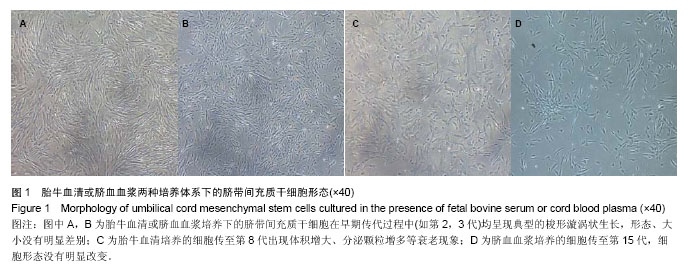
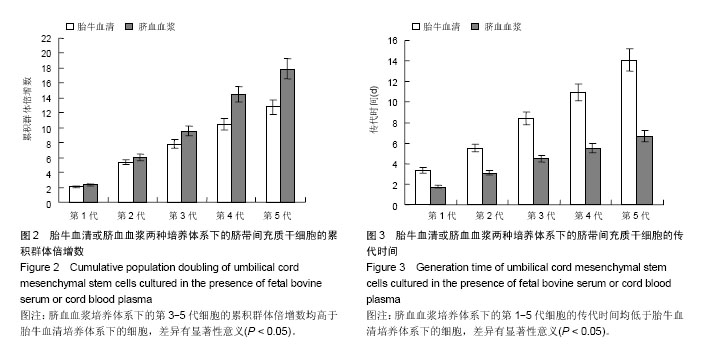
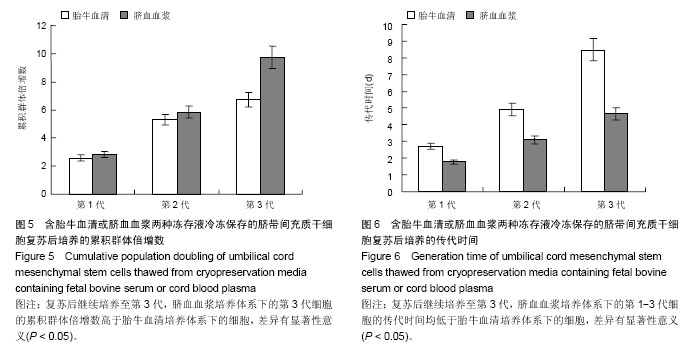
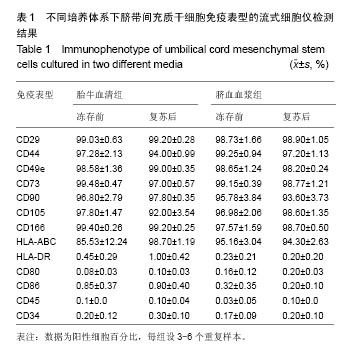
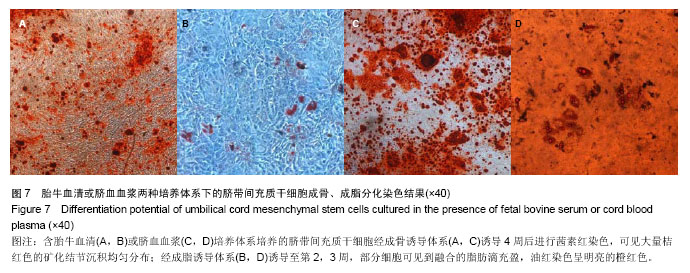
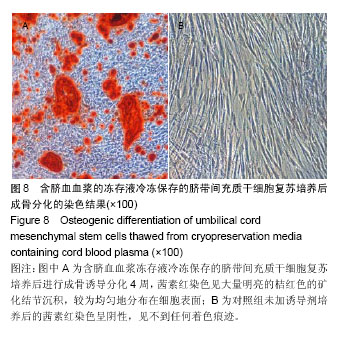
| [1] Sharma RR, Pollock K, Hubel A,et al.Mesenchymal stem or stromal cells: a review of clinical applications and manufacturing practices.Transfusion. 2014;54(5):1418-1437.
[2] Salem HK, Thiemermann C.Mesenchymal stromal cells: current understanding and clinical status.Stem Cells. 2010; 28(3):585-596.
[3] Patel AN, Genovese J.Potential clinical applications of adult human mesenchymal stem cell (Prochymal®) therapy.Stem Cells Cloning. 2011;4:61-72.
[4] Chanda D, Kumar S, Ponnazhagan S.Therapeutic potential of adult bone marrow-derived mesenchymal stem cells in diseases of the skeleton.J Cell Biochem. 2010;111(2):249-257.
[5] Schallmoser K, Rohde E, Reinisch A,et al.Rapid large-scale expansion of functional mesenchymal stem cells from unmanipulated bone marrow without animal serum.Tissue Eng Part C Methods. 2008;14(3):185-196.
[6] Prockop DJ."Stemness" does not explain the repair of many tissues by mesenchymal stem/multipotent stromal cells (MSCs).Clin Pharmacol Ther. 2007;82(3):241-243.
[7] Spees JL, Gregory CA, Singh H,et al.Internalized antigens must be removed to prepare hypoimmunogenic mesenchymal stem cells for cell and gene therapy.Mol Ther. 2004;9(5): 747-756.
[8] Mannello F, Tonti GA.Concise review: no breakthroughs for human mesenchymal and embryonic stem cell culture: conditioned medium, feeder layer, or feeder-free; medium with fetal calf serum, human serum, or enriched plasma; serum-free, serum replacement nonconditioned medium, or ad hoc formula? All that glitters is not gold!Stem Cells. 2007; 5(7):1603-1609.
[9] Tekkatte C, Gunasingh GP, Cherian KM,et al."Humanized" stem cell culture techniques: the animal serum controversy. Stem Cells Int. 2011;2011:504723.
[10] Bieback K, Hecker A, Kocaömer A,et al.Human alternatives to fetal bovine serum for the expansion of mesenchymal stromal cells from bone marrow.Stem Cells. 2009;27(9):2331-2341.
[11] Jung J, Moon N, Ahn JY,et al.Mesenchymal stromal cells expanded in human allogenic cord blood serum display higher self-renewal and enhanced osteogenic potential.Stem Cells Dev. 2009;18(4):559-571.
[12] 吴洁莹,廖灿,许遵鹏,等.广州脐血库造血干细胞保存及临床应用[J].中华生物医学工程杂志,2010,16(5): 438-443.
[13] Stolzing A, Jones E, McGonagle D,et al.Age-related changes in human bone marrow-derived mesenchymal stem cells: consequences for cell therapies.Mech Ageing Dev. 2008; 129(3):163-173.
[14] Jung S, Panchalingam KM, Rosenberg L,et al. Ex vivo expansion of human mesenchymal stem cells in defined serum-free media.Stem Cells Int. 2012;2012:123030.
[15] Dahl JA, Duggal S, Coulston N,et al.Genetic and epigenetic instability of human bone marrow mesenchymal stem cells expanded in autologous serum or fetal bovine serum.Int J Dev Biol. 2008;52(8):1033-1042.
[16] Stute N, Holtz K, Bubenheim M,et al. Autologous serum for isolation and expansion of human mesenchymal stem cells for clinical use.Exp Hematol. 2004;32(12):1212-1225.
[17] Felka T, Schäfer R, De Zwart P,et al.Animal serum-free expansion and differentiation of human mesenchymal stromal cells.Cytotherapy. 2010;12(2):143-153.
[18] Turnovcova K, Ruzickova K, Vanecek V,et al. Properties and growth of human bone marrow mesenchymal stromal cells cultivated in different media.Cytotherapy. 2009;11(7):874-885.
[19] Poloni A, Maurizi G, Rosini V,et al.Selection of CD271(+) cells and human AB serum allows a large expansion of mesenchymal stromal cells from human bone marrow. Cytotherapy. 2009;11(2):153-162.
[20] Tateishi K, Ando W, Higuchi C,et al.Comparison of human serum with fetal bovine serum for expansion and differentiation of human synovial MSC: potential feasibility for clinical applications.Cell Transplant. 2008;17(5):549-557.
[21] Le Blanc K, Samuelsson H, Lönnies L,et al. Generation of immunosuppressive mesenchymal stem cells in allogeneic human serum.Transplantation. 2007;84(8):1055-1059.
[22] Flemming A, Schallmoser K, Strunk D,et al. Immunomodulative efficacy of bone marrow-derived mesenchymal stem cells cultured in human platelet lysate.J Clin Immunol. 2011;31(6):1143-1156.
[23] Cholewa D, Stiehl T, Schellenberg A,et al.Expansion of adipose mesenchymal stromal cells is affected by human platelet lysate and plating density.Cell Transplant. 2011; 20(9):1409-1422.
[24] Trojahn Kølle SF, Oliveri RS, Glovinski PV,et al.Pooled human platelet lysate versus fetal bovine serum-investigating the proliferation rate, chromosome stability and angiogenic potential of human adipose tissue-derived stem cells intended for clinical use.Cytotherapy. 2013;15(9):1086-1097.
[25] Hemeda H, Giebel B, Wagner W.Evaluation of human platelet lysate versus fetal bovine serum for culture of mesenchymal stromal cells.Cytotherapy. 2014;16(2):170-180.
[26] 吕江涛,田少奇,孙康,等.血小板裂解液对人脐带间充质干细胞体外成骨分化的影响[J].中国组织工程研究,2012,16(41): 7637-7641.
[27] Abdelrazik H, Spaggiari GM, Chiossone L,et al.Mesenchymal stem cells expanded in human platelet lysate display a decreased inhibitory capacity on T- and NK-cell proliferation and function.Eur J Immunol. 2011;41(11):3281-3290.
[28] Phadnis SM, Joglekar MV, Venkateshan V,et al.Human umbilical cord blood serum promotes growth, proliferation, as well as differentiation of human bone marrow-derived progenitor cells.In Vitro Cell Dev Biol Anim. 2006;42(10): 283-286.
[29] Shetty P, Bharucha K, Tanavde V.Human umbilical cord blood serum can replace fetal bovine serum in the culture of mesenchymal stem cells.Cell Biol Int. 2007;31(3):293-298.
[30] 旷文勇,周新伏,李睿娟,等.脐带血清和成人自体血清体外培养人骨髓间充质干细胞的比较[J].中国组织工程研究与临床康复, 2009,13(45):8917-8921.
[31] 吴刚,何辉,曹月诚,等.不同血清培养条件下骨髓间充质干细胞的增殖[J].中国组织工程研究与临床康复,2010,14(49):9151- 9154.
[32] Murphy MB, Blashki D, Buchanan RM,et al.Adult and umbilical cord blood-derived platelet-rich plasma for mesenchymal stem cell proliferation, chemotaxis, and cryo-preservation.Biomaterials. 2012;33(21):5308-5316.
[33] 丁妍,卢智勇,袁雅红,等.脐血浆与胎牛血清培养人脐带间充质干细胞的比较[J]. 生物医学工程学杂志,2013,30(6):1279-1282.
[34] Dominici M, Le Blanc K, Mueller I,et al.Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315-317.
[35] Horwitz EM, Le Blanc K, Dominici M,et al.Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement.Cytotherapy. 2005;7(5):393-395.
[36] Hemeda H, Kalz J, Walenda G,et al.Heparin concentration is critical for cell culture with human platelet lysate.Cytotherapy. 2013;15(9):1174-1181.
[37] Jung S, Sen A, Rosenberg L,et al.Identification of growth and attachment factors for the serum-free isolation and expansion of human mesenchymal stromal cells.Cytotherapy. 2010; 12(5): 637-657.
[38] Lindroos B, Boucher S, Chase L,et al.Serum-free, xeno-free culture media maintain the proliferation rate and multipotentiality of adipose stem cells in vitro.Cytotherapy. 2009;11(7):958-972.
[39] Liu CH, Wu ML, Hwang SM. Optimization of serum free medium for cord blood mesenchymal stem cells. Biochem Eng J. 2007; 33: 1-9.
[40] 周婷婷,卫超,陈晓东,等.无血清培养体系原代培养脐带间充质干细胞[J].中国组织工程研究,2013,17(27):4980-4987. |