAMUEL RE, SHUKLA A, PAIK DH, et al. Osteoconductive protamine-based polyelectrolyte multilayer functionalized surfaces. Biomaterials.2011;32(30):7491-502.
[40] SÁNCHEZ PL, FERNÁNDEZ-SANTOS ME, COSTANZA S, et al. Acellular human heart matrix: A critical step toward whole heart grafts.Biomaterials.2015;61:279-289.
[41] SONG X, SHI L, CHEN L, et al. Endothelial cells modified by adenovirus vector containing nine copies hypoxia response elements and human vascular endothelial growth factor as the novel seed cells for bone tissue engineering.Acta Biochim Biophys Sin (Shanghai).2017;49(11): 973-978.
[42] KO IK, PENG L, PELOSO A, et al.Bioengineered transplantable porcine livers with re-endothelialized vasculature. Biomaterials.2015;40:72-79.
[43] KIM JJ, HOU L, HUANG NF. Vascularization of three-dimensional engineered tissues for regenerative medicine applications. Acta Biomater.2016;41:17-26.
[44] REN X, MOSER PT, GILPIN SE, et al. Engineering pulmonary vasculature in decellularized rat and human lungs. Nat Biotechnol. 2015;33(10):1097-1102.
[45] HONG M, JO H, ANKENY RF, et al.Influence of mesenchymal stem cells on the response of endothelial cells to laminar flow and shear stress.Cells Tissues Organs.2013;198(4): 289-299.
[46] MAHDAVI SN, BAHARARA J, TAKBIRI M, KHAJEH AS. In vitro Decellularization of Rabbit Lung Tissue. Cell J. 2013; 15(1): 83-88.
[47] SULLIVAN DC, MIRMALEK-SANI SH, DEEGAN DB, et al. Decellularization methods of porcine kidneys for whole organ engineering using a high-throughput system. Biomaterials. 2012;33(31): 7756-7764.
[48] REMLINGER NT, WEARDEN PD, GILBERT TW.Procedure for decellularization of porcine heart by retrograde coronary perfusion.J Vis Exp.2012;(70):e50059.
[49] ARENAS-HERRERA JE, KO IK, ATALA A, YOO JJ. Decellularization for whole organ bioengineering. Biomed Mater. 2013;8(1):014106.
[50] BADYLAK SF, TAYLOR D, UYGUN K. Whole-organ tissue engineering: decellularization and recellularization of three-dimensional matrix scaffolds. Annu Rev Biomed Eng. 2011;13:27-53.
[51] GUI L, CHAN SA, BREUER CK, et al. Novel utilization of serum in tissue decellularization. Tissue Eng C Methods.2010; 16(2):173-184.
[52] TAKEBE T, SEKINE K, ENOMURA M, et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature.2013;499(7459):481-484.
[53] BAPTISTA PM, SIDDIQUI MM, LOZIER G, et al. The use of whole organ decellularization for the generation of a vascularized liver organoid. Hepatology.2011;53(2):604-617.
[54] MURPHY SV, ATALA A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32(8):773-785.
[55] MA X, QU X, ZHU W, et al. Deterministically patterned biomimetic human iPSC-derived hepatic model via rapid 3D bioprinting. Proc Natl Acad Sci U S A.2016;113(8):2206-2211.
[56] LEE JW, CHOI YJ, YONG WJ, et al. Development of a 3D cell printed construct considering angiogenesis for liver tissue engineering. Biofabrication. 2016;8(1): 015007.
[57] YUAN L, JIANG J, LIU X, et al. HBV infection-induced liver cirrhosis development in dual-humanised mice with human bone mesenchymal stem cell transplantation. Gut. 2019; 68(11):2044-2056.
|