中国组织工程研究 ›› 2024, Vol. 28 ›› Issue (19): 3104-3110.doi: 10.12307/2024.144
• 干细胞综述 stem cell review • 上一篇 下一篇
外泌体与皮肤创伤的修复
肖梓腾1,王婷禹2,张雯雯2,谭凤怡1,苏海威1,李思婷1,吴雅慧1,周艳芳2,彭新生3
- 广东医科大学,1第二临床医学院,2基础医学院,3药学院,广东省东莞市 523808
-
收稿日期:2023-03-09接受日期:2023-04-26出版日期:2024-07-08发布日期:2023-09-26 -
通讯作者:周艳芳,博士,教授,广东医科大学基础医学院,广东省东莞市 523808 彭新生,博士,教授,广东医科大学药学院,广东省东莞市 523808 -
作者简介:肖梓腾,男,2001年生,广东省茂名市人,汉族,广东医科大学本科在读,主要从事皮肤组织工程、外泌体相关研究。 -
基金资助:广东省自然科学基金(2021A1515012437),项目负责人:周艳芳;广东省教育厅普通高校科研项目(2021KTSCX040),项目负责人:周艳芳;广东省教育厅普通高校科研项目(2021ZDZX2039),项目负责人:彭新生;国家级大学生创新创业训练计划项目(202210571040),项目负责人:肖梓腾
Exosomes and skin wound healing
Xiao Ziteng1, Wang Tingyu2, Zhang Wenwen2, Tan Fengyi1, Su Haiwei1, Li Siting1, Wu Yahui1, Zhou Yanfang2, Peng Xinsheng3
- 1Second Clinical Medical College, 2School of Basic Medicine, 3School of Pharmacy, Guangdong Medical University, Dongguan 523808, Guangdong Province, China
-
Received:2023-03-09Accepted:2023-04-26Online:2024-07-08Published:2023-09-26 -
Contact:Zhou Yanfang, PhD, Professor, School of Basic Medicine, Guangdong Medical University, Dongguan 523808, Guangdong Province, China Peng Xinsheng, PhD, Professor, School of Pharmacy, Guangdong Medical University, Dongguan 523808, Guangdong Province, China -
About author:Xiao Ziteng, Second Clinical Medical College, Guangdong Medical University, Dongguan 523808, Guangdong Province, China -
Supported by:Guangdong Provincial Natural Science Foundation, No. 2021A1515012437 (to ZYF); Guangdong Provincial Department of Education Regular University Research Project, No. 2021KTSCX040 (to ZYF); General University Research Project of Guangdong Provincial Department of Education, No. 2021ZDZX2039 (to PXS); National College Student Innovation and Entrepreneurship Training Program Project, No. 202210571040 (to XZT)
摘要:
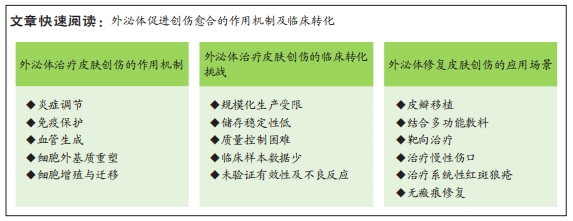
文题释义:
外泌体:是一种直径在40-100 nm的细胞外盘状囊泡。外泌体携带的生物活性物质的类型及功能根据细胞类型的不同而有所不同,主要包括DNA、RNA、蛋白质、脂质、细胞因子等,是细胞间信息交流的重要组成部分。目前外泌体是疾病诊断与治疗、靶向给药、生物疫苗、组织修复再生等领域研究的热点。皮肤创伤修复:是一个复杂的组织修复和重塑过程,涉及炎症反应、细胞增殖和迁移、细胞外基质合成和沉积、血管生成和重塑等生物学进程,需要通过依靠各种类型细胞在时间和空间的紧密协调才能有效恢复受损组织的外观和功能。修复过程受多种内源性及外源性因素影响,若伤口修复相关细胞行为和功能紊乱可导致伤口愈合不良,甚至无法愈合。
背景:外泌体在创伤修复的所有阶段均有作用,目前已有大量外泌体应用于皮肤创伤修复的研究且被证实具有极大的应用潜力。
目的:总结与探讨外泌体治疗皮肤创伤的主要机制及临床应用现状,以促进外泌体的临床转化。方法:检索PubMed数据库、clinicaltrials.gov数据库、中国知网数据库、美国食品药品监督管理局数据库和中国临床试验注册中心,检索时限为各数据库建库至2023年3月,英文检索关键词为“exosomes,wound healing,stem cells,chronic wound,immunoregulation,inflammation,skin,therapeutic use,isolation,characterization,infections”,中文检索关键词为“外泌体,创伤修复,干细胞,免疫调节,临床应用”,最终纳入79篇文献进行总结。
结果与结论:①外泌体能通过炎症调节、免疫保护、血管生成、细胞增殖和迁移、胶原重塑失衡调节等方式改善和加速创面愈合。②干细胞源外泌体制备技术、相关机制研究均较为成熟,是目前主流的研究方向。非干细胞外泌体有方便、经济、易于生产等优点,可作为临床应用的补充。③外泌体的临床应用仍处在起步阶段,但具有极大的应用潜力。各种外泌体改良技术为未来开拓临床个性化服务奠定了基础,需要进一步研究。④外泌体的临床转化面临诸多挑战,如:产量低,高度异质性,未建立统一的分离、纯化和质量控制标准,储存困难等。
https://orcid.org/0009-0005-3333-5362 (肖梓腾)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
肖梓腾, 王婷禹, 张雯雯, 谭凤怡, 苏海威, 李思婷, 吴雅慧, 周艳芳, 彭新生. 外泌体与皮肤创伤的修复[J]. 中国组织工程研究, 2024, 28(19): 3104-3110.
Xiao Ziteng, Wang Tingyu, Zhang Wenwen, Tan Fengyi, Su Haiwei, Li Siting, Wu Yahui, Zhou Yanfang, Peng Xinsheng. Exosomes and skin wound healing[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 3104-3110.
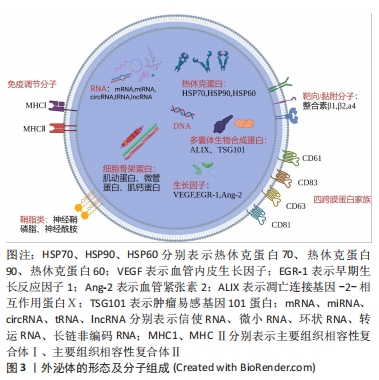
外泌体的分离纯化主要基于其化学、物理和生物特征,常用于分离外泌体的方法主要包括超速离心法、超滤法、聚合物沉淀法、免疫亲和分离法和排阻色谱法,但仍以超速离心法作为外泌体分离的“金标准”[8]。然而,现有的传统技术仍存在产量低、纯度低、回收率低、成本高、提取过程复杂等问题,研究人员更倾向于结合不同的分离技术互补以优化分离结果[9]。鉴于外泌体的高度异质性及目前研究现状,开发通用的外泌体分离方法比较困难。为突破外泌体转化应用的瓶颈,提高分离效率,探索新的分离技术并结合多种技术选择性应用是当前可行的解决方案。
外泌体的表征需要从3个方面考虑,包括外泌体表面蛋白标志物、外泌体大小的纳米颗粒追踪分析鉴定和外泌体形态的透射电子显微镜鉴定[10]。国际细胞外囊泡学会建议需要评估2种类型的蛋白质以证明提取的成分为外泌体,主要包括热休克蛋白(如热休克蛋白70、热休克蛋白90)、四跨膜蛋白超家族(如CD63、CD81)、内体分拣转运复合物相关蛋白(如肿瘤易感基因101蛋白)[11]。虽然可根据外泌体的形态来完成表征,但纯化的外泌体形态的多样性对其提出挑战。ZABEO等[12]观察到从人肥大细胞系HMC-1纯化的外泌体具有高度的形态变异性,这表明即使从单个细胞系中提取,也可能存在形态和功能的不同亚群。
2.2 正常伤口愈合过程 皮肤创口的愈合是一个动态连续的生理过程,需要各种细胞类型及其衍生分子的精细协调以恢复受损组织。典型的伤口修复过程根据其细胞事件、生化反应的不同可以分为一系列重叠阶段,包括止血期、炎症期、增殖期和重塑期。止血在皮肤损伤后立即发生,血小板与胶原蛋白的相互作用导致血小板的活化和聚集,在凝血酶激活的纤维蛋白网作用下最终转化为稳定的凝块。炎症阶段的主要任务是清除组织碎片和病原体,为新组织的产生提供伤口床。免疫细胞的活动在该阶段中起着核心作用,通过直接的活动和旁分泌进一步控制出血并阻止感染。增殖期的开始伴随着炎症期的缓解和免疫细胞的凋亡。这个阶段是伤口实际闭合的阶段,主要包括再上皮化、血管重建和细胞外基质的生成,完成伤口的填充和覆盖。在伤口的重塑阶段,细胞增殖受到抑制,炎症进一步消退,大量胶原蛋白沉积,形成的细胞外基质及胶原纤维被重塑和重新排列,新组织的柔韧性和强度得到提高,最终形成瘢痕组织。愈合过程出现缺陷会导致慢性伤口或者增生性瘢痕[13]。高龄、感染、静脉疾病、糖尿病是慢性伤口发生发展的常见原因。
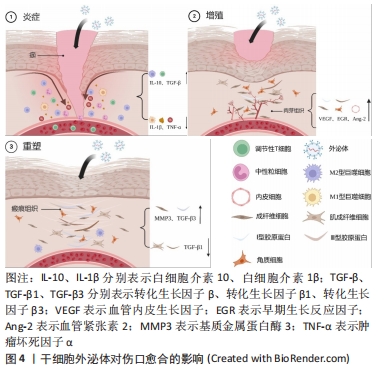
2.3 干细胞来源的外泌体:一种新型的伤口愈合疗法 干细胞疗法被认为在皮肤伤口治疗领域具有很大应用前景,其中又以间充质干细胞受到的关注最多。间充质干细胞具有强大的自我更新能力和多向分化潜力,并可通过调节微环境促进伤口的愈合,大量临床试验已初步证实其促进组织修复的有效性和安全性[14]。然而,细胞保留和存活能力差、细胞培养耗时、致瘤性等问题限制其进一步应用[15]。研究表明,干细胞主要通过旁分泌信号促进修复和再生,外泌体作为其中最为关键的组成部分,具有和干细胞相同的临床益处[16],见图4。与直接的细胞移植相比,外泌体疗法克服干细胞的局限性,不仅保留相应母体细胞的基本特征和功能,而且具有免疫原性低、来源丰富、给药方式便捷、节省时间等突出优点,在促进伤口愈合方面具有巨大的临床潜力[17]。此外,外泌体可以通过过滤灭菌,更适合作为产品生产,所以人们更倾向于使用外泌体而非干细胞进行治疗[18]。
2.3.1 炎症 中性粒细胞是炎症阶段第一种被募集的细胞,它们能通过吞噬作用控制细菌播散,产生活性氧和释放细胞毒性分子,同时促进细胞碎片的分解与清除[19]。伤口愈合不良和中性粒细胞的超额募集有关[20]。
研究证实,间充质干细胞外泌体(mesenchymal stem cells exosome,MSCs-exo)能够阻断中性粒细胞的浸润,防止过度的炎症,促进伤口的愈合[21]。中性粒细胞浸润的抑制是因为伤口环境炎性细胞因子分泌减少,还是MSCs-exo直接作用于中性粒细胞还有待确定。另外,MSCs-exo能够改善中性粒细胞的功能,提高其吞噬能力和活性氧水平,显著降低中性粒细胞的百分比[22]。中性粒细胞过度释放细胞因子似乎可以解释糖尿病患者的中性粒细胞功能障碍[23],而MSCs-exo促进糖尿病伤口愈合的机制是否与影响中性粒细胞的释放功能有关是一个值得考虑的问题。
M1型巨噬细胞在炎症的早期占主导地位,表现为吞噬活性和促炎因子(如白细胞介素1、白细胞介素6、白细胞介素12、肿瘤坏死因子和氧化代谢物)的分泌,以清除病原体和受损组织[24]。在炎症后期,随着M1型巨噬细胞对凋亡中性粒细胞的摄取增加和辅助型T细胞2细胞因子(如白细胞介素4,白细胞介素13和白细胞介素10)的释放,促炎的M1型巨噬细胞转化为抗炎的M2型巨噬细胞,通过纤维化和血管生成局限炎症并促进伤口修复,为进入增殖期做准备[25-26]。巨噬细胞表型的转换决定伤口愈合的速度和质量,向M2表型转型的失败与伤口愈合不良密切相关[27]。已有研究表明MSCs-exo能促进M2型巨噬细胞的极化。LIU等[28]发现褪黑激素预处理的MSCs-exo通过增加M2/M1极化有效减少炎症,有利于伤口的愈合。将MSCs-exo皮下注射到糖尿病大鼠全层伤口中可以观察到M2型巨噬细胞的标志物CD206相对增加,显著减少炎症细胞的浸润[29]。人表皮干细胞外泌体降低细胞因子信号转导抑制剂3的表达并激活Janus激酶2/信号转导和转录激活子3(JAK2/STAT3)信号通路诱导M2型巨噬细胞极化,促进糖尿病伤口愈合[30]。值得一提的是,人脱落乳牙干细胞衍生的外泌体可以增强巨噬细胞自噬,显著促进皮肤组织中白细胞介素10的释放,有利于伤口高质量愈合[31]。
从急性到慢性伤口的潜在机制也需要关注,免疫细胞功能紊乱是慢性伤口的一大特征。T细胞募集发生在炎症反应的后期,调节性T细胞通过抑制CD4+ T辅助性淋巴细胞和CD8+ T辅助性淋巴细胞功能限制炎症,调节性T细胞的耗竭将损伤正常的急性伤口愈合[32-33]。T细胞浸润伤口对伤口愈合至关重要,这一过程的延迟和损伤会导致愈合不足[34]。干细胞来源的外泌体在免疫调节方面具有巨大潜力。在体外实验中,人脐血间充质干细胞衍生的外泌体通过提高miR-17b水平靶向Krüppel样因子19表达,调节17型辅助T细胞/调节性T细胞平衡,最终抑制炎症因子表达[35]。鼠脂肪干细胞外泌体对17型辅助T细胞/调节性T细胞平衡的调节作用能有效抑制肥厚性瘢痕形成[36]。MSCs-exo能够激活调节性T细胞自噬和信号转导及转录激活因子5信号通路以增强调节性T细胞的抑制能力和功能稳定性。另外,伤口反复感染会导致慢性伤口,树突状细胞作为主要抗原呈递细胞协调先天性和适应性免疫,是防止感染的重要因素。一项研究显示MSCs-exo在诱导耐受性树突状细胞形成的同时抑制淋巴细胞的活性,增强白细胞介素10和转化生长因子β的分泌[37]。用间充质干细胞衍生的细胞外囊泡处理的树突状细胞抑制T细胞增殖,可减少白细胞介素6和白细胞介素12p70炎性细胞因子的分泌[38]。最近有研究表明,间充质干细胞分泌因子可抑制金黄色葡萄球菌生物膜形成,并破坏已生成的生物膜,与抗生素有协同作用[39]。马外周血来源间充质干细胞分泌组抑制皮肤伤口中各种常见的细菌生长,包括耐甲氧西林金黄色葡萄球菌,这与半胱氨酸蛋白酶的分泌有关[40]。然而,关于外泌体在皮肤创面抗菌特性的研究尚未开展,需要进一步研究外泌体在其中参与的作用及不同来源细胞外泌体抗菌效果的差异。外泌体炎症调节、免疫保护的作用机制,见表1。
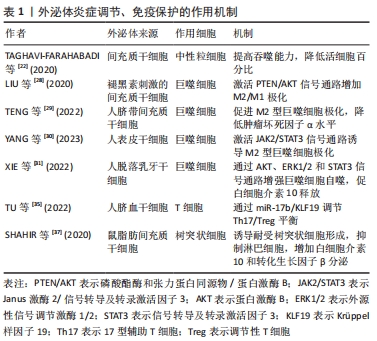
2.3.2 增殖 增殖阶段主要有4个过程,包括成纤维细胞增殖、细胞外基质沉积、血管生成和再上皮化。大量证据表明,外泌体对4个过程均有积极的治疗作用。
MSCs-exo可以刺激成纤维细胞的增殖、迁徙和细胞外基质产生。成纤维细胞作为参与产生细胞外基质的关键效应细胞,在伤口愈合中承担重要的作用。人脂肪干细胞外泌体富集的长链非编码RNA GAS5能够减轻人真皮成纤维细胞中的氧化应激,并显著提高成纤维细胞的迁徙能力。若耗竭GAS5则显著降低外泌体促进伤口愈合的能力[41]。 此外,人羊膜间充质干细胞外泌体携带的miR-135a能够增强成纤维细胞的活性,抑制钙黏蛋白E、神经钙黏蛋白和大肿瘤抑制激酶2表达,同时促进α平滑肌肌动蛋白表达[42]。MSCs-exo对成纤维细胞的调控是多层次、多途径的,这些信号通路在伤口愈合中很重要。人脂肪间充质干细胞衍生的外泌体通过WNT/β-连环蛋白信号通路的激活促进伤口愈合,并且以剂量依赖性方式促进成纤维细胞的迁移和增殖,极大提高Ⅰ型胶原蛋白和Ⅲ型胶原蛋白的表达水平[43]。骨髓间充质干细胞外泌体可通过p44/42有丝分裂原激活蛋白激酶途径增强成纤维细胞修复功能[44]。缺氧诱导的脂肪干细胞外泌体则通过磷脂酰肌醇-3-激酶/蛋白激酶B(PI3K/AKT)信号通路促进糖尿病伤口愈合[45]。
MSCs-exo还可以刺激修复相关生长因子表达,促进内皮细胞的增殖、迁移和血管内皮生长因子分泌,最终促进血管生成。FU等[46]发现人羊膜间充质干细胞外泌体可改善高葡萄糖环境下人脐静脉内皮细胞的功能特性。进一步研究发现,人羊膜间充质干细胞外泌体富集PANTR1、H19、OIP5-AS1和NR2F1-AS1等一系列与血管生成相关的长链非编码RNA。在外泌体中还发现血管生成素2和早期生长反应因子1,MSCs-exo可通过血管生成素2与早期生长反应因子1的递送增强血管生成[47-48]。近年来,关于干细胞外泌体转移RNA的研究颇多,例如
miR-125a-3p[49]、miR-383[50]、miR-132和miR-146a的转移均可刺激血管生成促进伤口愈合[51]。伤口环境代谢活跃,需要新血管形成以维持修复的正常进行,血管生成是愈合过程至关重要的一步。如果血管生成受损以至于不能够提供充足的代谢营养素则会破坏愈合过程,导致慢性伤口的形成。
最后,MSCs-exo加速伤口的再上皮化。在这期间,角质形成细胞增殖伴随上皮-间充质转化,通过板状爬行覆盖伤口,初步建立起皮肤屏障[52]。体外研究发现,脂肪干细胞外泌体的使用降低角质形成细胞活性氧水平,使其免受缺氧和氧化应激损伤的同时增强迁移和增殖的能力,这种作用或许是通过WNT/β-连环蛋白信号[53]、蛋白激酶B/低氧诱导因子1α信号通路的传导实现的[54]。另外,研究证明miRNA在其中也是关键的一环。人脂肪干细胞外泌体中高表达的miR-21增强人永生化表皮细胞迁移和增殖能力,同时经磷脂酰肌醇-3-激酶/蛋白激酶B信号通路影响基质金属蛋白酶2和组织金属蛋白酶抑制因子1蛋白表达[55]。值得一提的是,成纤维细胞可以通过分泌角质形成细胞衍生的生长因子刺激角质形成细胞增殖和迁移[56]。MSCs-exo能通过调节角质形成细胞和成纤维细胞的特征,增强再上皮化来促进伤口愈合。人脂肪干细胞外泌体中含有高水平的miR-100-5p,miR-100-5p通过肌微管素相关蛋白3上调3,4,5-三磷酸磷脂酰肌醇并激活蛋白激酶B和细胞外信号调节激酶信号通路促进表皮干细胞增殖,提示外泌体可以通过刺激皮肤中驻留的干细胞促进修复[57]。见表2。在现有研究中,MSCs-exo在再上皮化过程作用的探索仍相对较少,加之啮齿动物与人类伤口闭合的差异性在一定程度上会影响结果的准确性,需要更多的研究来确定MSCs-exo如何影响再上皮化过程[58]。

2.3.3 重塑 如何减少瘢痕的形成是创口重塑阶段主要的工作内容。在这个阶段,Ⅲ型胶原蛋白逐渐转化为Ⅰ型胶原蛋白并形成更厚、更有组织性的胶原束,成纤维细胞分化为肌成纤维细胞以收缩伤口,基质金属蛋白酶将对细胞外基质进行修饰改造,最终形成瘢痕组织。若愈合过程任何阶段受到破坏则可能会导致萎缩性瘢痕、增生性瘢痕、瘢痕疙瘩和慢性伤口[59]。
外泌体在重塑阶段的调节是多方面的。MSCs-exo已被证明可以减轻病理性瘢痕,而这种能力主要依赖于miR-138-5p下调sirtuin 1,抑制成纤维细胞的生长和蛋白表达[60]。人羊膜上皮细胞外泌体中miR-let-7d通过降低二价金属转运蛋白1的表达减少铁摄取,减轻肥厚性瘢痕纤维化[61]。此外,miR-192-5p在脂肪干细胞外泌体中高度富集,可抑制成纤维细胞中的白细胞介素17RA/Smad通路,降低胶原蛋白沉积速度、抑制成纤维细胞向肌成纤维细胞转分化以及增生性瘢痕形成[62]。ZHANG等[63]的研究进一步阐述外泌体在重塑阶段的作用。他们发现人胎盘间充质干细胞外泌体能够改善伤口愈合的质量,促进皮肤附属物的生成,增加Ⅲ型胶原蛋白分泌同时减少Ⅰ型胶原蛋白生成,改善愈合皮肤中的胶原模式。外泌体还可以通过促进血管生成和抑制炎症间接调节成纤维细胞和肌成纤维细胞,减少瘢痕形成[64]。用人羊水干细胞外泌体处理小鼠伤口可以加速伤口愈合并缓解瘢痕形成,进一步研究显示人羊水干细胞外泌体靶向CXCR146,通过miR-5a-4p减少炎症相关细胞因子的分 泌[65]。JIANG等[66]的研究也佐证了这一点,MSCs-exo可通过调节炎症和减少胶原蛋白沉积来抑制瘢痕形成。
2.4 非干细胞源外泌体在伤口愈合中的作用 到目前为止,研究用于伤口愈合的外泌体主要来自干细胞,然而干细胞需要更多资料来证明其生物安全性,故研究者考虑选用更稳定的动物来源蛋白外泌体作为替代方案。最近研究发现,小鼠来源血清外泌体通过刺激成纤维细胞增殖、促进人永生化表皮细胞迁移并抑制其热诱导的凋亡以加速小鼠烫伤伤口的愈合[67]。在糖尿病小鼠伤口模型中,血清外泌体促进细胞外基质形成和CD31、纤连蛋白和胶原蛋白α的表达,并通过增强血管形成缩短伤口愈合时间[68]。牛奶外泌体抑制促炎递质白细胞介素6、肿瘤坏死因子α以及促炎趋化因子环氧化酶2和诱导型一氧化氮合酶的表达,调节转化生长因子β亚型水平以促进无瘢痕愈合[69]。更重要的是,牛奶外泌体产量相对较高且易于获取,是大规模生产的理想来源。MI等[70]发现唾液外泌体在体外诱导人脐静脉内皮细胞增殖、迁移和血管生成并促进体内皮肤伤口愈合。进一步实验证明,唾液外泌体中的泛素偶联酶E2O降低smad家族成员6水平,进而激活骨形态发生蛋白2,最终诱导血管生成。
另外,植物来源的外泌体样纳米囊泡在最近也得到关注,其特征类似于哺乳动物外泌体,直径为40-150 nm,能够跨界产生生物学效应。相比于哺乳动物外泌体,植物外泌体不携带人畜共患和人类病原体,具有一定的靶向性和大规模生产的巨大潜力,部分植物外泌体还具备一定的伤口愈合特性[71-72]。目前,关于植物外泌体的研究方兴未艾,但作为一种更经济、更安全的替代方案,可以预见植物外泌体的广阔前景。
干细胞外泌体基于其亲本细胞强大的免疫调节能力、自我更新能力、多向分化潜力及免疫原性低和来源广泛等诸多优点,仍是目前主流的研究方向,其制备技术、相关机制研究均较为成熟。与干细胞来源外泌体相比,非干细胞来源外泌体的优点集中在获取方便、成本低廉、易于生产且具有促进伤口修复的能力。然而,非干细胞源外泌体普遍存在表征困难、分离和纯化标准不统一、命名不明确的问题,对生物发生、释放、内化和作用机制的理解仍然十分有限。当前关于不同来源外泌体的横向研究较少,需要更多研究比较不同来源外泌体的优缺点和作用机制,而非干细胞源外泌体作为外泌体应用必要的补充,其研究有利于临床应对更复杂的皮肤伤口。
2.5 临床应用现状 外泌体研究如今仍处在初级阶段,实现临床前研究到临床应用仍具有极大的探索价值。根据 ClinicalTrials.gov的检索结果,只有4项关于皮肤伤口愈合的临床试验且均未发表相关结果,见表3,而中国临床试验注册中心尚未有相关临床试验的注册,目前仍缺乏足够的临床试验证明其有效性和安全性。美国食品药品监督管理局曾发布了患者使用未经注册外泌体产品引起严重不良反应的报告并对消费者使用外泌体产品发出警告,目前仍未有注册的外泌体产品。因此,亟需更多高质量的临床研究。一项前瞻性、双盲、随机临床试验显示,用人脂肪干细胞外泌体和一氧化碳联合激光处理痤疮瘢痕较对照组有明显效果,且治疗后相关红斑减轻,人脂肪干细胞外泌体与治疗装置的联合使用可以产生协同作用[73]。人脂肪干细胞外泌体已被证明在急性口服毒性、皮肤致敏及体外光敏化实验中均不会引起任何刺激或毒性,初步证实人脂肪干细胞外泌体的安全性[74]。另外,QIAO等[75]的Meta分析提示外泌体疗法能够提高糖尿病伤口愈合质量,这一观点与BAILEY等[76]的研究相似。

由于外泌体来源不同及高度异质性,难以建立严格的质量控制指标;大规模生产技术不成熟、储存效率低下等问题尚未解决;给药的安全性和有效性还没有得到证实,这些问题显著放缓了外泌体的临床转化速度。目前研究主要集中在鼠模型与人源或鼠源外泌体应用上,然而由于人体皮肤与鼠皮肤组织的种族差异和2种外泌体在蛋白质组与基因组表达水平的差异,通过临床前研究难以预测外泌体在人体皮肤的应用[77]。在未来的研究中,需要进行大规模的随机双盲试验并进行高质量的随访,以获得最佳外泌体治疗参数;采用恰当的方法评估外泌体在伤口的分布、清除和治疗效率;建立标准化的外泌体分离方案。
外泌体在治疗皮肤创口时可协同传统治疗方法,如换药和负压治疗等,也可应用于皮瓣修复,减少皮瓣坏死[78]。此外,外泌体的给药途径也是值得考虑的问题,有研究显示静脉注射相比于局部注射更有利于伤口愈合,这可能是因为局部注射期间不仅会导致外泌体的损失,还会进一步破坏伤口的愈合过程[79]。然而,静脉注射受影响因素较多,难以维持在较高浓度,采用敷料或许是更好的解决方案。当外泌体被应用在临床时,如何提高外泌体伤口愈合功效和产量是一个值得探讨的问题。为此,众多学者做出许多努力,诸如工程外泌体的设计、结合生物活性支架、预处理细胞、采用中空纤维生物反应器和微流控装置等技术可有效推进其临床转化。针对不同的伤口病因、并发症和潜在的生物学缺陷,或许将采用不同的疗法以促进伤口更好地愈合。

| [1] EMING SA, MARTIN P, TOMIC-CANIC M. Wound repair and regeneration: mechanisms, signaling, and translation. Sci Transl Med. 2014;6(265):265sr6. [2] MATHIEU M, MARTIN-JAULAR L, LAVIEU G, et al. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat Cell Biol. 2019;21(1):9-17. [3] VALADI H, EKSTRÖM K, BOSSIOS A, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654-659. [4] BARILE L, VASSALLI G. Exosomes: Therapy delivery tools and biomarkers of diseases. Pharmacol Ther. 2017;174:63-78. [5] ZHANG XA, HUANG C. Tetraspanins and cell membrane tubular structures. Cell Mol Life Sci. 2012;69(17):2843-2852. [6] RECORD M, CARAYON K, POIROT M, et al. Exosomes as new vesicular lipid transporters involved in cell-cell communication and various pathophysiologies. Biochim Biophys Acta. 2014;1841(1):108-120. [7] YÁÑEZ-MÓ M, SILJANDER PR, ANDREU Z, et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015; 4:27066. [8] XU WM, LI A, CHEN JJ, et al. Research Development on Exosome Separation Technology. J Membr Biol. 2023;256(1):25-34. [9] LIU WZ, MA ZJ, KANG XW. Current status and outlook of advances in exosome isolation. Anal Bioanal Chem. 2022;414(24):7123-7141. [10] ZHANG Y, BI J, HUANG J, et al. Exosome: A Review of Its Classification, Isolation Techniques, Storage, Diagnostic and Targeted Therapy Applications. Int J Nanomedicine. 2020;15:6917-6934. [11] THÉRY C, WITWER KW, AIKAWA E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750. [12] ZABEO D, CVJETKOVIC A, LÄSSER C, et al. Exosomes purified from a single cell type have diverse morphology. J Extracell Vesicles. 2017;6(1):1329476. [13] FRYKBERG RG, BANKS J. Challenges in the Treatment of Chronic Wounds. Adv Wound Care (New Rochelle). 2015;4(9):560-582. [14] HSIEH MW, WANG WT, LIN CY, et al. Stem Cell-Based Therapeutic Strategies in Diabetic Wound Healing. Biomedicines. 2022;10(9):2085. [15] HOSSEINI M, DALLEY AJ, SHAFIEE A. Convergence of Biofabrication Technologies and Cell Therapies for Wound Healing. Pharmaceutics. 2022;14(12):2749. [16] NIKFARJAM S, REZAIE J, ZOLBANIN NM, et al. Mesenchymal stem cell derived-exosomes: a modern approach in translational medicine. J Transl Med. 2020; 18(1):449. [17] WU J, CHEN LH, SUN SY, et al. Mesenchymal stem cell-derived exosomes: The dawn of diabetic wound healing. World J Diabetes. 2022;13(12):1066-1095. [18] HAN C, SUN X, LIU L, et al. Exosomes and Their Therapeutic Potentials of Stem Cells. Stem Cells Int. 2016;2016:7653489. [19] PHILLIPSON M, KUBES P. The Healing Power of Neutrophils. Trends Immunol. 2019;40(7):635-647. [20] WILGUS TA, ROY S, MCDANIEL JC. Neutrophils and Wound Repair: Positive Actions and Negative Reactions. Adv Wound Care (New Rochelle). 2013;2(7): 379-388. [21] SHOJAATI G, KHANDAKER I, FUNDERBURGH ML, et al. Mesenchymal Stem Cells Reduce Corneal Fibrosis and Inflammation via Extracellular Vesicle-Mediated Delivery of miRNA. Stem Cells Transl Med. 2019;8(11):1192-1201. [22] TAGHAVI-FARAHABADI M, MAHMOUDI M, MAHDAVIANI SA, et al. Improving the function of neutrophils from chronic granulomatous disease patients using mesenchymal stem cells’ exosomes. Hum Immunol. 2020;81(10-11):614-624. [23] LIN Q, ZHOU W, WANG Y, et al. Abnormal Peripheral Neutrophil Transcriptome in Newly Diagnosed Type 2 Diabetes Patients. J Diabetes Res. 2020;2020:9519072. [24] BONIAKOWSKI AE, KIMBALL AS, JACOBS BN, et al. Macrophage-Mediated Inflammation in Normal and Diabetic Wound Healing. J Immunol. 2017;199(1): 17-24. [25] CHEN C, LIU T, TANG Y, et al. Epigenetic regulation of macrophage polarization in wound healing. Burns Trauma. 2023;11:tkac057. [26] XU X, GU S, HUANG X, et al. The role of macrophages in the formation of hypertrophic scars and keloids. Burns Trauma. 2020;8:tkaa006. [27] GANESH GV, RAMKUMAR KM. Macrophage mediation in normal and diabetic wound healing responses. Inflamm Res. 2020;69(4):347-363. [28] LIU W, YU M, XIE D, et al. Melatonin-stimulated MSC-derived exosomes improve diabetic wound healing through regulating macrophage M1 and M2 polarization by targeting the PTEN/AKT pathway. Stem Cell Res Ther. 2020;11(1):259. [29] TENG L, MAQSOOD M, ZHU M, et al. Exosomes Derived from Human Umbilical Cord Mesenchymal Stem Cells Accelerate Diabetic Wound Healing via Promoting M2 Macrophage Polarization, Angiogenesis, and Collagen Deposition. Int J Mol Sci. 2022;23(18):10421. [30] YANG H, XU H, WANG Z, et al. Analysis of miR-203a-3p/SOCS3-mediated induction of M2 macrophage polarization to promote diabetic wound healing based on epidermal stem cell-derived exosomes. Diabetes Res Clin Pract. 2023; 197:110573. [31] XIE Y, YU L, CHENG Z, et al. SHED-derived exosomes promote LPS-induced wound healing with less itching by stimulating macrophage autophagy. J Nanobiotechnology. 2022;20(1):239. [32] BOOTHBY IC, COHEN JN, ROSENBLUM MD. Regulatory T cells in skin injury: At the crossroads of tolerance and tissue repair. Sci Immunol. 2020;5(47):eaaz9631. [33] HAERTEL E, JOSHI N, HIEBERT P, et al. Regulatory T cells are required for normal and activin-promoted wound repair in mice. Eur J Immunol. 2018;48(6): 1001-1013. [34] BROCKMANN L, GIANNOU AD, GAGLIANI N, et al. Regulation of TH17 Cells and Associated Cytokines in Wound Healing, Tissue Regeneration, and Carcinogenesis. Int J Mol Sci. 2017;18(5):1033. [35] TU J, ZHENG N, MAO C, et al. UC-BSCs Exosomes Regulate Th17/Treg Balance in Patients with Systemic Lupus Erythematosus via miR-19b/KLF13. Cells. 2022; 11(24):4123. [36] WANG H, LI Y, YUE Z, et al. Adipose-Derived Stem Cell Exosomes Inhibit Hypertrophic Scaring Formation by Regulating Th17/Treg Cell Balance. Biomed Res Int. 2022;2022:9899135. [37] SHAHIR M, MAHMOUD HASHEMI S, ASADIRAD A, et al. Effect of mesenchymal stem cell-derived exosomes on the induction of mouse tolerogenic dendritic cells. J Cell Physiol. 2020;235(10):7043-7055. [38] REIS M, MAVIN E, NICHOLSON L, et al. Mesenchymal Stromal Cell-Derived Extracellular Vesicles Attenuate Dendritic Cell Maturation and Function. Front Immunol. 2018;9:2538. [39] CHOW L, JOHNSON V, IMPASTATO R, et al. Antibacterial activity of human mesenchymal stem cells mediated directly by constitutively secreted factors and indirectly by activation of innate immune effector cells. Stem Cells Transl Med. 2020;9(2):235-249. [40] MARX C, GARDNER S, HARMAN RM, et al. The mesenchymal stromal cell secretome impairs methicillin-resistant Staphylococcus aureus biofilms via cysteine protease activity in the equine model. Stem Cells Transl Med. 2020; 9(7):746-757. [41] PATEL RS, IMPRESO S, LUI A, et al. Long Noncoding RNA GAS5 Contained in Exosomes Derived from Human Adipose Stem Cells Promotes Repair and Modulates Inflammation in a Chronic Dermal Wound Healing Model. Biology (Basel). 2022;11(3):426. [42] CHEN T, GAO S, HAO Y, et al. Experimental study of human amniotic mesenchymal stem cell exosome promoting fibroblasts migration through microRNA-135a. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2020;34(2):234-239. [43] LI C, AN Y, SUN Y, et al. Adipose Mesenchymal Stem Cell-Derived Exosomes Promote Wound Healing Through the WNT/β-catenin Signaling Pathway in Dermal Fibroblasts. Stem Cell Rev Rep. 2022;18(6):2059-2073. [44] ZHOU J, DING Y, ZHANG Y, et al. Exosomes from bone marrow-derived mesenchymal stem cells facilitate corneal wound healing via regulating the p44/42 MAPK pathway. Graefes Arch Clin Exp Ophthalmol. 2023;261(3):723-734. [45] WANG J, WU H, PENG Y, et al. Hypoxia adipose stem cell-derived exosomes promote high-quality healing of diabetic wound involves activation of PI3K/Akt pathways. J Nanobiotechnology. 2021;19(1):202. [46] FU S, ZHANG H, LI X, et al. Exosomes Derived from Human Amniotic Mesenchymal Stem Cells Facilitate Diabetic Wound Healing by Angiogenesis and Enrich Multiple lncRNAs. Tissue Eng Regen Med. 2023;20(2):295-308. [47] LIU J, YAN Z, YANG F, et al. Exosomes Derived from Human Umbilical Cord Mesenchymal Stem Cells Accelerate Cutaneous Wound Healing by Enhancing Angiogenesis through Delivering Angiopoietin-2. Stem Cell Rev Rep. 2021;17(2): 305-317. [48] SUN Y, JU Y, FANG B. Exosomes from human adipose-derived mesenchymal stromal/stem cells accelerate angiogenesis in wound healing: implication of the EGR-1/lncRNA-SENCR/DKC1/VEGF-A axis. Hum Cell. 2022;35(5):1375-1390. [49] PI L, YANG L, FANG BR, et al. Exosomal microRNA-125a-3p from human adipose-derived mesenchymal stem cells promotes angiogenesis of wound healing through inhibiting PTEN. Mol Cell Biochem. 2022;477(1):115-127. [50] HAN ZF, CAO JH, LIU ZY, et al. Exosomal lncRNA KLF3-AS1 derived from bone marrow mesenchymal stem cells stimulates angiogenesis to promote diabetic cutaneous wound healing. Diabetes Res Clin Pract. 2022;183:109126. [51] HEO JS, KIM S. Human adipose mesenchymal stem cells modulate inflammation and angiogenesis through exosomes. Sci Rep. 2022;12(1):2776. [52] SORG H, SORG CGG. Skin wound healing: of players, patterns and processes. Eur Surg Res. 2022. doi: 10.1159/000528271. [53] MA T, FU B, YANG X, et al. Adipose mesenchymal stem cell-derived exosomes promote cell proliferation, migration, and inhibit cell apoptosis via Wnt/β-catenin signaling in cutaneous wound healing. J Cell Biochem. 2019;120(6):10847-10854. [54] ZHANG Y, HAN F, GU L, et al. Correction to: Adipose mesenchymal stem cell exosomes promote wound healing through accelerated keratinocyte migration and proliferation by activating the AKT/HIF-1α axis. J Mol Histol. 2020;51(4):467. [55] YANG C, LUO L, BAI X, et al. Highly-expressed micoRNA-21 in adipose derived stem cell exosomes can enhance the migration and proliferation of the HaCaT cells by increasing the MMP-9 expression through the PI3K/AKT pathway. Arch Biochem Biophys. 2020;681:108259. [56] LV H, LIU H, SUN T, et al. Exosome derived from stem cell: A promising therapeutics for wound healing. Front Pharmacol. 2022;13:957771. [57] LIU Z, YANG Y, JU J, et al. miR-100-5p Promotes Epidermal Stem Cell Proliferation through Targeting MTMR3 to Activate PIP3/AKT and ERK Signaling Pathways. Stem Cells Int. 2022;2022:1474273. [58] GRAMBOW E, SORG H, SORG CGG, et al. Experimental Models to Study Skin Wound Healing with a Focus on Angiogenesis. Med Sci (Basel). 2021;9(3):55. [59] BRAY ER, OROPALLO AR, GRANDE DA, et al. Extracellular Vesicles as Therapeutic Tools for the Treatment of Chronic Wounds. Pharmaceutics. 2021;13(10):1543. [60] ZHAO W, ZHANG R, ZANG C, et al. Exosome Derived from Mesenchymal Stem Cells Alleviates Pathological Scars by Inhibiting the Proliferation, Migration and Protein Expression of Fibroblasts via Delivering miR-138-5p to Target SIRT1. Int J Nanomedicine. 2022;17:4023-4038. [61] ZHAO B, SHI X, FENG D, et al. MicroRNA let-7d attenuates hypertrophic scar fibrosis through modulation of iron metabolism by reducing DMT1 expression. J Mol Histol. 2023;54(1):77-87. [62] LI Y, ZHANG J, SHI J, et al. Exosomes derived from human adipose mesenchymal stem cells attenuate hypertrophic scar fibrosis by miR-192-5p/IL-17RA/Smad axis. Stem Cell Res Ther. 2021;12(1):221. [63] ZHANG Y, SHI L, LI X, et al. Placental stem cells-derived exosomes stimulate cutaneous wound regeneration via engrailed-1 inhibition. Front Bioeng Biotechnol. 2022;10:1044773. [64] LI C, WEI S, XU Q, et al. Application of ADSCs and their Exosomes in Scar Prevention. Stem Cell Rev Rep. 2022;18(3):952-967. [65] WGEALLA MMAMA, LIANG H, CHEN R, et al. Amniotic fluid derived stem cells promote skin regeneration and alleviate scar formation through exosomal miRNA-146a-5p via targeting CXCR4. J Cosmet Dermatol. 2022;21(10):5026-5036. [66] JIANG L, ZHANG Y, LIU T, et al. Exosomes derived from TSG-6 modified mesenchymal stromal cells attenuate scar formation during wound healing. Biochimie. 2020;177:40-49. [67] LI Y, YU Y, XIE Z, et al. Serum-derived exosomes accelerate scald wound healing in mice by optimizing cellular functions and promoting Akt phosphorylation. Biotechnol Lett. 2021;43(8):1675-1684. [68] CHEN L, QIN L, CHEN C, et al. Serum exosomes accelerate diabetic wound healing by promoting angiogenesis and ECM formation. Cell Biol Int. 2021;45(9):1976-1985. [69] AHN G, KIM YH, AHN JY. Multifaceted effects of milk-exosomes (Mi-Exo) as a modulator of scar-free wound healing. Nanoscale Adv. 2020;3(2):528-537. [70] MI B, CHEN L, XIONG Y, et al. Saliva exosomes-derived UBE2O mRNA promotes angiogenesis in cutaneous wounds by targeting SMAD6. J Nanobiotechnology. 2020;18(1):68. [71] DAD HA, GU TW, ZHU AQ, et al. Plant Exosome-like Nanovesicles: Emerging Therapeutics and Drug Delivery Nanoplatforms. Mol Ther. 2021;29(1):13-31. [72] NARAUSKAITĖ D, VYDMANTAITĖ G, RUSTEIKAITĖ J, et al. Extracellular Vesicles in Skin Wound Healing. Pharmaceuticals (Basel). 2021;14(8):811. [73] KWON HH, YANG SH, LEE J, et al. Combination Treatment with Human Adipose Tissue Stem Cell-derived Exosomes and Fractional CO2 Laser for Acne Scars: A 12-week Prospective, Double-blind, Randomized, Split-face Study. Acta Derm Venereol. 2020;100(18):adv00310. [74] HA DH, KIM SD, LEE J, et al. Toxicological evaluation of exosomes derived from human adipose tissue-derived mesenchymal stem/stromal cells. Regul Toxicol Pharmacol. 2020;115:104686. [75] QIAO Z, WANG X, ZHAO H, et al. The effectiveness of cell-derived exosome therapy for diabetic wound: A systematic review and meta-analysis. Ageing Res Rev. 2023;85:101858. [76] BAILEY AJM, LI H, KIRKHAM AM, et al. MSC-Derived Extracellular Vesicles to Heal Diabetic Wounds: a Systematic Review and Meta-Analysis of Preclinical Animal Studies. Stem Cell Rev Rep. 2022;18(3):968-979. [77] XIONG M, ZHANG Q, HU W, et al. The novel mechanisms and applications of exosomes in dermatology and cutaneous medical aesthetics. Pharmacol Res. 2021;166:105490. [78] 张璟琳,冷敏,朱博恒,等.干细胞源外泌体促进糖尿病创面愈合的机制及应用[J].中国组织工程研究,2022,26(7):1113-1118. [79] HU L, WANG J, ZHOU X, et al. Exosomes derived from human adipose mensenchymal stem cells accelerates cutaneous wound healing via optimizing the characteristics of fibroblasts. Sci Rep. 2016;6:32993. |
| [1] | 余伟杰, 刘爱峰, 陈继鑫, 郭天赐, 贾易臻, 冯汇川, 杨家麟. 机器学习在腰椎间盘突出症诊治中的优势和应用策略[J]. 中国组织工程研究, 2024, 28(9): 1426-1435. |
| [2] | 杨玉芳, 杨芷姗, 段棉棉, 刘毅恒, 唐正龙, 王 宇. 促红细胞生成素在骨组织工程中的应用及前景[J]. 中国组织工程研究, 2024, 28(9): 1443-1449. |
| [3] | 陈凯佳, 刘景云, 曹 宁, 孙建波, 周 燕, 梅建国, 任 强. 组织工程技术在股骨头坏死治疗中的应用及前景[J]. 中国组织工程研究, 2024, 28(9): 1450-1456. |
| [4] | 白 晨, 杨文骞, 孟志超, 王宇泽. 损伤前交叉韧带修复及促进移植物愈合的策略[J]. 中国组织工程研究, 2024, 28(9): 1457-1463. |
| [5] | 林泽玉, 徐 林. 痛风致骨破坏机制的研究与进展[J]. 中国组织工程研究, 2024, 28(8): 1295-1300. |
| [6] | 徐灿丽, 何文星, 汪 磊, 吴芳婷, 王佳慧, 段雪琳, 赵铁建, 赵 斌, 郑 洋. 肝脏类器官研究的文献计量学分析[J]. 中国组织工程研究, 2024, 28(7): 1099-1104. |
| [7] | 刘瀚峰, 王晶晶, 余云生. 人造外泌体治疗心肌梗死:应用现状及前景[J]. 中国组织工程研究, 2024, 28(7): 1118-1123. |
| [8] | 马树微, 何 生, 韩 冰, 张缭云. 间充质干细胞来源外泌体治疗动物急性肝衰竭的Meta分析[J]. 中国组织工程研究, 2024, 28(7): 1137-1142. |
| [9] | 孙宇康, 宋丽娟, 温春丽, 丁智斌, 田 昊, 马 东, 马存根, 翟晓艳. 基于Web of Science近十年干细胞治疗心肌梗死的可视化分析[J]. 中国组织工程研究, 2024, 28(7): 1143-1148. |
| [10] | 冯睿钦, 韩 娜, 张 蒙, 谷馨怡, 张丰识. 1%富血小板血浆联合骨髓间充质干细胞促进周围神经损伤的修复[J]. 中国组织工程研究, 2024, 28(7): 985-992. |
| [11] | 王 雯, 郑芃芃, 孟浩浩, 刘 浩, 袁长永. 过表达Sema3A促进牙髓干细胞和MC3T3-E1的成骨分化[J]. 中国组织工程研究, 2024, 28(7): 993-999. |
| [12] | 邱晓燕, 李碧欣, 黎敬弟, 范垂钦, 马 廉, 王鸿武. MAFA-PDX1过表达慢病毒感染人脐带间充质干细胞向胰岛素分泌细胞的分化[J]. 中国组织工程研究, 2024, 28(7): 1000-1006. |
| [13] | 刘麒薇, 张俊辉, 杨 袁, 王金娟. 脐带间充质干细胞治疗多囊卵巢综合征的作用及机制[J]. 中国组织工程研究, 2024, 28(7): 1015-1020. |
| [14] | 梅静怡, 刘 江, 肖 聪, 刘 鹏, 周浩浩, 林展翼. 组织工程血管构建过程中平滑肌细胞增殖变化及代谢模式[J]. 中国组织工程研究, 2024, 28(7): 1043-1049. |
| [15] | 王姗姗, 舒 晴, 田 峻. 物理因子促进干细胞的成骨分化[J]. 中国组织工程研究, 2024, 28(7): 1083-1090. |
外泌体作为细胞旁分泌的重要组成部分在各种疾病中均发挥重要作用,并成为再生医学领域研究的热点。外泌体继承亲本细胞的特征,能通过炎症调节、免疫保护、血管生成、细胞增殖和迁移、胶原重塑失衡调节等方式改善和加速创面愈合,具有广阔的应用前景。各种改良外泌体及生产技术的更新,诸如预处理细胞、工程外泌体、微控流分离技术的涌现让其临床应用成为可能。
目前关于外泌体应用于皮肤创伤修复的研究在近几年呈爆发性增长,文章拟就外泌体在皮肤创伤愈合中最新的研究进展作一综述,对前人的研究补充说明,针对创伤修复的病理生理学过程及作用机制进行阐述,比较不同来源外泌体治疗的优缺点,回顾外泌体临床应用现状并结合自己的理解探索未来研究方向,为其进一步临床转化及临床个性化服务铺平道路。 中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1.1 检索人及检索时间 第一作者在2023年1月进行检索。
1.1.2 检索文献时限 2023年前发表的中英文文献,重点检索近5年发表的英文文献。
1.1.3 检索数据库 PubMed数据库、clinicaltrials.gov数据库、中国知网数据库、美国食品药品监督管理局数据库和中国临床试验注册中心。
1.1.4 检索途径 主题词、标题/摘要、关键词检索。
1.1.5 检索词 英文检索词为“exosomes,wound healing,stem cells,chronic wound,immunoregulation,inflammation,skin,therapeutic use,isolation,characterization,infections”;中文检索词为“外泌体,创伤修复,干细胞,免疫调节,临床应用”。
1.1.6 检索文献类型 研究原著和综述。
1.1.7 检索策略 PudMed 数据库检索策略见图1。

1.2 入组标准
1.2.1 纳入标准 ①论据充分,立意新颖,有权威性的文献;②关于外泌体临床应用与临床试验的文献;③描述外泌体治疗皮肤伤口机制的文献。
1.2.2 排除标准 ①观点陈旧,时间过长的文献;②研究内容相关性差,重复性的文献;③无法完整获取的文献。
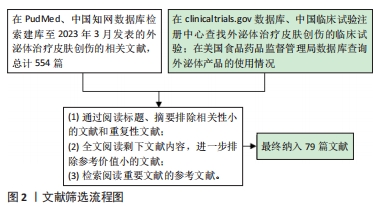
1.3 文献质量评估和数据的提取 初步检索相关文献总计554篇,根据纳入标准和排除标准进行筛选,最终纳入79篇文献,其中来源于PubMed数据库78篇,来源于中国知网数据库1篇。文献筛选流程图,见图2。
3.2 作者综述区别于他人他篇的特点 该综述结合国内外关于外泌体治疗皮肤创伤最新的研究成果和相关综述,对不同来源外泌体在伤口修复不同阶段的作用机制进行总结,并结合当前外泌体的临床数据和临床应用现状提出自己对外泌体未来在皮肤创伤修复研究方向的见解,全面详尽地对外泌体的临床转化进行展望。
3.3 综述的局限性 由于篇幅所限,综述尚未具体讨论改良外泌体在皮肤创伤治疗中的应用和比较各种改良技术的优劣,关于非干细胞源外泌体用于创伤修复的研究仍不够深入,概括比较粗略。
3.4 综述的重要意义 外泌体作为一种新型的无细胞治疗方式,在皮肤再生方面具有广阔的应用前景。文章总结干细胞外泌体在创伤修复各个阶段的作用,详细阐述外泌体通过调节炎症反应、免疫保护、促进血管生成、增强细胞增殖和迁移能力和胶原重塑失衡调节等具体机制促进伤口愈合,帮助学者迅速掌握最新研究成果,为后续研究提供切实可靠的理论依据和实践基础。同时比较干细胞外泌体与非干细胞外泌体的优缺点,全面深入探讨当前外泌体在临床应用的现状和临床转化的痛点与难点,为未来临床转化方向及更广阔的应用研究奠定基础。
3.5 课题专家组对未来的建议 鉴于外泌体的高度异质性,建立标准化的分离、质量控制、纯化及表征技术是很长一段时间需要面临的问题。微流体分离技术作为一种可以和多种外泌体分离方法兼容的高通量技术,其发展为建立标准化的生产程序提供了可能。外泌体的长期储存是建立标准化外泌体生产程序的重要组成部分,冷冻干燥法可以有效保持其生物活性与蛋白含量并极大延长其储存时间,是一种理想的解决方案。大量临床前研究是临床转化的支撑,在未来还需要更多的临床前研究确定对于伤口愈合最合适的细胞来源,进一步发展工程外泌体、外泌体结合生物活性支架等改善外泌体治疗效果的技术,共同探索外泌体在伤口病理生理过程中所起的作用,为未来外泌体的临床转化寻找新的突破口。在未来的临床试验中应聚焦于探索最佳的给药途径和剂量,建立完善的疗效评价系统,验证其安全性,为临床促创面愈合开辟新的路径。 中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
 #br#
#br#
文题释义:
外泌体:是一种直径在40-100 nm的细胞外盘状囊泡。外泌体携带的生物活性物质的类型及功能根据细胞类型的不同而有所不同,主要包括DNA、RNA、蛋白质、脂质、细胞因子等,是细胞间信息交流的重要组成部分。目前外泌体是疾病诊断与治疗、靶向给药、生物疫苗、组织修复再生等领域研究的热点。皮肤创伤修复:是一个复杂的组织修复和重塑过程,涉及炎症反应、细胞增殖和迁移、细胞外基质合成和沉积、血管生成和重塑等生物学进程,需要通过依靠各种类型细胞在时间和空间的紧密协调才能有效恢复受损组织的外观和功能。修复过程受多种内源性及外源性因素影响,若伤口修复相关细胞行为和功能紊乱可导致伤口愈合不良,甚至无法愈合。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
外泌体作为一种新型的无细胞治疗方式,在皮肤再生方面具有广阔的应用前景。本文总结干细胞外泌体创伤修复各个阶段的作用,详细阐述外泌体通过调节炎症反应、免疫保护、促进血管生成、增强细胞增殖和迁移能力和胶原重塑失衡调节等具体机制促进伤口愈合,帮助学者迅速掌握最新研究成果,为后续研究提供切实可靠的理论依据和实践基础。同时比较干细胞外泌体与非干细胞外泌体的优缺点,全面深入探讨当前外泌体在临床应用的现状和临床转化的痛点与难点,为未来临床转化方向及更广阔的应用研究奠定基础。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||