中国组织工程研究 ›› 2021, Vol. 25 ›› Issue (1): 133-140.doi: 10.3969/j.issn.2095-4344.2148
• 干细胞综述 stem cell review • 上一篇 下一篇
细胞因子和富血小板血浆对肌腱干细胞的影响
于承浩1,张 益 2,戚 超 2,陈进利 2,高甲科 2,于腾波 2
- 1青岛大学,山东省青岛市 266000;2青岛大学附属医院,山东省青岛市 266000
-
收稿日期:2020-03-27修回日期:2020-04-02接受日期:2020-05-09出版日期:2021-01-08发布日期:2020-11-20 -
通讯作者:于腾波,博士,青岛大学附属医院,山东省青岛市 266000 -
作者简介:于承浩,男,1995年生,山东省德州市人,青岛大学在读硕士,主要从事关节方面研究。 -
基金资助:山东省自然科学基金项目(ZR2019MH097)
Effect of cytokines and platelet-rich plasma on tendon derived stem cells
Yu Chenghao1, Zhang Yi2, Qi Chao2, Chen Jinli2, Gao Jiake2, Yu Tengbo2
- 1Qingdao University, Qingdao 266000, Shandong Province, China; 2Affiliated Hospital of Qingdao University, Qingdao 266000, Shandong Province, China
-
Received:2020-03-27Revised:2020-04-02Accepted:2020-05-09Online:2021-01-08Published:2020-11-20 -
Contact:Yu Tengbo, MD, Affiliated Hospital of Qingdao University, Qingdao 266000, Shandong Province, China -
About author:Yu Chenghao, Master candidate, Qingdao University, Qingdao 266000, Shandong Province, China -
Supported by:the Natural Science Foundation of Shandong Province, No. ZR2019MH097
摘要:
文题释义:
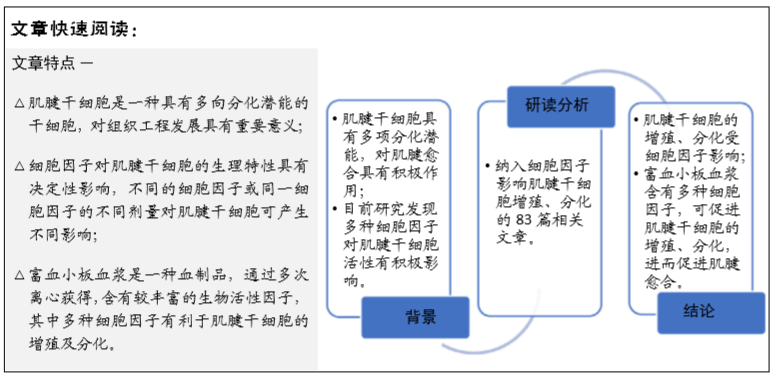
肌腱干细胞:是肌腱组织经消化后提取的具有自我更新及多向分化能力的细胞,不同于正常肌腱细胞,其表达干细胞标记物,近年来多项研究证明肌腱干细胞是组织移植治疗肌腱损伤的优势细胞。
富血小板血浆:是指全血通过梯度离心分层后得到的富含血小板的血浆部分,含有多种较丰富的生物活性因子,其血小板浓度为全血的3倍。
背景:肌腱干细胞存在于肌腱中,对于肌腱修复具有独特的功能,不同细胞因子对肌腱干细胞的增殖、分化功能具有不同影响;富血小板血浆是指全血通过梯度离心分层后得到的富含血小板的血浆部分,含有多种较丰富的细胞因子,有助于促使韧带和肌腱再生。
目的:探讨细胞因子和富血小板血浆对肌腱干细胞增殖、分化影响的最新进展。
方法:由第一作者以“tendon derived stem cells,tendon stem/progenitor cells,tendon stem cell,platelet-rich plasma,ligament injury”为英文关键词,以“肌腱干细胞,富血小板血浆,肌腱损伤”为中文关键词。检索 2007 至 2019 年期间收录在 PubMed、中国知网、万方等数据库中的文献,排除与文章研究目的无关及重复性的文献,纳入符合标准的 83篇文献进行综述。
结果与结论:肌腱干细胞是进行细胞移植治疗肌腱损伤的理想细胞,其增殖、分化受细胞因子影响,富血小板血浆包含大量细胞因子,具有刺激肌腱干细胞增殖、分化的功能,亦有成为细胞移植载体的潜力。探究细胞因子和富血小板血浆与肌腱干细胞增殖、分化的关系,将为肌腱干细胞的临床应用寻觅一条新途径。
https://orcid.org/0000-0001-5576-2975 (于承浩)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
于承浩, 张 益, 戚 超, 陈进利, 高甲科, 于腾波. 细胞因子和富血小板血浆对肌腱干细胞的影响[J]. 中国组织工程研究, 2021, 25(1): 133-140.
Yu Chenghao, Zhang Yi, Qi Chao, Chen Jinli, Gao Jiake, Yu Tengbo. Effect of cytokines and platelet-rich plasma on tendon derived stem cells[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(1): 133-140.
2.2 肌腱干细胞相对于其他干细胞的优势 肌腱干细胞和骨髓间充质干细胞与其他多种干细胞类型相比具有多种优势如增殖快、肌腱修复特异性强、再生时间短、成肌腱性优越等;同时,肌腱干细胞与骨髓间充质干细胞相比具有较高的成软骨和成骨分化潜能。BI等[7]研究发现,与小鼠骨髓间充质干细胞相比,小鼠肌腱干细胞表达更多的scleraxis、软骨寡聚蛋白、SOX9和Runx2,人肌腱干细胞比人骨髓间充质干细胞表达更高水平的腱调蛋白。TAN等[10]发现,与骨髓间充质干细胞相比,肌腱干细胞表达更高水平的Oct4,具有更强的增殖和克隆形成能力。在另一项小鼠研究中,肌腱干细胞治疗跟腱断裂时,Ⅲ型胶原蛋白、基因表达高于骨髓间充质干细胞,从而表现出更高的再生潜能[5]。骨髓间充质干细胞在兔肌腱损伤模型肌腱内形成了异位骨[15]。因此,肌腱干细胞相对于骨髓间充质干细胞在腱骨愈合方面更具优势,是理想的肌腱-骨交界处再生的候选细胞[9]。
2.3 细胞因子对肌腱干细胞的作用 大量研究证实肌腱干细胞在肌腱疾病和肌腱组织工程进展中起重要作用,肌腱干细胞的生物学特性受到多种因素的影响,这就是为什么许多肌腱相关损伤的治疗策略主要集中在肌腱干细胞上。
2.3.1 细胞因子对肌腱干细胞活性的作用 募集足够数量的肌腱干细胞是有效愈合的前提,而肌腱中肌腱干细胞数量极少,仅占1%-4%[7],因此需要为肌腱干细胞增殖制定标准化的分离、扩增、长期培养和明确的分化方案。
目前研究发现多种细胞因子对肌腱干细胞活性有积极影响。炎症因子白细胞介素6、白细胞介素10可促进肌腱干细胞的增殖与迁移。DENG等[16]及CHEN等[17]发现晚期炎症因子白细胞介素6、白细胞介素10可激活肌腱干细胞活性,使更多的细胞处于G2/M期,促进细胞快速增殖。另外,低、高剂量(0.5 nmol和5.0 nmol)的P物质均可显著增强肌腱干细胞的增殖能力[18]。HAN等[19]研究表明肝细胞生长因子激活HGF/c‐Met、MAPK/ERK1/2、PI3K/AKT信号通路,这些信号通路与肌腱干细胞扩散和迁移呈正相关。转化生长因子β1和胰岛素样生长因子1促进了肌腱干细胞的增殖和表型维
持[20]。色素上皮衍生因子衍生的短肽可在体外增强肌腱干细胞的增殖能力[21]。研究发现部分因素可对肌腱干细胞增殖产生不利影响。肌腱干细胞增殖能力受供者年龄影响。CD44是与细胞增殖呈现负相关的一项细胞因子,随着年龄的增长, 肌腱干细胞增殖能力降低,肌腱干细胞中CD44表达上调,CITED2表达下调[22]。此外,高糖环境促进肌腱干细胞中CD44的表达而抑制肌腱的增殖[23]。转化生长因子β2可以通过下调CITED2基因的表达而抑制肌腱干细胞的增殖[24]。罗毅等[25]发现转化生长因子β3可促进肌腱干细胞中成软骨标志物Sox9、ColⅡ的表达,诱导肌腱干细胞向软骨细胞分化。
局部环境氧化应激影响肌腱干细胞的细胞活性。LEE
等[26]应用不同浓度(100,200,500 μmol/L)的维生素C及过氧化氢处理人肌腱干细胞,发现低浓度的维生素C促进了细胞增殖、活力和迁移,而高浓度的维生素C及所有浓度的过氧化氢均影响了集落的形成,抑制了细胞的迁移、活力、凋亡和增殖,其原因可能是低水平维生素C由NOXs家族介导,通过激活ERK1/2和Jun-1/2途径促进肌腱干细胞的增殖和迁移。原花青素能保护过氧化氢诱导肌腱干细胞的氧化损伤,其保护作用可能是原花青素通过上调NRF-2信号通路激活GCLM、HO-1和NQO-1的表达[27],进而抵抗氧化损伤。常氧可以促进肌腱干细胞向成骨方向转化。在一项体外研究中,LI等[28]将肌腱干细胞分别在常氧与低氧中培养,发现低氧培养的肌腱干细胞增殖能力明显增强,而在常氧中培养的肌腱干细胞中细胞外信号调节激酶(ERK)1/2信号通路的激活更为明显,且成骨相关标志物(碱性磷酸酶、骨钙素和Runx2)的基因表达上调。
2.3.2 细胞因子对肌腱干细胞分化的作用 肌腱干细胞作为间充质干细胞的一种类型,具有成腱性、成骨性、成软骨性、成脂性等多项分化潜能[7],并在人和动物模型中形成肌腱、软骨、骨和腱-骨连接样组织。肌腱干细胞来源于肌腱,其成腱性对于肌腱组织工程治疗具有重要作用,可通过注射细胞因子诱导肌腱中肌腱干细胞向肌腱细胞分化,从而促进肌腱功能性愈合。首先,肌腱干细胞具有自发分化为肌腱的特性。GUO等[29]在大鼠肌腱干细胞体外研究中发现成腱转录因子Scx、Mkx、Egr1和Eya1在培养过程中上调。Scx是肌腱分化的关键调控因子,有人将含有Scx基因的慢病毒载体转导到肌腱干细胞后,增加了大多数与肌腱和软骨(Col2、Sox9、Acan和Bgn)相关标志物的表达,其中Ⅰ型胶原表达增加促进了损伤肌腱组织的愈合。SHUKUNAMI等[30]证明Scx以肌腱细胞系依赖的方式积极调节腱调蛋白的表达。
细胞因子具有调节细胞生成代谢、细胞免疫反应以及修复等生物学作用,是体内生理病理反应的重要调控因子。目前相关研究已经证明细胞因子对肌腱干细胞的分化方向也具有重要的调节作用。
结缔组织生长因子可促进肌腱干细胞的增殖及调节炎症因子的表达。TARAFDER等[31]将结缔组织生长因子注入全横断大鼠髌腱,可上调白细胞介素10的表达,并显著增加肌腱干细胞的数量。DENG等[16]及CHEN等[17]发现白细胞介素6、白细胞介素10可激活JAK/STAT3通路,降低Ⅰ型胶原和Ⅲ型胶原的表达水平。将结缔组织生长因子与其他细胞因子同时应用于肌腱干细胞,可对细胞分化产生不同的影响。XU等[32]将骨形态发生蛋白12和结缔组织生长因子共转染入大鼠膑腱肌腱干细胞,并将该肌腱干细胞移植入大鼠缺损的膑腱中,发现转染后肌腱干细胞的成腱分化基因激活,而成骨、脂肪生成和软骨标记基因均下调,同时骨形态发生蛋白12和结缔组织生长因子共转染组肌腱组织结构较对照组更为完整,最终破坏负荷高于对照组。LUI等[33]研究表明肌腱干细胞经结缔组织生长因子和抗坏血酸处理后形成细胞片,具有较高的成腱性,并维持了肌腱干细胞高软骨-成骨基因表达;接下来,Lui等[34]在一项大鼠肌腱缺损模型的体内及体外实验证明,结缔组织生长因子和抗坏血酸预处理的肌腱干细胞可通过smad 1/5/8信号通路上调肌腱相关蛋白的表达,促进肌腱干细胞成腱分化,具有更好的促进肌腱愈合作用。尽管结缔组织生长因子的功能前景看好,但它仍面临着几个问题,包括未知的受体、剂量的选择和不同细胞类型的功能等[35]。
P物质对肌腱干细胞的分化具有双重影响。研究发现,低剂量(0.5 nmol)P物质可诱导腱性相关基因Ⅰ型胶原的表
达[18],然而大剂量P物质(5.0 nmol)诱导非腱性相关基因过氧化物酶体增殖物激活受体γ和Ⅱ型胶原的表达。二甘聚糖被认为是肌腱干细胞生态位的重要组成部分之一,可促进肌腱干细胞的成腱性分化。ZHANG等[36]研究表明低质量浓度(50 μg/L)二甘聚糖干预后肌腱干细胞成腱标志物(SCX、THBS-4和TNMD)的表达水平普遍上调,并且发现二甘聚糖通过刺激骨形态发生蛋白7而促使Smad1/5/8磷酸化增加进而调控肌腱干细胞的成腱分化。在进一步实验中发现,Smad8磷酸化显著增高,Smad1/5磷酸化较少,这可能是Smad8蛋白被单独激活,从而使肌腱干细胞成腱性增强[36]。
骨形态发生蛋白2抑制肌腱干细胞的成腱性分化,促进非腱性分化。RUI等[37]发现骨形态发生蛋白2刺激后肌腱干细胞的成腱标志物(Dcn、Bgn和Fmod)表达下调,抑制成腱性分化,而GAG和Acan表达上调,细胞的非腱性分化增强。芮云峰等[38]进一步实验证明骨形态发生蛋白2可促进肌腱干细胞的成软骨标志物SOX9、ColⅡ的表达,促进细胞软骨分化。LUI等[39]在大鼠膑腱肌腱干细胞研究中,细胞受到骨形态发生蛋白2刺激后,肌腱干细胞总的Smad1/5/8磷酸化水平增高。骨形态发生蛋白2可能是通过Smad1/5/8信号通路影响肌腱干细胞的分化。LIU等[40]研究发现将胰岛素样生长因子1与骨形态发生蛋白2共同作用于肌腱干细胞时,成脂分化标志物PPARγ的表达明显上调,脂肪细胞数量也显著增加,并进一步证明其是通过激活CREB与Smad介导前列腺素E2来促进肌腱干细胞的成脂性分化。进一步研究发现,骨形态发生蛋白2和转化生长因子β1抑制肌腱干细胞的成腱相关蛋白(SCX、SOX9和TNMD)的表达,但促进小鼠和人骨髓间充质干细胞编码成骨相关蛋白(RUNX2和Osterix)的表达。前列腺素E2对人肌腱干细胞具有双相作用。低质量浓度
(<1 μg/L)的前列腺素E2在维持体内肌腱稳态中发挥重要作用,而高质量浓度(>1 μg/L)的前列腺素E2可降低细胞活性,并上调ColⅡ和骨钙素的表达,诱导非肌腱细胞分化[41]。ZHOU等[42]实验发现前列腺素E2通过促进骨形态发生蛋白2的产生诱导肌腱干细胞成骨性分化,PI3K-Akt信号通路在分化中占有重要地位,同时发现该信号因子位于Smad通路下游。
肝细胞生长因子抑制肌腱干细胞成骨性分化。HAN等[19]研究表明肝细胞生长因子可激活HGF/c-Met、MAPK/ERK1/2、PI3K/AKT信号通路,抑制肌腱干细胞成骨标志物基因ColⅠa1、ALP及Runx2的表达。碱性成纤维细胞生长因子可促进肌腱干细胞的成腱性分化。TOKUNAGA等[43]应用碱性成纤维细胞生长因子处理大鼠肌腱干细胞,细胞中肌腱相关基因(SCX、Tnmd、Sox9等)表达上调。VIGANò等[44]在实验中得到同样的结果。近期有研究表明,长链非编码RNA TUG1可通过促进碱性成纤维细胞生长因子泛素化进而促进肌腱干细胞的成骨分化,为肌腱修复提供了潜在的靶点[45]。
另外,碱性成纤维细胞生长因子过表达显著增加了肌腱干细胞中ColⅢ和SCX的表达[46]。VH298是一种针对HIF的新化合物,它可以促进肌腱干细胞的分化。在一项大鼠研究中,QIU等[47]应用30 μmol/L VH298处理肌腱干细胞,肌腱分化相关基因(ColⅠα、ColⅢα、decorin、Tnmd、tenascin C)及软骨分化相关基因(ColⅡα、SRY-box9及aggrecan)的表达显著增加。
各种细胞因子对肌腱干细胞的分化具有不同的影响,且细胞因子含量不同亦可得到不同的结论,如何搭配细胞因子,诱导肌腱干细胞向既定方向发展,仍需进一步实验探索。
2.4 其他因素对肌腱干细胞分化的影响 生物体内的血糖、胆固醇、维生素C以及药物都会对肌腱干细胞的分化产生影响。高糖可抑制肌腱干细胞的成腱性分化,诱导非肌腱分化。LIN等[48]应用不同浓度葡萄糖 (5.5,15,25 mmol/L)对体外培养的大鼠肌腱干细胞进行处理,研究发现高糖(15,
25 mmol/L)处理24 h及48 h后,肌腱干细胞中的成腱性标志物Scx、ColⅠ的mRNA表达及ColⅠ蛋白、Tnmd蛋白表达均下调。SHI等[23]在一项糖尿病小鼠肌腱干细胞研究中发现,高糖环境会使肌腱干细胞成腱标志物ColⅠ和Tnmd降低,同时肌腱干细胞骨软骨形成标志物BMP2、ALP、OPN、OCN、ColⅡ和SOX9显著升高,导致细胞的成骨、软骨分化能力明显增强。徐宏亮等[49]发现高糖环境抑制肌腱干细胞成脂标志物PPARγ的表达,且经过成脂分化培养后脂肪细胞数量少于对照组,说明高糖环境可以抑制肌腱干细胞向肌腱及脂肪方向分化。
高胆固醇抑制肌腱干细胞的成腱性分化。LI等[50]在大鼠肌腱干细胞体外实验中,应用不同浓度的胆固醇处理大鼠肌腱干细胞3 d,发现肌腱干细胞中肌腱相关基因(ColⅠ/Ⅲ、Tnmd、SCX等)表达降低,胆固醇浓度越高,降低越明显,肌腱相关基因表达降低与活性氧激活的NF-κB信号有关,胆固醇可能通过活性氧激活的NF-κB信号抑制肌腱干细胞中肌腱相关基因的表达,从而抑制肌腱干细胞的成腱分化。
Wnt信号通路是一个高度保守的通路,它调控着广泛的生物学过程,包括细胞分化、迁移和极性[51]。LUI等[52]发现Wnt3a可以增加肌腱干细胞中碱性磷酸酶活性、钙结节的形成和成骨标志物的表达。TIAN等[53]将肌腱干细胞置于含有黄芩素培养基中体外诱导14 d,ALP、Runx2、OCN、OSX等成骨分化标记物显著上调,同时Wnt/β-catenin信号通路也增强。这说明黄芩素可能通过刺激Wnt/β-catenin信号通路促使肌腱干细胞成骨分化,进而促进腱骨连接处愈合。
另有研究发现,塞来昔布可抑制肌腱干细胞的腱性分化,但对细胞增殖无影响[54]。JIANG等[55]发现瘦素通过mTORC1信号促进肌腱干细胞中碱性磷酸酶、Runx2、骨钙素表达,促进成骨分化和异位骨化形成,而雷帕霉素可以阻断瘦素诱导的异位骨化形成。WANG等[56]发现阿司匹林促进了TNC、TNMD、SCX的表达,另外生长分化因子7表达上调,能够激活Smad1/5信号通路,促进肌腱生成。白藜芦醇促进肌腱干细胞中成骨标志物Runx2和OCN的表达,且通过调节β-catenin信号通路来调控细胞的成骨分化[57]。
综上所述,所有这些因素都可能通过靶向作用于肌腱干细胞来影响细胞的生理功能,进而影响肌腱的愈合效果,寻找合适的治疗靶点对改善治疗效果具有重要意义。
2.5 富血小板血浆对肌腱干细胞的影响 如今,富血小板血浆已经成为研究热点之一。应用富血小板血浆治疗有注射及凝胶移植两种方式,且已被广泛应用于骨科及运动医学手术中,以促进肌肉骨骼组织的愈合[58]。现使用的富血小板血浆有4种:纯富血小板血浆、纯富血小板纤维蛋白、富白细胞富血小板血浆和富白细胞富血小板纤维蛋白[59]。在临床应用中,注射富血小板血浆可以改善肩袖损伤[60-61]、网球肘[62-64]、跟腱病[65]、髌腱病患者的疼痛强度和功能评分[66]。 目前,富血小板血浆对肌腱干细胞增殖分化的影响仍处于探索阶段,见表1。
富血小板血浆可促进肌腱干细胞增殖和迁移。多项研究表明,富血小板血浆在体外对肌腱干细胞的增殖具有良好的激活作用[67-69]。XU等[70]研究发现富血小板血浆不仅提高了细胞的活力,而且通过DNA合成启动了细胞的增殖,使大部分细胞保持在增殖期。KIM等[71]同样发现骨髓浓缩液联合富血小板血浆可增加肌腱干细胞的增殖速度,同时降低成软骨和成骨能力。这说明应用富血小板血浆治疗可以为肌腱修复提供足够的细胞数量。另外,富血小板血浆还能促进肌腱干细胞迁移和募集。富血小板血浆释放的因子中包括基质细胞衍生因子1,该因子被认为是一种强大的骨髓间充质干细胞募集诱导因子[72]。咸杰等[73]发现富血小板血浆与肌腱干细胞的共混物可抑制基质金属蛋白酶1的表达,从而延缓肌腱炎症性反应。因此,富血小板血浆可以募集肌腱干细胞,促进细胞增殖。
富血小板血浆可促进肌腱干细胞向腱细胞分化,抑制肌腱干细胞的非腱性分化。研究发现,富血小板血浆在体外对肌腱干细胞的肌腱相关性基因和蛋白表达具有良好的激活作用[69],被认为是肌腱干细胞分化为腱细胞而不是非腱细胞的“保障”因素。ZHANG等[74]应用富血小板凝块释放物(platelet-rich clot releasate,PRCR)培养肌腱干细胞,发现腱细胞相关基因(ColⅠ和ColⅢ、Tenascin C和Tnmd)的表达增加,但不表达非腱细胞相关基因,包括PPAR-γ(脂肪细胞)、SOX-9(软骨细胞)和Runx2(骨细胞),且富血小板凝块释放物通过降低脂联素(脂肪细胞)、ColⅡ(软骨细胞)和骨连素(骨细胞)的水平,从而有效抑制肌腱干细胞向非腱细胞分化。这与CHEN等[75]的研究结果相一致。另外,富血小板血浆以活化FAK和ERK1/2信号通路的方式促进肌腱干细胞的腱性分化。CHEN等[75]研究中富血小板血浆可诱导肌腱干细胞的FAK和ERK1/2信号通路活化,且对这两条通路中任一条进行抑制时,肌腱干细胞的成腱性标志物ColⅠ、SCX、Tenascin C表达减少。随后,TARAFDER等[35]对FAK或ERK1/2激动剂的小分子进行了筛选,发现Oxotremorine M (Oxo-M)和PPBP maleate (4-PPBP)具有极高的诱导肌腱干细胞成肌腱分化潜能。通过对大鼠膑腱缺损模型研究,发现Oxo-M和4-PPBP治疗2周后肌腱具有天然的正常宏观结构,功能性肌腱修复约为天然肌腱的83.4%,而对照组仅为隆起的瘢痕愈合。
不同类型的富血小板血浆对肌腱干细胞具有不同的影响。富白细胞富血小板血浆中的白细胞浓度比全血中的白细胞浓度高4倍,比纯富血小板血浆高8倍[76]。纯富血小板血浆比富白细胞富血小板血浆更有利于肌腱干细胞增殖及成腱分化。ZHANG等[77]研究发现纯富血小板血浆可促进肌腱干细胞增殖,在10%浓度时效果最为显著,并诱导肌腱干细胞产生胶原蛋白,纯富血小板血浆可产生更多的血管内皮生长因子、表皮生长因子、转化生长因子β1和血小板衍生生长因子。转化生长因子β1可以促进肌腱干细胞的增殖,并有利于维持细胞表型[20]。富白细胞富血小板血浆对肌腱干细胞有不利影响,进而影响损伤肌腱的愈合。富白细胞富血小板血浆培养的肌腱干细胞产生更多的炎症因子,如白细胞介素1β、肿瘤坏死因子α、基质金属蛋白酶1、基质金属蛋白酶13、膜相关前列腺素合成酶及前列腺素E2[76,78]。这些研究结果与YUAN等[79]的研究结果相符合。白细胞介素1β及前列腺素E2是肌腱急性损伤时特征性上调的炎症因子。研究发现白细胞介素1β可显著降低肌腱干细胞标志物SCX和Tnmd的表达,并下调肌腱干细胞中COLⅠ、COLⅢ、双聚糖和纤维调素的基因表达。最重要的是,它可以显著不可逆地抑制肌腱干细胞的腱性分化[80]。而前列腺素E2在高浓度时会抑制肌腱干细胞的增殖并诱导成骨性分化[81]。富白细胞富血小板血浆诱导COLⅢ的表达高于纯富血小板血浆,而COLⅠ的表达低于纯富血小板血浆[78]。由于肌腱中ColⅠ是主要成分,而在正常肌腱中只有少量的ColⅢ存在,在瘢痕愈合的肌腱中存在大量的ColⅢ,使用富白细胞富血小板血浆治疗损伤的肌腱可能会导致愈合肌腱中瘢痕形成。因此,富白细胞富血小板血浆抑制肌腱干细胞的增殖,促进肌腱干细胞非腱性分化,降低生长因子水平,诱导肌腱干细胞的凋亡。
富血小板血浆中富含血小板,血小板中包含不同的α颗粒的亚群,亚群的分泌受凝血酶受体的调节。激活不同的受体亦可以影响富血小板血浆对肌腱干细胞的作用。ZHANG
等[77]的一项研究分别激活富血小板血浆的PAR1和PAR4,然后将其作用于人肌腱干细胞,发现PAR1-PRP选择性释放血管内皮生长因子,使肌腱干细胞产生了一种“血管样”的细胞模式,并增加了COLⅠ及MMP-1的表达;而PAR4-PRP选择性释放内皮抑素,以平行的方式诱导细胞组织并增加MMP-2的表达。据研究,血管内皮生长因子在胎儿和受伤的人体肌腱细胞中高度表达;MMP-1在瘢痕组织中低水平表
达[82],而MMP-2在瘢痕组织中高表达。在损伤部位表达的血管内皮生长因子可以有双重影响,既能促进血管生成,也可能导致组织中瘢痕的形成[83]。由于急性损伤的特点是高水平的血管内皮生长因子,因此PAR1-PRP不宜用于创伤部位已含有丰富血管内皮生长因子的急性肌腱损伤的治疗,以免增加瘢痕形成而影响愈合,而慢性愈合因缺乏足够的血管形成而愈合停滞,更适合使用PAR1-PRP进行治疗[77]。内皮抑素可减少血管生成,因此应用PAR4-PRP治疗急性损伤将减少过量血管内皮生长因子的影响,从而减少瘢痕形成。在动物实验中,PAR1-PRP组比PAR4-PRP组诱导了更多的血管形成,且产生了更多无序的瘢痕组织,而后者诱导了更多的有序的组织生成[77]。因此,应用不同的凝血酶受体激活剂将对肌腱愈合产生不同影响。

| [1] KJAER M. Role of extracellular matrix in adaptation of tendon and skeletal muscle to mechanical loading. Physiol Rev. 2004;84(2): 649-698. [2] PENNISI E. Tending tender tendons. Science. 2002;295(5557):1011. [3] BUTLER DL, JUNCOSA N, DRESSLER MR. Functional efficacy of tendon repair processes. Annu Rev Biomed Eng. 2004;6:303-329. [4] CHEUNG TS, LAU PM, LU H, et al. Cytotoxic and sublethal effects of silver nanoparticles on tendon-derived stem cells - implications for tendon engineering. Toxicol Res (Camb). 2015;5(1):318-330. [5] AL-ANI MKH, XU K, SUN Y, et al. Study of Bone Marrow Mesenchymal and Tendon-Derived Stem Cells Transplantation on the Regenerating Effect of Achilles Tendon Ruptures in Rats. Stem Cells Int. 2015; 2015: 984146. [6] WU T, LIU Y, WANG B, et al. The Use of Cocultured Mesenchymal Stem Cells with Tendon-Derived Stem Cells as a Better Cell Source for Tendon Repair. Tissue Eng Part A. 2016;22(19-20):1229-1240. [7] BI Y, EHIRCHIOU D, KILTS TM, et al. Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche. Nat Med. 2007;13(10):1219-1227. [8] NI M, LUI PP, RUI YF, et al. Tendon-derived stem cells (TDSCs) promote tendon repair in a rat patellar tendon window defect model. J Orthop Res. 2012;30(4):613-619. [9] LUI PP, CHAN KM. Tendon-derived stem cells (TDSCs): from basic science to potential roles in tendon pathology and tissue engineering applications. Stem Cell Rev Rep. 2011;7(4):883-897. [10] TAN Q, LUI PP, RUI YF, et al. Comparison of potentials of stem cells isolated from tendon and bone marrow for musculoskeletal tissue engineering. Tissue Eng Part A. 2012;18(7-8):840-851. [11] ZHANG Q, CHENG B. Tendon-derived stem cells as a new cell source for tendon tissue engineering. Front Biosci (Landmark Ed). 2013;18:756-764. [12] LIN Y, ZHANG L, LIU NQ, et al. In vitro behavior of tendon stem/progenitor cells on bioactive electrospun nanofiber membranes for tendon-bone tissue engineering applications. Int J Nanomedicine. 2019;14:5831-5848. [13] WANG T, THIEN C, WANG C, et al. 3D uniaxial mechanical stimulation induces tenogenic differentiation of tendon-derived stem cells through a PI3K/AKT signaling pathway. FASEB J. 2018;32(9):4804-4814. [14] SCREEN HR, BERK DE, KADLER KE, et al. Tendon functional extracellular matrix. J Orthop Res. 2015;33(6):793-799. [15] HARRIS MT, BUTLER DL, BOIVIN GP, et al. Mesenchymal stem cells used for rabbit tendon repair can form ectopic bone and express alkaline phosphatase activity in constructs. J Orthop Res. 2004;22(5):998-1003. [16] DENG G, LI K, CHEN S, et al. Interleukin‑10 promotes proliferation and migration, and inhibits tendon differentiation via the JAK/Stat3 pathway in tendon‑derived stem cells in vitro. Mol Med Rep. 2018;18(6):5044-5052. [17] CHEN S, DENG G, LI K, et al. Interleukin-6 Promotes Proliferation but Inhibits Tenogenic Differentiation via the Janus Kinase/Signal Transducers and Activators of Transcription 3 (JAK/STAT3) Pathway in Tendon-Derived Stem Cells. Med Sci Monit. 2018;24:1567-1573. [18] ZHOU Y, ZHOU B, TANG K. The effects of substance p on tendinopathy are dose-dependent: an in vitro and in vivo model study. J Nutr Health Aging. 2015;19(5):555-561. [19] HAN P, CUI Q, LU W, et al. Hepatocyte growth factor plays a dual role in tendon-derived stem cell proliferation, migration, and differentiation. J Cell Physiol. 2019;234(10):17382-17391. [20] HOLLADAY C, ABBAH SA, O’DOWD C, et al. Preferential tendon stem cell response to growth factor supplementation. J Tissue Eng Regen Med. 2016;10(9):783-798. [21] HO TC, TSAI SH, YEH SI, et al. PEDF-derived peptide promotes tendon regeneration through its mitogenic effect on tendon stem/progenitor cells. Stem Cell Res Ther. 2019;10(1):2. [22] ZHOU Z, AKINBIYI T, XU L, et al. Tendon-derived stem/progenitor cell aging: defective self-renewal and altered fate. Aging Cell. 2010; 9(5):911-915. [23] SHI L, LI YJ, DAI GC, et al. Impaired function of tendon-derived stem cells in experimental diabetes mellitus rat tendons: implications for cellular mechanism of diabetic tendon disorder. Stem Cell Res Ther. 2019;10(1):27. [24] HU C, ZHANG Y, TANG K, et al. Downregulation of CITED2 contributes to TGFβ-mediated senescence of tendon-derived stem cells. Cell Tissue Res. 2017;368(1):93-104. [25] 罗毅,汪敏加,苏全生,等.富血小板血浆(PRP)凝胶联合TGF-β3对肌腱干细胞成软骨分化影响的实验研究[J].广西大学学报(自然科学版), 2019,44(6): 1781-1789. [26] LEE YW, FU SC, YEUNG MY, et al. Effects of Redox Modulation on Cell Proliferation, Viability, and Migration in Cultured Rat and Human Tendon Progenitor Cells. Oxid Med Cell Longev. 2017;2017:8785042. [27] SUN W, MENG J, WANG Z, et al. Proanthocyanidins Attenuation of H2O2-Induced Oxidative Damage in Tendon-Derived Stem Cells via Upregulating Nrf-2 Signaling Pathway. Biomed Res Int. 2017; 2017:7529104. [28] LI P, XU Y, GAN Y, et al. Role of the ERK1/2 Signaling Pathway in Osteogenesis of Rat Tendon-Derived Stem Cells in Normoxic and Hypoxic Cultures. Int J Med Sci. 2016;13(8):629-637. [29] GUO J, CHAN KM, ZHANG JF, et al. Tendon-derived stem cells undergo spontaneous tenogenic differentiation. Exp Cell Res. 2016;341(1):1-7. [30] SHUKUNAMI C, TAKIMOTO A, ORO M, et al. Scleraxis positively regulates the expression of tenomodulin, a differentiation marker of tenocytes. Dev Biol. 2006;298(1):234-247. [31] TARAFDER S, CHEN E, JUN Y, et al. Tendon stem/progenitor cells regulate inflammation in tendon healing via JNK and STAT3 signaling. FASEB J. 2017;31(9):3991-3998. [32] XU K, SUN Y, KH AL-ANI M, et al. Synergistic promoting effects of bone morphogenetic protein 12/connective tissue growth factor on functional differentiation of tendon derived stem cells and patellar tendon window defect regeneration. J Biomech. 2018;66:95-102. [33] LUI PP, WONG OT, LEE YW. Application of tendon-derived stem cell sheet for the promotion of graft healing in anterior cruciate ligament reconstruction. Am J Sports Med. 2014;42(3):681-689. [34] LUI PP, WONG OT, LEE YW. Transplantation of tendon-derived stem cells pre-treated with connective tissue growth factor and ascorbic acid in vitro promoted better tendon repair in a patellar tendon window injury rat model. Cytotherapy. 2016;18(1):99-112. [35] TARAFDER S, RICUPERO C, MINHAS S, et al. A Combination of Oxo-M and 4-PPBP as a potential regenerative therapeutics for tendon injury. Theranostics. 2019;9(14):4241-4254. [36] ZHANG YJ, QING Q, ZHANG YJ, et al. Enhancement of tenogenic differentiation of rat tendon-derived stem cells by biglycan. J Cell Physiol. 2019 Feb 4. doi: 10.1002/jcp.28247. Online ahead of print. [37] RUI YF, LUI PP, WONG YM, et al. BMP-2 stimulated non-tenogenic differentiation and promoted proteoglycan deposition of tendon-derived stem cells (TDSCs) in vitro. J Orthop Res. 2013;31(5):746-753. [38] 芮云峰,郭永刚,林禹丞,等.BMP-2诱导人跟腱来源肌腱干细胞成软骨分化的实验研究[J].中国修复重建外科杂志, 2013,27(12): 1492-1498. [39] LUI PP, WONG Y. Higher BMP/Smad sensitivity of tendon-derived stem cells (TDSCs) isolated from the collagenase-induced tendon injury model: possible mechanism for their altered fate in vitro. BMC Musculoskelet Disord. 2013;14:248. [40] LIU J, CHEN L, ZHOU Y, et al. Insulin-like growth factor-1 and bone morphogenetic protein-2 jointly mediate prostaglandin E2-induced adipogenic differentiation of rat tendon stem cells. PLoS One. 2014; 9(1):e85469. [41] ZHANG J, WANG JH. Prostaglandin E2 (PGE2) exerts biphasic effects on human tendon stem cells. PLoS One. 2014;9(2):e87706. [42] ZHOU W, LIN X, CHU J, et al. Magnolol prevents ossified tendinopathy by inhibiting PGE2-induced osteogenic differentiation of TDSCs. Int Immunopharmacol. 2019;70:117-124. [43] TOKUNAGA T, SHUKUNAMI C, OKAMOTO N, et al. FGF-2 Stimulates the Growth of Tenogenic Progenitor Cells to Facilitate the Generation of Tenomodulin-Positive Tenocytes in a Rat Rotator Cuff Healing Model. Am J Sports Med. 2015;43(10):2411-2422. [44] VIGANÒ M, PERUCCA ORFEI C, COLOMBINI A, et al. Different culture conditions affect the growth of human tendon stem/progenitor cells (TSPCs) within a mixed tendon cells (TCs) population. J Exp Orthop. 2017;4(1):8. [45] YU Y, CHEN Y, ZHENG YJ, et al. LncRNA TUG1 promoted osteogenic differentiation through promoting bFGF ubiquitination. In Vitro Cell Dev Biol Anim. 2020;56(1):42-48. [46] GUO D, LI H, LIU Y, et al. Fibroblast growth factor-2 promotes the function of tendon-derived stem cells in Achilles tendon restoration in an Achilles tendon injury rat model. Biochem Biophys Res Commun. 2020;521(1):91-97. [47] QIU S, JIA Y, TANG J, et al. Von Hippel-Lindau (VHL) protein antagonist, VH298, promotes functional activities of tendon-derived stem cells and accelerates healing of entheses in rats by inhibiting ubiquitination of hydroxy-HIF-1α. Biochem Biophys Res Commun. 2018;505(4):1063-1069. [48] LIN YC, LI YJ, RUI YF, et al. The effects of high glucose on tendon-derived stem cells: implications of the pathogenesis of diabetic tendon disorders. Oncotarget. 2017;8(11):17518-17528. [49] 徐宏亮,石柳,李荥娟,等.糖尿病大鼠髌腱来源肌腱干细胞体外成脂分化潜能的实验研究[J].东南大学学报(医学版), 2018,37(3): 390-395. [50] LI K, DENG G, DENG Y, et al. High Cholesterol Inhibits Tendon-Related Gene Expressions in Tendon-Derived Stem Cells Through Reactive Oxygen Species-Activated Nuclear factor-κB SignalingJ Cell Physiol. 2019;234(10):18017-18028. [51] HUANG AH, LU HH, SCHWEITZER R. Molecular regulation of tendon cell fate during development. J Orthop Res. 2015;33(6):800-812. [52] LUI PP, LEE YW, WONG YM, et al. Expression of Wnt pathway mediators in metaplasic tissue in animal model and clinical samples of tendinopathy. Rheumatology (Oxford). 2013;52(9):1609-1618. [53] TIAN X, JIANG H, CHEN Y, et al. Baicalein Accelerates Tendon-Bone Healing via Activation of Wnt/β-Catenin Signaling Pathway in Rats. Biomed Res Int. 2018;2018:3849760. [54] ZHANG K, ZHANG S, LI Q, et al. Effects of celecoxib on proliferation and tenocytic differentiation of tendon-derived stem cells. Biochem Biophys Res Commun. 2014;450(1):762-766. [55] JIANG H, CHEN Y, CHEN G, et al. Leptin accelerates the pathogenesis of heterotopic ossification in rat tendon tissues via mTORC1 signaling. J Cell Physiol. 2018;233(2):1017-1028. [56] WANG Y, HE G, TANG H, et al. Aspirin promotes tenogenic differentiation of tendon stem cells and facilitates tendinopathy healing through regulating the GDF7/Smad1/5 signaling pathway. J Cell Physiol. 2020;235(5):4778-4789. [57] 王文,秦胜男,陈鸿辉,等.白藜芦醇通过抑制β-catenin表达诱导并促进人肌腱干细胞的成骨分化[J]. 中国矫形外科杂志, 2015, 23(19):1793-1800. [58] WANG J, ZHANG J, NIRMALA X, et al. Advancements in the treatment and repair of tendon injuries. Current Tissue Engineering. 2014; 3(2): 71-81. [59] DOHAN EHRENFEST DM, RASMUSSON L, ALBREKTSSON T. Classification of platelet concentrates: from pure platelet-rich plasma (P-PRP) to leucocyte- and platelet-rich fibrin (L-PRF). Trends Biotechnol. 2009; 27(3):158-167. [60] KIM SJ, YEO SM, NOH SJ, et al. Effect of platelet-rich plasma on the degenerative rotator cuff tendinopathy according to the compositions. J Orthop Surg Res. 2019;14(1):408. [61] MALAVOLTA EA, GRACITELLI MEC, ASSUNÇÃO JH, et al. Clinical and Structural Evaluations of Rotator Cuff Repair With and Without Added Platelet-Rich Plasma at 5-Year Follow-up: A Prospective Randomized Study. Am J Sports Med. 2018;46(13):3134-3141. [62] MARTIN JI, ATILANO L, BULLY P, et al. Needle tenotomy with PRP versus lidocaine in epicondylopathy: clinical and ultrasonographic outcomes over twenty months. Skeletal Radiol. 2019;48(9):1399-1409. [63] MARTIN JI, ATILANO L, MERINO J, et al. Platelet-rich plasma versus lidocaine as tenotomy adjuvants in people with elbow epicondylopathy: a randomized controlled trial. J Orthop Surg Res. 2019;14(1):109. [64] BODEN AL, SCOTT MT, DALWADI PP, et al. Platelet-rich plasma versus Tenex in the treatment of medial and lateral epicondylitis. J Shoulder Elbow Surg. 2019;28(1):112-119. [65] KEARNEY RS, PARSONS N, JI C, et al. Platelet rich plasma versus placebo for the management of Achilles tendinopathy: protocol for the UK study of Achilles tendinopathy management (ATM) multi-centre randomised trial. BMJ Open. 2020;10(2):e034076. [66] ANDRIOLO L, ALTAMURA SA, REALE D, et al. Nonsurgical Treatments of Patellar Tendinopathy: Multiple Injections of Platelet-Rich Plasma Are a Suitable Option: A Systematic Review and Meta-analysis. Am J Sports Med. 2019;47(4):1001-1018. [67] HOPPE S, ALINI M, BENNEKER LM, et al. Tenocytes of chronic rotator cuff tendon tears can be stimulated by platelet-released growth factors. J Shoulder Elbow Surg. 2013;22(3):340-349. [68] JO CH, KIM JE, YOON KS, et al. Platelet-rich plasma stimulates cell proliferation and enhances matrix gene expression and synthesis in tenocytes from human rotator cuff tendons with degenerative tears. Am J Sports Med. 2012;40(5):1035-1045. [69] ZHANG J, WANG JH. Characterization of differential properties of rabbit tendon stem cells and tenocytes. BMC Musculoskelet Disord. 2010;11:10. [70] XU K, AL-ANI MK, SUN Y, et al. Platelet-rich plasma activates tendon-derived stem cells to promote regeneration of Achilles tendon rupture in rats. J Tissue Eng Regen Med. 2017;11(4):1173-1184. [71] KIM SJ, SONG DH, PARK JW, et al. Effect of Bone Marrow Aspirate Concentrate-Platelet-Rich Plasma on Tendon-Derived Stem Cells and Rotator Cuff Tendon Tear. Cell Transplant. 2017;26(5):867-878. [72] GUO K, YAO X, WU W, et al. HIF-1α/SDF-1/CXCR4 axis reduces neuronal apoptosis via enhancing the bone marrow-derived mesenchymal stromal cell migration in rats with traumatic brain injury. Exp Mol Pathol. 2020;114:104416. [73] 咸杰,何本祥,吴骁,等.富血小板血浆与肌腱干细胞共混物干预跟腱病模型兔肌腱组织形态及金属蛋白酶1的表达[J].中国组织工程研究, 2018,22(6):921-926. [74] ZHANG J, WANG JH. PRP treatment effects on degenerative tendinopathy - an in vitro model study. Muscles Ligaments Tendons J. 2014;4(1):10-17. [75] CHEN L, LIU JP, TANG KL, et al. Tendon derived stem cells promote platelet-rich plasma healing in collagenase-induced rat achilles tendinopathy. Cell Physiol Biochem. 2014;34(6):2153-2168. [76] ZHANG L, CHEN S, CHANG P, et al. Harmful Effects of Leukocyte-Rich Platelet-Rich Plasma on Rabbit Tendon Stem Cells In Vitro. Am J Sports Med. 2016;44(8):1941-1951. [77] ZHANG J, NIE D, WILLIAMSON K, et al. Selectively activated PRP exerts differential effects on tendon stem/progenitor cells and tendon healing. J Tissue Eng. 2019;10:2041731418820034. [78] ZHOU Y, ZHANG J, WU H, et al. The differential effects of leukocyte-containing and pure platelet-rich plasma (PRP) on tendon stem/progenitor cells - implications of PRP application for the clinical treatment of tendon injuries. Stem Cell Res Ther. 2015;6(1):173. [79] YUAN T, GUO SC, HAN P, et al. Applications of leukocyte- and platelet-rich plasma (L-PRP) in trauma surgery. Curr Pharm Biotechnol. 2012; 13(7):1173-1184. [80] ZHANG K, ASAI S, YU B, et al. IL-1β irreversibly inhibits tenogenic differentiation and alters metabolism in injured tendon-derived progenitor cells in vitro. Biochem Biophys Res Commun. 2015;463(4):667-672. [81] ZHANG J, WANG JH. BMP-2 mediates PGE(2) -induced reduction of proliferation and osteogenic differentiation of human tendon stem cells. J Orthop Res. 2012;30(1):47-52. [82] ZHANG J, WANG JH. The effects of mechanical loading on tendons--an in vivo and in vitro model study. PLoS One. 2013;8(8):e71740. [83] CHEN J, YU Q, WU B, et al. Autologous tenocyte therapy for experimental Achilles tendinopathy in a rabbit model. Tissue Eng Part A. 2011;17(15-16):2037-2048. |
| [1] | 蒲 锐, 陈子扬, 袁凌燕. 不同细胞来源外泌体保护心脏的特点与效应[J]. 中国组织工程研究, 2021, 25(在线): 1-. |
| [2] | 林清凡, 解一新, 陈婉清, 叶振忠, 陈幼芳. 人胎盘源间充质干细胞条件培养液可上调缺氧状态下BeWo细胞活力和紧密连接因子的表达[J]. 中国组织工程研究, 2021, 25(在线): 4970-4975. |
| [3] | 袁家威, 张海涛, 揭 珂, 曹厚然, 曾意荣. 基于网络药理学研究桃红四物汤治疗假体周围感染的潜在靶点和机制[J]. 中国组织工程研究, 2021, 25(9): 1428-1433. |
| [4] | 张 超, 吕 欣. 髋臼骨折固定后的异位骨化:危险因素、预防及其治疗进展[J]. 中国组织工程研究, 2021, 25(9): 1434-1439. |
| [5] | 周继辉, 李新志, 周 游, 黄 卫, 陈文瑶. 髌骨骨折修复内植物选择的多重问题[J]. 中国组织工程研究, 2021, 25(9): 1440-1445. |
| [6] | 王德斌, 毕郑刚. 尺骨鹰嘴骨折-脱位解剖力学、损伤特点、固定修复及3D技术应用的相关问题[J]. 中国组织工程研究, 2021, 25(9): 1446-1451. |
| [7] | 顾 霞, 赵 敏, 王平义, 李一梅, 李文华. 低氧诱导因子1α与低氧相关疾病信号通路的关系[J]. 中国组织工程研究, 2021, 25(8): 1284-1289. |
| [8] | 姬志祥, 蓝常贡. 尿酸盐转运蛋白在痛风中的多态性和治疗相关性[J]. 中国组织工程研究, 2021, 25(8): 1290-1298. |
| [9] | 袁 美, 张新新, 郭祎莎, 毕 霞. 循环microRNA在血管性认知障碍诊断中的应用[J]. 中国组织工程研究, 2021, 25(8): 1299-1304. |
| [10] | 蒋红英, 朱 亮, 余 曦, 黄 靖, 向小娜, 兰正燕, 何红晨. 富血小板血浆干预脊髓损伤患者压力性损伤的作用[J]. 中国组织工程研究, 2021, 25(8): 1149-1153. |
| [11] | 吴 训, 孟娟红, 张建运, 王 亮. 浓缩生长因子修复兔髁突全层软骨损伤[J]. 中国组织工程研究, 2021, 25(8): 1166-1171. |
| [12] | 申晋波, 张 林. 急性力竭运动导致大鼠跟腱微损伤的超微结构变化及机制[J]. 中国组织工程研究, 2021, 25(8): 1190-1195. |
| [13] | 柴 乐, 吕建兰, 胡劲涛, 胡华辉, 许庆军, 余进伟, 全仁夫. 诱导急性脊髓损伤模型大鼠炎症反应信号通路的变化[J]. 中国组织工程研究, 2021, 25(8): 1218-1223. |
| [14] | 张秀梅, 翟运开, 赵 杰, 赵 萌. 类器官模型国内外数据库近10年文献研究热点分析[J]. 中国组织工程研究, 2021, 25(8): 1249-1255. |
| [15] | 李中峰, 陈明海, 凡一诺, 魏秋实, 何 伟, 陈镇秋. 右归饮治疗激素性股骨头坏死作用机制的网络药理学分析[J]. 中国组织工程研究, 2021, 25(8): 1256-1263. |
近年来,基于细胞水平的治疗为肌腱修复和重建提供了一种新的替代方法,包括肌腱干细胞、骨髓间充质干细胞和脂肪间充质干细胞[5-6]。研究表明,肌腱干细胞具有集落形成、多重分化和自我更新能力等干细胞的基本特点[7],能促进肌腱损伤后更快更好地恢复[5,8],是肌腱修复的一个有希望的细胞来源[9-10]。肌腱干细胞在肌腱修复中的作用可分为3种情况:一是作为组织工程肌腱再生的外源性或者内源性的种子细胞,通过将肌腱干细胞移植到肌腱缺损处,诱导肌腱干细胞的成腱性分化,促进细胞分泌Ⅰ型胶原及相关细胞因子,重塑损伤肌腱的功能;二是在急性肌腱损伤(撕裂)修复中
“扮演角色”,通过加入相应细胞因子激活肌腱中少量的肌腱干细胞,促使其增殖及分化,减少瘢痕愈合,从而促进肌腱的修复;三是在肌腱退化过程中的作用,肌腱干细胞的多重分化特性使细胞可在一定环境下向骨细胞、软骨细胞、脂肪细胞分化,而这3种细胞导致肌腱退化,比如钙化性肌腱炎。
富血小板血浆是指全血通过梯度离心分层后得到的富含血小板的血浆部分,含有多种较丰富的生物活性因子。富血小板血浆一般应用于软骨及骨损伤的治疗,获得良好的治疗效果,因为富血小板血浆易于取得,且含有的生长因子丰富,具有极高的研究价值。另外,富血小板血浆被凝血酶等激活物激活后形成的胶冻状物,是具有完整的三维结构和良好组织相容性的纤维蛋白支架,有望成为肌腱干细胞进行组织移植治疗肌腱修复的载体。富血小板血浆含有多种生长因子,有助于促进组织愈合,这些生长因子包括血小板衍生生长因子、转化生长因子β、血管内皮生长因子、表皮生长因子、胰岛素样生长因子1、成纤维细胞生长因子和肝细胞生长因子等,对肌腱干细胞的增殖、分化具有不同的作用,因此文章就细胞因子和富血小板血浆对肌腱干细胞的作用作一综述。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
progenitor cells,tendon stem cell, cytokine,platelet-rich plasma,
ligament injury”“肌腱干细胞,细胞因子,富血小板血浆,肌腱损伤”为关键词,检索2007至2019年期间的文献,检索文献类型:综述性论文、研究性论文、著作等。
1.2 文献的筛选标准 ①细胞因子和富血小板血浆对肌腱干细胞的影响;②选择近期发表或在权威杂志上发表的文章;③具有时效性,能反映研究领域最新进展的综述。
1.3 资料提取与文献质量评价 排除与文章研究目的无关的文献,排除研究陈旧性、重复性研究文献,共纳入83篇符合标准的文献进行综述。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程

文题释义:#br# 肌腱干细胞:是肌腱组织经消化后提取的具有自我更新及多向分化能力的细胞,不同于正常肌腱细胞,其表达干细胞标记物,近年来多项研究证明肌腱干细胞是组织移植治疗肌腱损伤的优势细胞。#br#
富血小板血浆:是指全血通过梯度离心分层后得到的富含血小板的血浆部分,含有多种较丰富的生物活性因子,其血小板浓度为全血的3倍。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
肌腱干细胞具有分化为肌腱细胞、促进肌腱愈合的天然优势,各因素对肌腱干细胞的影响已相继展开探索,这些因素是未来动物实验的指引,也能够为临床应用提供帮助;同时,支持肌腱干细胞移植的最佳载体正在被寻找,细胞依附性、孔隙率、合适的降解时间、足够的抗拉伸强度都是需要考虑的问题。富血小板血浆中富含各类细胞因子,而各种细胞因子对肌腱干细胞具有协同或者相反的作用,富血小板血浆对于肌腱干细胞的影响仍不清晰,选择更适合的种类与浓度还需要进一步的探索。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||