Melanoma is a highly malignant tumor. The development process is as follows: melanoma can get
in situ proliferation and form tumor foci, then beak through the basement membrane and obtain tissue infiltration, finally transfer to other parts through blood vessels or lymphatic
[27]. During the development process, melanoma can express some protease, promote the degeneration of basement, membrane components, thus promoting the transformation of tumor cells
[23, 28]. Reports have indicated that the melanoma tissue model
in vitro established with melanoma cells and dermal matrix materials can reflect the invasion characteristics of melanoma
[13], and the previous of the group have proved this point
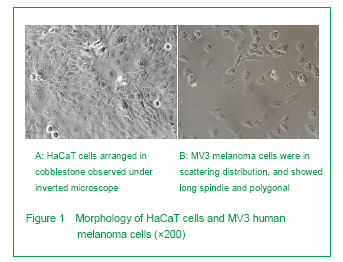
[23, 25]. MV3 cell is a kind of human melanoma cell line, it can be obtained through subcutaneously inoculating the human melanoma into the nude mice
[29]. The cell line had highly tumorigenic and metastatic in the nude mice experiment, is the better cell line for the research of the expressions of human melanoma cell surface molecules and the transformation of melanoma. De-epidermized dermis is the skin tissue without epidermis and living cell components, but maintains the basement membrane. Meanwhile, the dermal matrix surface and adnexal tube wall still maintains the basement membrane components, such as glycogen and collagen
[12]. So the de-epidermized dermis can provide an environment for growth of the melanoma
in vitro, and simulate the process of skin
in situ melanoma cells breaking through the basement membrane into the dermis
[23]. Due to the differences in the seed cells, dermal matrix materials and medium components, there are some differences in the structure and function of the
in vitro established melanoma tissues. The authors have constructed the
in vitro melanoma model with this cell line
[23-24], and made primary research on the invasion mechanism of melanoma
[24-25]. But in the previous studies, the authors commonly used the human keratinocytes and MV3 melanoma cells. Due to the difference of seed cell source, there may be differences in the skin models
[30]. HaCaT cells are immortalized keratinocyte cell lines, though there are differences between HaCaT cells and normal human keratinocytes
[31], the HaCaT cells are similar with the normal human keratinocytes
[18], HaCaT cells can be used for the
in vitro establishment of skin models
[30-31].
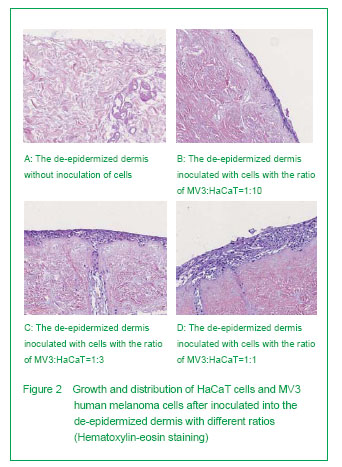
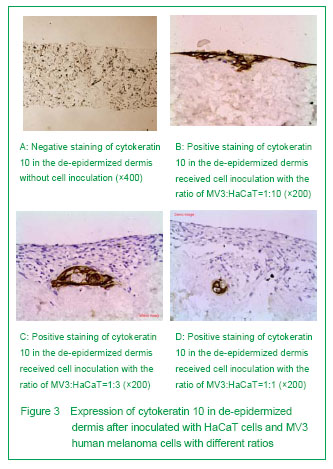
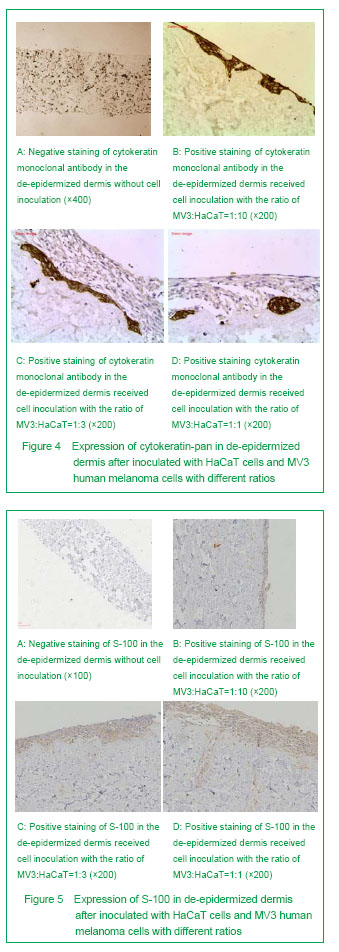
Based on the previous studies, we constructed the artificial skin model of melanoma through inoculating MV3 melanoma cells and HaCaT cells on the de-epidermized dermis. The results showed that in the skin melanoma model constructed by the authors, the surface cells showed a multilayer distribution, the spinous layer cells were arranged in paving, the keratinocytes in partial areas showed a significant atypia and disorder polodial with mild-moderate atypia, and the pathological division phase could be observed, the atypia was most significant in the epidermal cells on the surface, and the structure was similar with that of normal human epidermis. Cytokeratin is a group of protein, some cytokeratin specifically expressed in the keratinocytes, and cytokeratin 10 is one of the specific markers of keratinocytes[32]. The simulation showed the immunostaining of cytokeratin 10 and cytokeratin-pan was positive, and distributed layer-by-layer. The experiment showed that if the proportion of HaCaT cells was high in the tissue model, the expressions of cytokeratin 10 and cytokeratin-pan will more significant, the stratification will more clear. When the concentration of HaCaT cells decreased, the positive cell mass of cytokeratin 10 and cytokeratin-pan staining was in slug distribution. S-100 may be one of the best markers of melanoma[33]. Staining of S-100 in skin melanoma showed the MV3 cells were distributed layer-by-layer, and some of the cells were in tumor-like distribution. Some tumor cells can break through the basement membrane and form new tumor foci in de-epidermized dermis, this is similar with the previous studies[23, 25, 34-35].
Studies have shown that keratinocytes can enhance the invasion vitality of melanoma cells, and this may be related with the secretion of growth factor and proteases by keratinocyte[13, 36-37]. The authors fund that with the increasing of the proportion of MV3 cells, the HaCaT cells were gradually compressed to the middle-lower layer of the model. With the increasing of the ratio of MV3: HaCaT, the invasion vitality of MV3 human melanoma cells was increased gradually. When the ratio of MV3: HaCaT =1:1, the invasion vitality of MV3 cells reached peak. The reason may be that tumor cells with higher proliferation rate can lead to cell integration, and produce mechanical stress when mixed grow with epidermal cells, thus resulting the tumor cells to penetrate the basement membrane[36]. It also may be because the inoculated MV3 cells are too less to form tumor foci, the invasion vitality is relatively small. In this experiment, the ratio of MV3: HaCaT was highest at 1:1, and the invasion vitality of MV3 cells did not observed yet if the proportion of MV3 cells higher than that of HaCaT cells, so the further observation is needed.
Compared with part of the previous experiments, there were no recombinant epidermal growth factor, bovine pituitary leaching solution
[24] and cholera toxin
[38]. Recombinant epidermal growth factor, bovine pituitary leaching solution and endotoxin may affect the growth of HaCaT cells. The medium for the culture of HaCaT cells is 10% Dulbecco’s modified Eagle’s medium without endotoxin content, which can avoid the influence above. The medium used in this experiment is easy to prepare with low cost, and the HaCaT cell medium is same with the MV3 cell medium, which is easy to construct the
in vitro skin models.
.jpg)