中国组织工程研究 ›› 2018, Vol. 22 ›› Issue (5): 807-814.doi: 10.3969/j.issn.2095-4344.0455
• 干细胞综述 stem cell review • 上一篇 下一篇
外环境因素对体细胞重编程诱导多能干细胞的影响
陈忠尧,曹泽宇,黄 艳,汲 婧,陈晓芳
- 北京航空航天大学生物与医学工程学院生物力学与力生物学教育部重点实验室,北京市 100191
-
修回日期:2017-09-14出版日期:2018-02-18发布日期:2018-02-18 -
通讯作者:陈晓芳,博士,副教授,硕士生导师,北京航空航天大学生物与医学工程学院生物力学与力生物学教育部重点实验室,北京市 100191 -
作者简介:陈忠尧,男,1991年生,河北省唐山市人,汉族,北京航空航天大学生物与医学工程学院在读硕士,主要从事再生医学和用于细胞表观遗传检测的芯片实验室研究。 -
基金资助:国家自然科学基金项目(81301334)
Microenvironmental cues influence the reprogramming of somatic cells to induced pluripotent stem cells
Chen Zhong-yao, Cao Ze-yu, Huang Yan, Ji Jing, Chen Xiao-fang
- Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100191, China
-
Revised:2017-09-14Online:2018-02-18Published:2018-02-18 -
Contact:Chen Xiao-fang, Ph.D., Associate professor, Master’s supervisor, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100191, China -
About author:Chen Zhong-yao, Master candidate, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100191, China -
Supported by:the National Natural Science Foundation of China, No. 81301334
摘要:
文章快速阅读:
.jpg)
文题释义: 外环境:是细胞膜以外的溶液和固体环境,以及细胞受到的物理刺激。在体外培养条件下外环境包括培养基内的可溶性因子,基质材料的理化性质,以及对细胞施加的力、电、磁等刺激因素。 细胞重编程:是利用特定转录因子把终末分化的体细胞转变为具有全能性,类似于胚胎干细胞的诱导多能干细胞的过程。许多外环境因素包括可溶性小分子,生长因子,基质材料的硬度,表面形貌,黏附分子,细胞间连接以及力、电、磁场等多种物理刺激对此过程具有重要影响。
中图分类号:
引用本文
陈忠尧,曹泽宇,黄 艳,汲 婧,陈晓芳. 外环境因素对体细胞重编程诱导多能干细胞的影响[J]. 中国组织工程研究, 2018, 22(5): 807-814.
Chen Zhong-yao, Cao Ze-yu, Huang Yan, Ji Jing, Chen Xiao-fang. Microenvironmental cues influence the reprogramming of somatic cells to induced pluripotent stem cells[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(5): 807-814.
Jeon等在polydimethylsiloxane(PDMS)材料上加工出了粗糙度为8的表面并在其上培养小鼠胚胎干细胞。结果表明,相比于粗糙度为1的光滑PDMS材料,粗糙度为8的PDMS材料促进了小鼠胚胎干细胞的自我更新,小鼠胚胎干细胞能够在PDMS材料表面长期维持多能性,而在普通培养皿表面经过长期传代后出现随机分化。粗糙的PDMS材料表面有利于小鼠胚胎干细胞的干性维持,不利于细胞贴壁和分化[9]。相反的,Chen等[10]利用腐蚀的方法增加玻璃表面粗糙度,他们发现光滑的玻璃表面(粗糙度为1)更有利于人胚胎干细胞贴壁、增殖和多能性的长期维持,而粗糙的玻璃表面(粗糙度为100)会引起人胚胎干细胞分化。2篇报道中结果的差异可能与所用材料本身的差异有关,另外,小鼠胚胎干细胞与人胚胎干细胞不同,其维持干性所需条件也有很大差别。Kong等[11]在聚苯乙烯材料上加工了不同形状的纳米柱,结果表明在没有碱性成纤维细胞生长因子的情况下,培养在具有纳米结构的基底上的人胚胎干细胞能够更好的维持干性。Bae等[12]在聚苯乙烯材料上加工了不同直径的纳米柱(100-350 nm),他们发现相比于光滑表面,具有纳米柱的表面不利于细胞形成稳定的黏着斑,限制了细胞迁移,增强了钙黏附蛋白E(E-cadherin)介导的细胞间连接,促进了多能性的维持。此外,微米和纳米级的纤维结构及合成的不规则颗粒结构都会影响胚胎干细胞的多能性[13-16]。
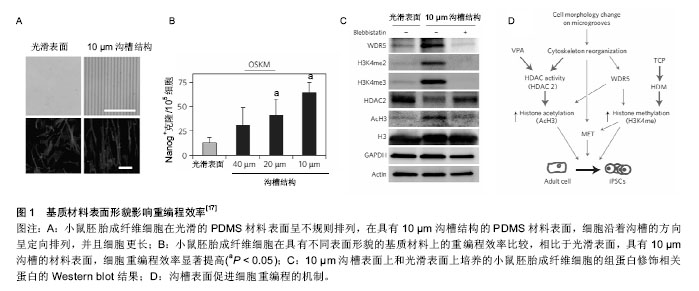
表面微纳结构也能够促进成纤维细胞向其他细胞类型的转变。Leong小组发现表面微沟槽能够促进转录因子Ascl1、Brn2和Myt1L诱导的成纤维细胞向神经细胞重编程[21]。表面结构对神经轴突的分支数也有显著影响,在微沟槽表面轴突分支减少,而在微柱表面分支增加。轴突分支数目的变化与成纤维细胞对外界力学环境的感受有关,非肌肌球蛋白Ⅱ在其中发挥重要作用[21]。Kim等发现具有纳米沟槽的聚氨酯丙烯酸酯表面能够有效提高转录因子Ascl1、Pitx3、Nurr1和Lmx1a介导的成纤维细胞向多巴胺神经元重编程。其分子机制是纳米沟槽引起的细胞骨架重构促进了细胞的间质-上皮转化和组蛋白H3K4me3修饰[22]。此外,微沟槽表面促进成纤维细胞向心肌细胞方向重编程也有报道,原理是微沟槽促进组蛋白乙酰化(AcH3)修饰[23]。这些研究说明了表面微结构引起的表观遗传变化有助于细胞克服重编程过程中的阻碍因素[20],进而提高重编程的效率。

| [1] Yamanaka S, Takahashi K. Induction of pluripotent stem cells from mouse fibroblast cultures. Tanpakushitsu Kakusan Koso. 2006;51(15):2346-2351.[2] Takahashi K, Yamanaka S. A decade of transcription factor-mediated reprogramming to pluripotency. Nat Rev Mol Cell Biol. 2016;17(3):183-193.[3] Theunissen TW, Jaenisch R. Molecular control of induced pluripotency. Cell Stem Cell. 2014;14(6):720-734.[4] Lin J, Li MR, Ti DD, et al. Microenvironment-evoked cell lineage conversion: Shifting the focus from internal reprogramming to external forcing. Ageing Res Rev. 2013; 12(1):29-38.[5] Dingal PC, Discher DE. Combining insoluble and soluble factors to steer stem cell fate. Nat Mater. 2014;13(6):532-537.[6] Crowder SW, Leonardo V, Whittaker T, et al. Material Cues as Potent Regulators of Epigenetics and Stem Cell Function. Cell Stem Cell. 2016;18(1):39-52.[7] Mashinchian O, Turner LA, Dalby MJ, et al. Regulation of stem cell fate by nanomaterial substrates. Nanomedicine (Lond). 2015;10(5):829-847.[8] Bettinger CJ, Langer R, Borenstein JT. Engineering substrate topography at the micro-and nanoscale to control cell function. Angew Chem Int Ed Engl. 2009;48(30):5406-5415.[9] Jeon K, Oh HJ, Lim H, et al. Self-renewal of embryonic stem cells through culture on nanopattern polydimethylsiloxane substrate. Biomaterials. 2012;33(21):5206-5220.[10] Chen W, Villa-Diaz LG, Sun Y, et al. Nanotopography influences adhesion, spreading, and self-renewal of human embryonic stem cells. ACS Nano. 2012;6(5):4094-4103.[11] Kong YP, Tu CH, Donovan PJ, et al. Expression of Oct4 in human embryonic stem cells is dependent on nanotopographical configuration. Acta Biomater. 2013; 9(5):6369-6380.[12] Bae D, Moon SH, Park BG, et al. Nanotopographical control for maintaining undifferentiated human embryonic stem cell colonies in feeder free conditions. Biomaterials. 2014;35(3): 916-928.[13] Nur-E-Kamal A, Ahmed I, Kamal J, et al. Three-dimensional nanofibrillar surfaces promote self-renewal in mouse embryonic stem cells. Stem Cells. 2006;24(2):426-433.[14] Carlson AL, Florek CA, Kim JJ, et al. Microfibrous substrate geometry as a critical trigger for organization, self-renewal, and differentiation of human embryonic stem cells within synthetic 3-dimensional microenvironments. FASEB J. 2012;26(8):3240-3251.[15] Lyu Z, Wang H, Wang Y, et al. Maintaining the pluripotency of mouse embryonic stem cells on gold nanoparticle layers with nanoscale but not microscale surface roughness. Nanoscale. 2014;6(12):6959-6969.[16] Jaggy M, Zhang P, Greiner AM, et al. Hierarchical Micro-Nano Surface Topography Promotes Long-Term Maintenance of Undifferentiated Mouse Embryonic Stem Cells. Nano Lett. 2015;15(10):7146-7154.[17] Downing TL, Soto J, Morez C, et al. Biophysical regulation of epigenetic state and cell reprogramming. Nat Mater. 2013; 12(12): 1154-1162.[18] Sakurai K, Talukdar I, Patil VS, et al. Kinome-wide functional analysis highlights the role of cytoskeletal remodeling in somatic cell reprogramming. Cell Stem Cell. 2014;14(4):523-534.[19] Li Y, Chu JS, Kurpinski K, et al. Biophysical regulation of histone acetylation in mesenchymal stem cells. Biophys J. 2011;100(8): 1902-1909.[20] Pasque V, Jullien J, Miyamoto K, et al. Epigenetic factors influencing resistance to nuclear reprogramming. Trends Genet. 2011;27(12):516-525.[21] Kulangara K, Adler AF, Wang H, et al. The effect of substrate topography on direct reprogramming of fibroblasts to induced neurons. Biomaterials. 2014;35(20):5327-5336.[22] Yoo J, Noh M, Kim H, et al. Nanogrooved substrate promotes direct lineage reprogramming of fibroblasts to functional induced dopaminergic neurons. Biomaterials. 2015;45:36-45.[23] Morez C, Noseda M, Paiva MA, et al. Enhanced efficiency of genetic programming toward cardiomyocyte creation through topographical cues. Biomaterials. 2015;70:94-104.[24] Chowdhury F, Li Y, Poh YC, et al. Soft substrates promote homogeneous self-renewal of embryonic stem cells via downregulating cell-matrix tractions. PLoS One. 2010;5(12): e15655.[25] Ranga A, Gobaa S, Okawa Y, et al. 3D niche microarrays for systems-level analyses of cell fate. Nat Commun. 2014;5:4324.[26] Keung AJ, Asuri P, Kumar S, et al. Soft microenvironments promote the early neurogenic differentiation but not self-renewal of human pluripotent stem cells. Integr Biol (Camb). 2012;4(9): 1049-1058.[27] Musah S, Morin SA, Wrighton PJ, et al. Glycosaminoglycan- binding hydrogels enable mechanical control of human pluripotent stem cell self-renewal. ACS Nano. 2012;6(11): 10168-10177.[28] Higuchi A, Kao SH, Ling QD, et al. Long-term xeno-free culture of human pluripotent stem cells on hydrogels with optimal elasticity. Sci Rep. 2015;5:18136.[29] Koestenbauer S, Zech NH, Juch H, et al. Embryonic stem cells: similarities and differences between human and murine embryonic stem cells. Am J Reprod Immunol 2006;55(3): 169-180.[30] Weinberger L, Ayyash M, Novershtern N, et al. Dynamic stem cell states: naive to primed pluripotency in rodents and humans. Nat Rev Mol Cell Biol. 2016;17(3):155-169.[31] Higuchi S, Watanabe TM, Kawauchi K, et al. Culturing of mouse and human cells on soft substrates promote the expression of stem cell markers. J Biosci Bioeng. 2014; 117(6):749-755.[32] Guo J, Wang Y, Sachs F, et al. Actin stress in cell reprogramming. Proc Natl Acad Sci U S A. 2014;111(49): E5252-5261.[33] Choi B, Park KS, Kim JH, et al. Stiffness of Hydrogels Regulates Cellular Reprogramming Efficiency Through Mesenchymal-to-Epithelial Transition and Stemness Markers. Macromol Biosci. 2016;16(2):199-206.[34] Zonca MR Jr, Yune PS, Heldt CL, et al. High-throughput screening of substrate chemistry for embryonic stem cell attachment, expansion, and maintaining pluripotency. Macromol Biosci. 2013;13(2):177-190.[35] Anderson DG, Levenberg S, Langer R. Nanoliter-scale synthesis of arrayed biomaterials and application to human embryonic stem cells. Nat Biotechnol. 2004;22(7):863-866.[36] Mei Y, Saha K, Bogatyrev SR, et al. Combinatorial development of biomaterials for clonal growth of human pluripotent stem cells. Nat Mater. 2010;9(9):768-778.[37] Brafman DA, Shah KD, Fellner T, et al. Defining long-term maintenance conditions of human embryonic stem cells with arrayed cellular microenvironment technology. Stem Cells Dev. 2009;18(8):1141-1154.[38] Joddar B, Ito Y. Artificial niche substrates for embryonic and induced pluripotent stem cell cultures. J Biotechnol. 2013; 168(2):218-228.[39] Villa-Diaz LG, Kim JK, Lahann J, et al. Derivation and long-term culture of transgene-free human induced pluripotent stem cells on synthetic substrates. Stem Cells Transl Med. 2014;3(12):1410-1417.[40] Zhou P, Wu F, Zhou T, et al. Simple and versatile synthetic polydopamine-based surface supports reprogramming of human somatic cells and long-term self-renewal of human pluripotent stem cells under defined conditions. Biomaterials. 2016;87:1-17.[41] Chen G, Gulbranson DR, Hou Z, et al. Chemically defined conditions for human iPSC derivation and culture. Nat Methods. 2011;8(5):424-429.[42] Kim HT, Lee KI, Kim DW, et al. An ECM-based culture system for the generation and maintenance of xeno-free human iPS cells. Biomaterials. 2013;34(4):1041-1050.[43] Nakagawa M, Taniguchi Y, Senda S, et al. A novel efficient feeder-free culture system for the derivation of human induced pluripotent stem cells. Sci Rep. 2014;4:3594.[44] Lu HF, Chai C, Lim TC, et al. A defined xeno-free and feeder-free culture system for the derivation, expansion and direct differentiation of transgene-free patient-specific induced pluripotent stem cells. Biomaterials. 2014;35(9):2816-2826.[45] Yamasaki S, Taguchi Y, Shimamoto A, et al. Generation of human induced pluripotent stem (Ips) cells in serum- and feeder-free defined culture and TGF-Β1 regulation of pluripotency. PLoS One. 2014;9(1):e87151.[46] Yoo J, Kim J, Baek S, et al. Cell reprogramming into the pluripotent state using graphene based substrates. Biomaterials. 2014;35(29):8321-8329.[47] Chang BS, Choi YJ, Kim JH. Collagen complexes increase the efficiency of iPS cells generated using fibroblasts from adult mice. J Reprod Dev. 2015;61(2):145-153.[48] Smith AW, Hoyne JD, Nguyen PK, et al. Direct reprogramming of mouse fibroblasts to cardiomyocyte-like cells using Yamanaka factors on engineered poly(ethylene glycol) (PEG) hydrogels. Biomaterials. 2013;34(28):6559-6571.[49] Phadnis SM, Loewke NO, Dimov IK, et al. Dynamic and social behaviors of human pluripotent stem cells. Sci Rep. 2015;5: 14209.[50] Lecuit T, Lenne PF. Cell surface mechanics and the control of cell shape, tissue patterns and morphogenesis. Nat Rev Mol Cell Biol. 2007;8(8):633-644.[51] Watanabe K, Ueno M, Kamiya D, et al. A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nat Biotechnol. 2007;25(6):681-686.[52] Chen G, Hou Z, Gulbranson DR, et al. Actin-myosin contractility is responsible for the reduced viability of dissociated human embryonic stem cells. Cell Stem Cell. 2010;7(2):240-248.[53] Li L, Wang BH, Wang S, et al. Individual cell movement, asymmetric colony expansion, rho-associated kinase, and E-cadherin impact the clonogenicity of human embryonic stem cells. Biophys J. 2010;98(11):2442-2451.[54] Rodin S, Antonsson L, Niaudet C, et al. Clonal culturing of human embryonic stem cells on laminin-521/E-cadherin matrix in defined and xeno-free environment. Nat Commun. 2014;5:3195.[55] Pieters T, van Roy F. Role of cell-cell adhesion complexes in embryonic stem cell biology. J Cell Sci. 2014;127(Pt 12): 2603-2613.[56] Samavarchi-Tehrani P, Golipour A, David L, et al. Functional genomics reveals a BMP-driven mesenchymal-to-epithelial transition in the initiation of somatic cell reprogramming. Cell Stem Cell. 2010;7(1):64-77.[57] Li R, Liang J, Ni S, et al. A mesenchymal-to-epithelial transition initiates and is required for the nuclear reprogramming of mouse fibroblasts. Cell Stem Cell. 2010;7(1):51-63.[58] Hu X, Zhang L, Mao SQ, et al. Tet and TDG mediate DNA demethylation essential for mesenchymal-to-epithelial transition in somatic cell reprogramming. Cell Stem Cell. 2014;14(4):512-522.[59] Shu X, Pei D. The function and regulation of mesenchymal-to-epithelial transition in somatic cell reprogramming. Curr Opin Genet Dev. 2014;28:32-37.[60] Megyola CM, Gao Y, Teixeira AM, et al. Dynamic migration and cell-cell interactions of early reprogramming revealed by high-resolution time-lapse imaging. Stem Cells. 2013;31(5): 895-905.[61] Chen T, Yuan D, Wei B, et al. E-cadherin-mediated cell-cell contact is critical for induced pluripotent stem cell generation. Stem Cells. 2010;28(8):1315-1325.[62] Redmer T, Diecke S, Grigoryan T, et al. E-cadherin is crucial for embryonic stem cell pluripotency and can replace OCT4 during somatic cell reprogramming. EMBO Rep. 2011;12(7): 720-726.[63] Bedzhov I, Alotaibi H, Basilicata MF, et al. Adhesion, but not a specific cadherin code, is indispensable for ES cell and induced pluripotency. Stem Cell Res. 2013;11(3):1250-1263.[64] Su G, Zhao Y, Wei J, et al. The effect of forced growth of cells into 3D spheres using low attachment surfaces on the acquisition of stemness properties. Biomaterials. 2013;34(13): 3215-3222.[65] Liu Y, Mukhopadhyay P, Pisano MM, et al. Repression of Zeb1 and hypoxia cause sequential mesenchymal-to- epithelial transition and induction of aid, Oct4, and Dnmt1, leading to immortalization and multipotential reprogramming of fibroblasts in spheres. Stem Cells. 2013;31(7):1350-1362.[66] Su G, Zhao Y, Wei J, et al. Direct conversion of fibroblasts into neural progenitor-like cells by forced growth into 3D spheres on low attachment surfaces. Biomaterials. 2013;34(24): 5897-5906.[67] Mirakhori F, Zeynali B, Rassouli H, et al. Induction of Neural Progenitor-Like Cells from Human Fibroblasts via a Genetic Material-Free Approach. PLoS One. 2015;10(8):e0135479.[68] Ni A, Wu MJ, Chavala SH. Sphere formation permits Oct4 reprogramming of ciliary body epithelial cells into induced pluripotent stem cells. Stem Cells Dev. 2014;23(24): 3065-3071.[69] Ezashi T, Das P, Roberts RM. Low O2 tensions and the prevention of differentiation of hES cells. Proc Natl Acad Sci U S A. 2005;102(13):4783-4788.[70] Hammoud AA, Kirstein N, Mournetas V, et al. Murine Embryonic Stem Cell Plasticity Is Regulated through Klf5 and Maintained by Metalloproteinase MMP1 and Hypoxia. PLoS One. 2016;11(1):e0146281.[71] Hawkins KE, Sharp TV, McKay TR. The role of hypoxia in stem cell potency and differentiation. Regen Med. 2013; 8(6): 771-782.[72] Yoshida Y, Takahashi K, Okita K, et al. Hypoxia enhances the generation of induced pluripotent stem cells. Cell Stem Cell. 2009;5(3):237-241.[73] Mathieu J, Zhang Z, Nelson A, et al. Hypoxia induces re-entry of committed cells into pluripotency. Stem Cells. 2013;31(9): 1737-1748.[74] Cheng L, Hu W, Qiu B, et al. Generation of neural progenitor cells by chemical cocktails and hypoxia. Cell Res. 2014;24(6): 665-679.[75] Wang Y, Shi S, Liu H, et al. Hypoxia Enhances Direct Reprogramming of Mouse Fibroblasts to Cardiomyocyte-Like Cells. Cell Reprogram. 2016;18(1):1-7.[76] Sia J, Sun R, Chu J, et al. Dynamic culture improves cell reprogramming efficiency. Biomaterials. 2016;92:36-45.[77] Luni C, Giulitti S, Serena E, et al. High-efficiency cellular reprogramming with microfluidics. Nat Methods. 2016;13(5): 446-452.[78] Baek S, Quan X, Kim S, et al. Electromagnetic fields mediate efficient cell reprogramming into a pluripotent state. ACS Nano. 2014;8(10):10125-10138.[79] Jin Y, Seo J, Lee JS, et al. Triboelectric Nanogenerator Accelerates Highly Efficient Nonviral Direct Conversion and In Vivo Reprogramming of Fibroblasts to Functional Neuronal Cells. Adv Mater. 2016;28(34):7365-7374.[80] Tajik A, Zhang Y, Wei F, et al. Transcription upregulation via force-induced direct stretching of chromatin. Nat Mater. 2016; 15(12):1287-1296. |
| [1] | 林清凡, 解一新, 陈婉清, 叶振忠, 陈幼芳. 人胎盘源间充质干细胞条件培养液可上调缺氧状态下BeWo细胞活力和紧密连接因子的表达[J]. 中国组织工程研究, 2021, 25(在线): 4970-4975. |
| [2] | 蒲 锐, 陈子扬, 袁凌燕. 不同细胞来源外泌体保护心脏的特点与效应[J]. 中国组织工程研究, 2021, 25(在线): 1-. |
| [3] | 张秀梅, 翟运开, 赵 杰, 赵 萌. 类器官模型国内外数据库近10年文献研究热点分析[J]. 中国组织工程研究, 2021, 25(8): 1249-1255. |
| [4] | 顾 霞, 赵 敏, 王平义, 李一梅, 李文华. 低氧诱导因子1α与低氧相关疾病信号通路的关系[J]. 中国组织工程研究, 2021, 25(8): 1284-1289. |
| [5] | 刘 聪, 刘 肃. miR-17-5p调控低氧诱导因子1α介导脂肪细胞分化及血管生成的分子机制[J]. 中国组织工程研究, 2021, 25(7): 1069-1074. |
| [6] | 王正东, 黄 娜, 陈婧娴, 郑作兵, 胡鑫宇, 李 梅, 苏 晓, 苏学森, 颜 南. 丁酸钠抑制氟中毒可诱导小胶质细胞活化及炎症因子表达增多[J]. 中国组织工程研究, 2021, 25(7): 1075-1080. |
| [7] | 汪显耀, 关亚琳, 刘忠山. 提高间充质干细胞治疗难愈性创面的策略[J]. 中国组织工程研究, 2021, 25(7): 1081-1087. |
| [8] | 万 然, 史 旭, 刘京松, 王岩松. 间充质干细胞分泌组治疗脊髓损伤的研究进展[J]. 中国组织工程研究, 2021, 25(7): 1088-1095. |
| [9] | 廖成成, 安家兴, 谭张雪, 王 倩, 刘建国. 口腔鳞状细胞癌干细胞的治疗靶点及应用前景[J]. 中国组织工程研究, 2021, 25(7): 1096-1103. |
| [10] | 赵 敏, 冯柳祥, 陈 垚, 顾 霞, 王平义, 李一梅, 李文华. 低氧环境下外泌体可作为疾病的标志物[J]. 中国组织工程研究, 2021, 25(7): 1104-1108. |
| [11] | 谢文佳, 夏天娇, 周卿云, 刘羽佳, 顾小萍. 小胶质细胞介导神经元损伤在神经退行性疾病中的作用[J]. 中国组织工程研究, 2021, 25(7): 1109-1115. |
| [12] | 李珊珊, 郭笑霄, 尤 冉, 杨秀芬, 赵 露, 陈 曦, 王艳玲. 感光细胞替代治疗视网膜变性疾病[J]. 中国组织工程研究, 2021, 25(7): 1116-1121. |
| [13] | 焦 慧, 张一宁, 宋雨晴, 林 宇, 王秀丽. 乳腺癌类器官研究进展及临床应用前景[J]. 中国组织工程研究, 2021, 25(7): 1122-1128. |
| [14] | 王诗琦, 张金生. 中医药调控缺血缺氧微环境对骨髓间充质干细胞增殖、分化及衰老的影响[J]. 中国组织工程研究, 2021, 25(7): 1129-1134. |
| [15] | 曾燕华, 郝延磊. 许旺细胞体外培养及纯化的系统性综述[J]. 中国组织工程研究, 2021, 25(7): 1135-1141. |
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.3 质量评估 对初步检索得到的文献进行逐篇阅读,选择与主题相关、数据详实可靠、引用次数较多的文献进行综述,对于部分重点文献还对其引用的参考文献和他引进行了检索和阅读。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
.jpg)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||