中国组织工程研究 ›› 2019, Vol. 23 ›› Issue (12): 1921-1929.doi: 10.3969/j.issn.2095-4344.1130
• 骨与关节综述 bone and joint review • 上一篇 下一篇
激素性股骨头缺血坏死中PI3K/Akt/mTOR信号通路对自噬的调控
李云隆1,赵振群2,刘万林2
- 1内蒙古医科大学研究生学院,内蒙古自治区呼和浩特市 010000;2内蒙古医科大学第二附属医院,内蒙古自治区呼和浩特市 010030
-
出版日期:2019-04-28发布日期:2019-04-28 -
通讯作者:赵振群,主任医师,内蒙古医科大学第二附属医院,内蒙古自治区呼和浩特市 010030 刘万林,教授,内蒙古医科大学第二附属医院,内蒙古自治区呼和浩特市 010030 -
作者简介:李云隆,男,1992年生,内蒙古自治区呼和浩特市人,汉族,内蒙古医科大学在读硕士,主要从事激素性股骨头缺血性坏死的发病机制研究。 -
基金资助:国家自然科学基金项目(81760391),项目名称:PI3K/Akt/mTOR介导的血管内皮细胞自噬在激素性骨坏死中的作用及其mTOR-siRNA对其影响的实验研究,项目负责人:赵振群
Regulation of PI3K/Akt/mTOR signaling pathway on autophagy in steroid-induced avascular necrosis of the femoral head
Li Yunlong1, Zhao Zhenqun2, Liu Wanlin2
- 1Graduate School of Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China; 2the Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China
-
Online:2019-04-28Published:2019-04-28 -
Contact:Zhao Zhenqun, Chief physician, the Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China Liu Wanlin, Professor, the Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China -
About author:Li Yunlong, Master candidate, Graduate School of Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China -
Supported by:the National Natural Science Foundation of China, No. 81760391 (to ZZQ)
摘要:
文章快速阅读:
.jpg)
中图分类号:
引用本文
李云隆,赵振群,刘万林. 激素性股骨头缺血坏死中PI3K/Akt/mTOR信号通路对自噬的调控[J]. 中国组织工程研究, 2019, 23(12): 1921-1929.
Li Yunlong, Zhao Zhenqun, Liu Wanlin. Regulation of PI3K/Akt/mTOR signaling pathway on autophagy in steroid-induced avascular necrosis of the femoral head[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(12): 1921-1929.
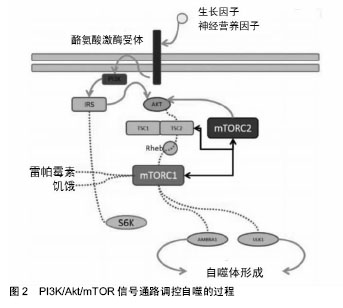
2.2.1 PI3K/Akt/mTOR信号通路组成

| [1] Okazaki S, Nagoya S, Matsumoto H, et al. Development of non-traumatic osteonecrosis of the femoral head requires toll-like receptor 7 and 9 stimulations and is boosted by repression on nuclear factor kappa B in rats. Lab Invest. 2015;95(1): 92-99.[2] Ao W, Ming R, Jincheng W. The pathogenesis of steroid-induced osteonecrosis of the femoral head: A systematic review of the literature. Gene. 2018;671:103-109. [3] Lian JB, Stein GS, Wijnen AJV, et al. MicroRNA control of bone formation and homeostasis. Nat Rev Endocrinol. 2012; 8(4): 212-227. [4] Li M, Gao P, Zhang J. Crosstalk between autophagy and apoptosis: potential and emerging therapeutic targets for cardiac diseases. Int J Mol Sci. 2016;17(3): 332.[5] Xue X, Feng Z, Li Z, et al. Salidroside inhibits steroid-induced avascular necrosis of the femoral head via the PI3K/Akt signaling pathway: In vitro and in vivo studies. Mol Med Rep. 2018; 17(3): 3751-3757.[6] Van Noorden R, Ledford H. Medicine Nobel for research on how cells 'eat themselves'. Nature. 2016;538(7623): 18-19.[7] Galluzzi L, Pietrocola F, Bravo-San Pedro J, et al. Autophagy in malignant transformation and cancer progression. Embo J. 2015; 34(7): 856-880.[8] Rebecca VW, Amaravadi RK. Emerging strategies to effectively target autophagy in cancer. Oncogene. 2016; 35(1): 1-11.[9] Tanida I. Autophagosome formation and molecular mechanism of autophagy. Antioxid Redox Signal. 2011;14(11): 2201-2214.[10] Shibutani S, Yoshimori T. A current perspective of autophagosome biogenesis. Cell Res. 2014;24(1): 58-68.[11] Zhang Z, Singh R, Aschner M. Methods for the Detection of Autophagy in Mammalian Cells. Curr Protoc Toxicol. 2016; 69:20.12.1-20.12.26.[12] Klionsky D, Abdelmohsen K, Abe A, et al. Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy. 2016;12(1): 1-222.[13] Carlsson S, Simonsen A. Membrane dynamics in autophagosome biogenesis. J Cell Sci. 2015;128(2): 193-205.[14] Russell R, Yuan H, Guan K. Autophagy regulation by nutrient signaling. Cell Res. 2014;24(1): 42-57.[15] Koyama-Honda I, Itakura E, Fujiwara TK, et al.Temporal analysis of recruitment of mammalian ATG proteins to the autophagosome formation site. Autophagy. 2013;9(10): 1491-1499.[16] Dall'Armi C1, Devereaux KA, Di Paolo G. The role of lipids in the control of autophagy. Curr. Biol. 2013;23: R33-45.[17] Johansen T, Lamark T. Selective autophagy mediated by autophagic adapter proteins. Autophagy. 2011;7(3): 279-296.[18] Suzuki K, Akioka M, Kondo-Kakuta C, et al. Fine mapping of autophagy-related proteins during autophagosome formation in Saccharomyces cerevisiae. J Cell Sci. 2013;126(Pt 11): 2534-2544.[19] Shen H, Mizushima N. At the end of the autophagic road: an emerging understanding of lysosomal functions in autophagy . Trends Biochem Sci. 2014;39(2): 61-71.[20] David HS, P rez-Rojas JM, Jacqueline HD, et al. The role of PI3K/AKT/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cellular Signalling. 2014;26(12): 2694-2701.[21] Bahrami A, Hassanian S, Shahidsales S, et al. Targeting RAS signaling pathway as a potential therapeutic target in the treatment of colorectal cancer. J Cell Physiol. 2018;233(3): 2058-2066.[22] Dibble C, Cantley L. Regulation of mTORC1 by PI3K signaling. Trends Cell Biol. 2015;25(9): 545-555.[23] Fruman D, Rommel C. PI3K and cancer: lessons, challenges and opportunities. Nat Rev Drug Discov. 2014; 13(2): 140-156.[24] Thorpe L, Yuzugullu H, Zhao J. PI3K in cancer: divergent roles of isoforms, modes of activation and therapeutic targeting . Nat Rev Cancer. 2015; 15(1): 7-24.[25] Duzgun Z, Eroglu Z, Biray Avci C. Role of mTOR in glioblastoma. Gene. 2016;575(2 Pt 1): 187-190.[26] Xia P, Xu X. PI3K/Akt/mTOR signaling pathway in cancer stem cells: from basic research to clinical application. Am J Cancer Res. 2015;5(5): 1602-1609.[27] Vanhaesebroeck B, Stephens L, Hawkins P. PI3K signalling: the path to discovery and understanding. Nat Rev Mol Cell Biol. 2012;13(3): 195-203.[28] Zou H, Li L, Garcia Carcedo I, et al. Synergistic inhibition of colon cancer cell growth with nanoemulsion-loaded paclitaxel and PI3K/mTOR dual inhibitor BEZ235 through apoptosis. Int J Nanomedicine. 2016;11:1947-1958.[29] Vanhaesebroeck B, Guillermet-Guibert J, Graupera M, et al. The emerging mechanisms of isoform-specific PI3K signalling. Nat Rev Mol Cell Biol. 2010;11(5): 329-341.[30] Avan A, Narayan R, Giovannetti E, et al. Role of Akt signaling in resistance to DNA-targeted therapy. World J Clin Oncol. 2016;7(5): 352-369.[31] Gao Y, Yuan C, Yuan W. Will targeting PI3K/Akt/mTOR signaling work in hematopoietic malignancies? Stem Cell Investig. 2016;3:31.[32] Jaber N, Dou Z, Chen J, et al. Class III PI3K Vps34 plays an essential role in autophagy and in heart and liver function . Proc Natl Acad Sci USA. 2012; 109(6): 2003-2008.[33] Yu J, Cui W. Proliferation, survival and metabolism: the role of PI3K/AKT/mTOR signalling in pluripotency and cell fate determination. Development. 2016; 143(17): 3050-3060.[34] Lin H, Lin C, Huo C, et al. AKT3 promotes prostate cancer proliferation cells through regulation of Akt, B-Raf, and TSC1/TSC2. Oncotarget. 2015; 6(29): 27097-27112.[35] Maiese K. Targeting molecules to medicine with mTOR, autophagy and neurodegenerative disorders. Br J Clin Pharmacol. 2016;82(5): 1245-1266.[36] Coffey R, Shi Y, Long M, et al. Ubiquilin-mediated Small Molecule Inhibition of Mammalian Target of Rapamycin Complex 1 (mTORC1) Signaling. J Biol Chem. 2016;291(10): 5221-5233.[37] Yang H, Rudge D, Koos J, et al. mTOR kinase structure, mechanism and regulation. Nature. 2013; 497(7448): 217-223.[38] Liu P, Gan W, Chin Y, et al. PtdIns(3,4,5)P3-Dependent Activation of the mTORC2 Kinase Complex. Cancer Discov. 2015; 5(11): 1194-209.[39] Heras-Sandoval D , Pérez-Rojas, Jazmin M, et al. The role of PI3K/AKT/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cell Signal. 2014;26(12): 2694-2701.[40] Wang K, Yang H, Jiang W, et al. Puquitinib mesylate (XC-302) induces autophagy via inhibiting the PI3K/AKT/mTOR signaling pathway in nasopharyngeal cancer cells. Int J Mol Med. 2015; 36(6): 1556-1562.[41] Bahrami A, Khazaei M, Hasanzadeh M, et al. Therapeutic potential of targeting pi3k/akt pathway in treatment of colorectal cancer: rational and progress. J Cell Biochem. 2018;119(3): 2460-2469.[42] Walker N, Belloli E, Stuckey L, et al. Mechanistic Target of Rapamycin Complex 1 (mTORC1) and mTORC2 as Key Signaling Intermediates in Mesenchymal Cell Activation. J Biol Chem. 2016;291(12): 6262-6271.[43] Hu Z, Yang B, Mo X, et al. Mechanism and Regulation of Autophagy and Its Role in Neuronal Diseases . Mol Neurobiol. 2015; 52(3): 1190-209.[44] Oh W, Jacinto E. mTOR complex 2 signaling and functions. Cell Cycle. 2011;10(14): 2305-2316.[45] Laplante M, Sabatini D. mTOR signaling in growth control and disease. Cell. 2012;149(2): 274-293.[46] Wang Z, Wang Y, Huang Y, et al. bFGF regulates autophagy and ubiquitinated protein accumulation induced by myocardial ischemia/reperfusion via the activation of the PI3K/Akt/mTOR pathway. Sci Rep. 2015; 5:9287.[47] Cao Z, Yang Y, Yu S, et al. Pogostone induces autophagy and apoptosis involving PI3K/Akt/mTOR axis in human colorectal carcinoma HCT116 cells. J Ethnopharmacol. 2017;202:20-27.[48] Sun X, Du F, Liu S. Modulation of autophagy in exJSRV-env- transfected cells through the Akt/mTOR and MAPK signaling pathway . Biochem Biophys Res Commun. 2017;485(3): 672-678.[49] Su R, Jin X, Zhang W, et al. Particulate matter exposure induces the autophagy of macrophages via oxidative stress-mediated PI3K/AKT/mTOR pathway. Chemosphere. 2017;167:444-453.[50] Li Y, Liu Y, Shi F, et al. Knockdown of Rap1b Enhances Apoptosis and Autophagy in Gastric Cancer Cells via the PI3K/Akt/mTOR Pathway. Oncol Res. 2016;24(5): 287-293.[51] Meng Y, Lin Z, Ge N, et al. Ursolic Acid Induces Apoptosis of Prostate Cancer Cells via the PI3K/Akt/mTOR Pathway . Am J Chin Med. 2015;43(7): 1471-1486.[52] Yu L, Mcphee C, Zheng L, et al. Termination of autophagy and reformation of lysosomes regulated by mTOR. Nature. 2010;465(7300): 942-946.[53] Zhao Z, Bai R, Liu W, et al. Roles of oxidative DNA damage of bone marrow hematopoietic cells in steroid-induced avascular necrosis of femoral head. Genet Mol Res. 2016;15(1). doi: 10.4238/gmr.15017706.[54] Bai R, Na Y, Liu W, et al. Quantitative assessment of ABCB1 polymorphisms and non-traumatic osteonecrosis of the femur head risk. Int J Clin Exp Med. 2016;9(11): 21542-21548.[55] 冯卫,刘万林,苏秀兰,等. 激素性股骨头缺血坏死与血管壁中主要促血管生长因子生理活性关系的实验研究[J]. 中华创伤骨科杂志, 2008, 10(10): 960-964.[56] 赵振群,刘万林,龚瑜林,等. 骨髓造血细胞DNA氧化损伤与骨细胞凋亡在早期激素性股骨头坏死中的表现[J]. 中国组织工程研究, 2015, 19(11): 1652-1657.[57] 赵振群,张志峰,刘万林,等. 激素性股骨头坏死过程中低氧诱导因子1α与骨细胞凋亡[J]. 中国组织工程研究, 2015, 19(51): 8201-8207.[58] Jia J, Yao W, Guan M, et al. Glucocorticoid dose determines osteocyte cell fate. FASEB J. 2011;25(10): 3366-3376.[59] Dai W, Wang L, Jin G, et al. Beta-ecdysone protects mouse osteoblasts from glucocorticoid-induced apoptosis in vitro. Planta Med. 2017;83(11): 888-894.[60] Ryu J, Ko J, Kim M, et al. Prednisolone induces apoptosis in corneal epithelial cells through the intrinsic pathway. Sci Rep, 2017; 7(1): 4135.[61] Feng Z, Zheng W, Tang Q, et al. Fludarabine inhibits STAT1-mediated up-regulation of caspase-3 expression in dexamethasone-induced osteoblasts apoptosis and slows the progression of steroid-induced avascular necrosis of the femoral head in rats. Apoptosis. 2017;22(8): 1001-1012.[62] Liu W, Zhao Z, Na Y, et al. Dexamethasone-induced production of reactive oxygen species promotes apoptosis via endoplasmic reticulum stress and autophagy in MC3T3-E1 cells. Int J Mol Med. 2018; 41(4): 2028-2036.[63] Han Y, Zhang L, Xing Y, et al. Autophagy relieves the function inhibition and apoptosis?promoting effects on osteoblast induced by glucocorticoid . Int J Mol Med. 2018; 41(2): 800-808.[64] Yang Y, Chen K, Li B, et al. Estradiol inhibits osteoblast apoptosis via promotion of autophagy through the ER-ERK-mTOR pathway. Apoptosis. 2013; 18(11): 1363-1375.[65] Wang J, Ma X, Feng Y, et al. Magnesium ions promote the biological behaviour of rat calvarial osteoblasts by activating the pi3k/akt signalling pathway. Biol Trace Elem Res. 2017; 179(2): 284-293.[66] Tong P, Xiao L, Ji W, et al. Research on the role of metabolism of fatty substance and osteoclast activity during the development of steroid-induced necrosis of femoral head. Zhongguo Gu Shang. 2009;22(2): 110-113.[67] Chen W, Kong X, Wan R, et al. Effects of huogu I formula (I) on correlated factors of bone regeneration in chickens with steroid-induced necrosis of femoral head. Chin J Integr Med. 2012;18(5): 378-384.[68] Kim CJ, Shin SH, Kim BJ, et al. The effects of kaempferol- inhibited autophagy on osteoclast formation. Int J Mol Sci. 2018;19(1): E125.[69] Liu S, Zhu L, Zhang J, et al. Anti-osteoclastogenic activity of isoliquiritigenin via inhibition of NF-κB-dependent autophagic pathway. Biochem Pharmacol. 2016;106:82-93.[70] Luo P, Gao F, Han J, et al. The role of autophagy in steroid necrosis of the femoral head: a comprehensive research review. Int Orthop. 2018;42(7): 1747-1753.[71] 孟晨阳,刘万林,白锐,等. 激素性股骨头缺血性坏死发病机制中的细胞自噬[J]. 中国组织工程研究, 2017, 21(8): 1280-1287.[72] Hocking L, Whitehouse C, Helfrich M. Autophagy: a new player in skeletal maintenance? J Bone Miner Res. 2012; 27(7): 1439-1447.[73] Manolagas S, Parfitt A. For whom the bell tolls: distress signals from long-lived osteocytes and the pathogenesis of metabolic bone diseases. Bone. 2013; 54(2): 272-278.[74] 王文选,赵振群,刘万林,等. Beclin 1和MAP1-LC3在家兔激素性股骨头缺血坏死中表达的实验研究[J]. 实用骨科杂志, 2017, 23(9): 811-815.[75] Liao Y, Zhang P, Yuan B, et al. Pravastatin protects against avascular necrosis of femoral head via autophagy. Front Physiol. 2018;9:307.[76] Zhu L, Chen J, Zhang J, et al. Parathyroid Hormone (PTH) Induces Autophagy to Protect Osteocyte Cell Survival from Dexamethasone Damage. Med Sci Monit. 2017;23: 4034-4040.[77] Hu J, Cui W, Ding W, et al. Globular Adiponectin Attenuated H2O2-Induced Apoptosis in Rat Chondrocytes by Inducing Autophagy Through the AMPK/ mTOR Pathway. Cell Physiol Biochem. 2017;43(1): 367-382.[78] Zhu Y, Zhou J, Ao R, et al. A-769662 protects osteoblasts from hydrogen dioxide-induced apoptosis through activating of AMP-activated protein kinase (AMPK). Int J Mol Sci. 2014; 15(6): 11190-1203.[79] She C, Zhu LQ, Zhen YF, et al. Activation of AMPK protects against hydrogen peroxide-induced osteoblast apoptosis through autophagy induction and NADPH maintenance: New implications for osteonecrosis treatment? Cell Signal. 2014; 26(1): 1-8.[80] Kim JH, Kang HM, Yu SB, et al. Cytoprotective Effect of Flavonoid-Induced Autophagy on Bisphosphonate Mediated Cell Death in Osteoblast. J Cell Biochem. 2018;119: 5571-5580.[81] Liu W, Mao L, Ji F, et al. Targeted activation of AMPK by GSK621 ameliorates H2O2-induced damages in osteoblasts. Oncotarget. 2017;8: 10543-10552. [82] Li X, Li Y, Li L, et al. Overactivated autophagy contributes to steroid-induced avascular necrosis of the femoral head. Exp Ther Med. 2017;14(1): 367-372.[83] Yu X, Long Y, Shen H. Differential regulatory functions of three classes of phosphatidylinositol and phosphoinositide 3-kinases in autophagy. Autophagy. 2015;11(10): 1711-1128. |
| [1] | 耿秋东, 葛海雅, 王和鸣, 李 楠. 基于网络药理学探讨龟鹿二仙胶治疗骨关节炎的作用及机制[J]. 中国组织工程研究, 2021, 25(8): 1229-1236. |
| [2] | 李中峰, 陈明海, 凡一诺, 魏秋实, 何 伟, 陈镇秋. 右归饮治疗激素性股骨头坏死作用机制的网络药理学分析[J]. 中国组织工程研究, 2021, 25(8): 1256-1263. |
| [3] | 刘立华, 孙 伟, 王云亭, 高福强, 程立明, 李子荣, 王江宁. 头颈部开窗减压治疗L1型激素性股骨头坏死:单中心前瞻性临床研究[J]. 中国组织工程研究, 2021, 25(6): 906-911. |
| [4] | 刘 钊, 徐西林, 申意伟, 张晓峰, 吕 航, 赵 军, 王政春, 刘旭卓, 王海涛. 塌陷预测方法联合分期分型对股骨头坏死治疗的指导作用与前景[J]. 中国组织工程研究, 2021, 25(6): 929-934. |
| [5] | 李时斌, 赖 渝, 周 毅, 廖建钊, 章晓云, 张 璇. 激素性股骨头坏死发病机制及相关信号通路的靶点效应[J]. 中国组织工程研究, 2021, 25(6): 935-941. |
| [6] | 郑小龙, 何晓铭, 龚水帝, 庞凤祥, 杨 帆, 何 伟, 刘少军, 魏秋实. 酒精性股骨头坏死患者的骨转换特点[J]. 中国组织工程研究, 2021, 25(5): 657-661. |
| [7] | 马泽涛, 曾 晖, 王德利, 翁 鉴, 冯 松. 微小RNA-138-5p与软骨细胞增殖和自噬的关系[J]. 中国组织工程研究, 2021, 25(5): 674-678. |
| [8] | 谢 阳, 张淑江, 刘梦兰, 罗 映, 杨 洋, 李作孝. 雷帕霉素保护实验性自身免疫性脑脊髓炎小鼠脊髓神经元的作用途径[J]. 中国组织工程研究, 2021, 25(5): 695-700. |
| [9] | 徐银琴, 史红美, 王光义. 通痹方热敷联合针刺治疗对退变椎间盘细胞凋亡相关基因Caspase-3、Bcl-2 mRNA的影响[J]. 中国组织工程研究, 2021, 25(5): 713-718. |
| [10] | 张雯雯, 金颂峰, 赵国梁, 宮丽鸿. 复方中药稳斑汤减少同型半胱氨酸诱导大鼠心肌微血管内皮细胞凋亡的作用机制[J]. 中国组织工程研究, 2021, 25(5): 723-728. |
| [11] | 刘 青, 万碧江. 针刀治疗胶原诱导性关节炎大鼠滑膜组织Bcl-2/Bax的表达[J]. 中国组织工程研究, 2021, 25(5): 729-734. |
| [12] | 曾祥洪, 梁博伟. 股骨头坏死保髋治疗的新策略[J]. 中国组织工程研究, 2021, 25(3): 431-437. |
| [13] | 左振魁, 韩佳瑞, 计树灵, 贺璐璐. 银杏酮酯预处理减轻辐射所致模型小鼠的急性肠损伤[J]. 中国组织工程研究, 2021, 25(23): 3666-3671. |
| [14] | 张 亮, 马晓燕, 王佳虹. 肾衰饮调控慢性肾功能衰竭大鼠肾脏细胞凋亡的机制[J]. 中国组织工程研究, 2021, 25(23): 3672-3677. |
| [15] | 童 洁, 廖 瑛, 陈政宇, 孙光华. 骨关节炎软骨细胞自噬及丝裂原活化蛋白激酶信号通路的调控[J]. 中国组织工程研究, 2021, 25(20): 3246-3251. |
中国组织工程研究杂志出版内容重点:人工关节;骨植入物;脊柱;骨折;内固定;数字化骨科;组织工程
1.1 资料来源 由第一作者应用计算机检索 PubMed 数据库、Web of science数据库、万方数据库中2008至2018年出版的文献,检索词分别为“Steroid,necrosis of the femoral head,autophagy,signal pathway,糖皮质激素,股骨头坏死,自噬”。
中国组织工程研究杂志出版内容重点:人工关节;骨植入物;脊柱;骨折;内固定;数字化骨科;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
.jpg)
.jpg)