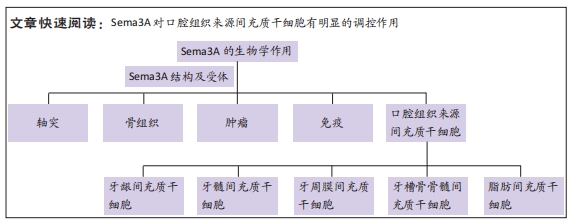
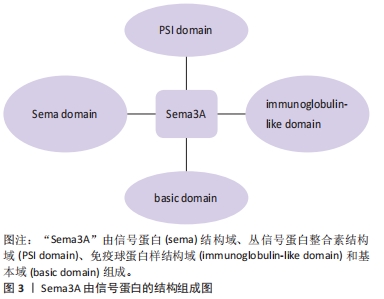
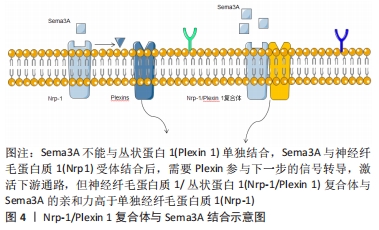
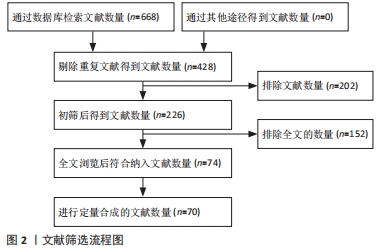
[1] FU X, LIU G, HALIM A, et al. Mesenchymal stem cell migration and tissue repair. Cells. 2019;8(8):784.
[2] ZHOU LL, LIU W, WU YM, et al. Oral mesenchymal stem/progenitor cells: the immunomodulatory masters. Stem Cells Int. 2020;2020: 1327405.
[3] BOTELHO J, CAVACAS MA, MACHADO V, et al. Dental stem cells: recent progresses in tissue engineering and regenerative medicine. Ann Med. 2017;49(8):644-651.
[4] KOLODKIN AL, MATTHES DJ, GOODMAN CS. The semaphorin genes encode a family of transmembrane and secreted growth cone guidance molecules. Cell. 1993;75(7):1389-1399.
[5] FIORE R, PUSCHEL AW. The function of semaphorins during nervous system development. Front Biosci. 2003;8:s484-s499.
[6] GHERARDI E, YOULES ME, MIGUEL RN, et al. Functional map and domain structure of MET, the product of the c-met protooncogene and receptor for hepatocyte growth factor/scatter factor. Proc Natl Acad Sci U S A. 2003;100(21):12039-12044.
[7] KOLODKIN AL, LEVENGOOD DV, ROWE EG, et al. Neuropilin is a semaphorin III receptor. Cell. 1997;90(4):753-762.
[8] ROHM B, OTTEMEYER A, LOHRUM M, et al. Plexin/neuropilin complexes mediate repulsion by the axonal guidance signal semaphorin 3A. Mech Dev. 2000;93(1-2):95-104.
[9] HOTA PK, BUCK M. Plexin structures are coming: opportunities for multilevel investigations of semaphorin guidance receptors, their cell signaling mechanisms, and functions. Cell Mol Life Sci. 2012;69(22): 3765-3805.
[10] LUO Y, RAIBLE D, RAPER JA. Collapsin: a protein in brain that induces the collapse and paralysis of neuronal growth cones. Cell. 1993;75(2): 217-227.
[11] GIGER RJ, WOLFER DP, DE WIT GM, et al. Anatomy of rat semaphorin III/collapsin-1 mRNA expression and relationship to developing nerve tracts during neuroembryogenesis. J Comp Neurol. 1996;375(3):378-392.
[12] GAVAZZI I. Semaphorin-neuropilin-1 interactions in plasticity and regeneration of adult neurons. Cell Tissue Res. 2001;305(2):275-284.
[13] FENSTERMAKER V, CHEN Y, GHOSH A, et al. Regulation of dendritic length and branching by semaphorin 3A. J Neurobiol. 2004;58(3):403-412.
[14] BIRGER A, OTTOLENGHI M, PEREZ L, et al. ALS-related human cortical and motor neurons survival is differentially affected bySema3A. Cell Death Dis. 2018;9(3):256.
[15] BEHAR O, GOLDEN JA, MASHIMO H, et al. Semaphorin III is needed for normal patterning and growth of nerves, bones and heart. Nature. 1996;383(6600):525-528.
[16] YU X, ZHENG F, DU Y, et al. Semaphorin 3A gets involved in the establishment of mouse tooth eruptive pathway. J Mol Histol. 2019; 50(5):427-434.
[17] TERMINI CM, PANG A, FANG T, et al. Neuropilin 1 regulates bone marrow vascular regeneration and hematopoietic reconstitution. Nat Commun. 2021;12(1):6990.
[18] ZHANG N, HUA Y, LI Y, et al. Sema3Aaccelerates bone formation during distraction osteogenesis in mice. Connect Tissue Res. 2021:1-11.
[19] LUCARINI G, SIMONETTI O, LAZZARINI R, et al. Vascular endothelial growth factor/semaphorin-3A ratio andSema3Aexpression in cutaneous malignant melanoma. Melanoma Res. 2020;30(5):433-442.
[20] HUANG C, WANG Y, HUANG JH, et al. Sema3A drastically suppresses tumor growth in oral cancer Xenograft model of mice. BMC Pharmacol Toxicol. 2017;18(1):55.
[21] TIAN T, ZHANG L, TANG K, et al. Sema3A Exon 9 expression is a potential prognostic marker of unfavorable recurrence-free survival in patients with tongue squamous cell carcinoma. DNA Cell Biol. 2020;39(4):555-562.
[22] BEJAR I, RUBINSTEIN J, BEJAR J, et al. Expression of semaphorin 3A in malignant and normal bladder tissue: immunohistochemistry staining and morphometric evaluation. Biology (Basel). 2021;10(2):109.
[23] IMOTO T, KONDO S, WAKISAKA N, et al. Overexpression of semaphorin 3A is a marker associated with poor prognosis in patients with nasopharyngeal carcinoma. Microorganisms. 2020;8(3):423.
[24] YAMADA D, TAKAHASHI K, KAWAHARA K, et al. Autocrine Semaphorin3A signaling is essential for the maintenance of stem-like cells in lung cancer. Biochem Biophys Res Commun. 2016;480(3):375-379.
[25] AVOUAC J, PEZET S, VANDEBEUQUE E, et al. Semaphorins: from angiogenesis to inflammation in rheumatoid arthritis. Arthritis Rheumatol. 2021;73(9):1579-1588.
[26] CATALANO A, CAPRARI P, MORETTI S, et al. Semaphorin-3A is expressed by tumor cells and alters T-cell signal transduction and function. Blood. 2006;107(8):3321-3329.
[27] GARCIA F, LEPELLETIER Y, SMANIOTTO S, et al. Inhibitory effect of semaphorin-3A, a known axon guidance molecule, in the human thymocyte migration induced by CXCL12. J Leukoc Biol. 2012;91(1):7-13.
[28] VADASZ Z, HAJ T, TOUBI E. The role of B regulatory cells and Semaphorin3A in atopic diseases. Int Arch Allergy Immunol. 2014; 163(4):245-251.
[29] RIENKS M, CARAI P, BITSCH N, et al. Sema3Apromotes the resolution of cardiac inflammation after myocardial infarction. Basic Res Cardiol. 2017;112(4):42.
[30] ADI SD, EIZA N, BEJAR J, et al. Semaphorin 3A is effective in reducing both inflammation and angiogenesis in a mouse model of bronchial asthma. Front Immunol. 2019;10:550.
[31] CASE N, RUBIN J. Beta-catenin--a supporting role in the skeleton. J Cell Biochem. 2010;110(3):545-553.
[32] EL-SAYED KM, PARIS S, GRAETZ C, et al. Isolation and characterisation of human gingival margin-derived STRO-1/MACS(+) and MACS(-) cell populations. Int J Oral Sci. 2015;7(2):80-88.
[33] HONG R, WANG Z, SUI A, et al. Gingival mesenchymal stem cells attenuate pro-inflammatory macrophages stimulated with oxidized low-density lipoprotein and modulate lipid metabolism. Arch Oral Biol. 2019;98:92-98.
[34] ZHANG F, SI M, WANG H, et al. IL-1/TNF-alpha inflammatory and anti-inflammatory synchronization affects gingival stem/progenitor cells’ regenerative attributes. Stem Cells Int. 2017;2017:1349481.
[35] FAWZY EL-SAYED KM, NGUYEN N, DöRFER CE. Ascorbic acid, inflammatory cytokines (IL-1β/TNF-α/IFN-γ), or their combination’s effect on stemness, proliferation, and differentiation of gingival mesenchymal stem/progenitor cells. Stem Cells Int. 2020;2020: 8897138.
[36] GRONTHOS S, MANKANI M, BRAHIM J, et al. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A. 2000;97(25):13625-13630.
[37] NUTI N, CORALLO C, CHAN BM, et al. Multipotent differentiation of human dental pulp stem cells: a literature review. Stem Cell Rev Rep. 2016;12(5):511-523.
[38] TAKEDA T, TEZUKA Y, HORIUCHI M, et al. Characterization of dental pulp stem cells of human tooth germs. J Dent Res. 2008;87(7):676-681.
[39] TOMASELLO L, MAUCERI R, COPPOLA A, et al. Mesenchymal stem cells derived from inflamed dental pulpal and gingival tissue: a potential application for bone formation. Stem Cell Res Ther. 2017; 8(1):179.
[40] CARNES DL, MAEDER CL, GRAVES DT. Cells with osteoblastic phenotypes can be explanted from human gingiva and periodontal ligament. J Periodontol. 1997;68(7):701-707.
[41] SEO BM, MIURA M, GRONTHOS S, et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004; 364(9429):149-155.
[42] IWASAKI K, KOMAKI M, YOKOYAMA N, et al. Periodontal ligament stem cells possess the characteristics of pericytes. J Periodontol. 2013; 84(10):1425-1433.
[43] LEE JS, AN SY, KWON IK, et al. Transdifferentiation of human periodontal ligament stem cells into pancreatic cell lineage. Cell Biochem Funct. 2014;32(7):605-611.
[44] TOMOKIYO A, WADA N, MAEDA H. Periodontal ligament stem cells: regenerative potency in periodontium. Stem Cells Dev. 2019;28(15): 974-985.
[45] ELEUTERIO E, TRUBIANI O, SULPIZIO M, et al. Proteome of human stem cells from periodontal ligament and dental pulp. PLoS One. 2013; 8(8):e71101.
[46] HASEGAWA D, HASEGAWA K, KANEKO H, et al. MEST regulates the stemness of human periodontal ligament stem cells. Stem Cells Int. 2020;2020:9672673.
[47] LI Y, YANG F, GAO M, et al. miR-149-3p regulates the switch between adipogenic and osteogenic differentiation of BMSCs by targeting FTO. Mol Ther Nucleic Acids. 2019;17:590-600.
[48] WAGNER W, HORN P, CASTOLDI M, et al. Replicative senescence of mesenchymal stem cells: a continuous and organized process. PLoS One. 2008;3(5):e2213.
[49] HUANG X, CHENG B, SONG W, et al. Superior CKIP-1 sensitivity of orofacial bone-derived mesenchymal stem cells in proliferation and osteogenic differentiation compared to long bone-derived mesenchymal stem cells. Mol Med Rep. 2020;22(2):1169-1178.
[50] ZUK PA, ZHU M, MIZUNO H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001; 7(2):211-228.
[51] CHU DT, NGUYEN THI PHUONG T, TIEN NLB, et al. Adipose tissue stem cells for therapy: an update on the progress of isolation, culture, storage, and clinical application. J Clin Med. 2019;8(7):917.
[52] STORTI G, SCIOLI MG, KIM BS, et al. Adipose-derived stem cells in bone tissue engineering: useful tools with new applications. Stem Cells Int. 2019;2019:3673857.
[53] GAUR S, AGNIHOTRI R. Application of adipose tissue stem cells in regenerative dentistry: a systematic review. J Int Soc Prev Community Dent. 2021;11(3):266-271.
[54] KANG BJ, RYU HH, PARK SS, et al. Comparing the osteogenic potential of canine mesenchymal stem cells derived from adipose tissues, bone marrow, umbilical cord blood, and Wharton’s jelly for treating bone defects. J Vet Sci. 2012;13(3):299-310.
[55] TIAN T, TANG K, WANG A, et al. The effects of Sema3Aoverexpression on the proliferation and differentiation of rat gingival mesenchymal stem cells in the LPS-induced inflammatory environment. Int J Clin Exp Pathol. 2019;12(10):3710-3718.
[56] SONG Y, LIU X, FENG X, et al. NRP1 accelerates odontoblast differentiation of dental pulp stem cells through classical Wnt/β-catenin signaling. Cell Reprogram. 2017;19(5):324-330.
[57] YOSHIDA S, WADA N, HASEGAWA D, et al. Semaphorin 3A induces odontoblastic phenotype in dental pulp stem cells. J Dent Res. 2016; 95(11):1282-1290.
[58] LIN Y, XING Q, QIN W, et al. Decreased expression of Semaphorin3A/Neuropilin-1 signaling axis in apical periodontitis. Biomed Res Int. 2017; 2017:8724503.
[59] MENICANIN D, BARTOLD PM, ZANNETTINO AC, et al. Identification of a common gene expression signature associated with immature clonal mesenchymal cell populations derived from bone marrow and dental tissues. Stem Cells Dev. 2010;19(10):1501-1510.
[60] WADA N, MAEDA H, HASEGAWA D, et al. Semaphorin 3A induces mesenchymal-stem-like properties in human periodontal ligament cells. Stem Cells Dev. 2014;23(18):2225-2236.
[61] CHANG X, ZHOU F, BU L, et al. Semaphorin 3A attenuates the hypoxia suppression of osteogenesis in periodontal ligament stem cells. J Periodontal Res. 2022;57(2):425-433.
[62] 姜朝阳,谢兴文,徐世红,等.骨髓间充质干细胞成骨分化相关信号通路[J].中华骨质疏松和骨矿盐疾病杂志,2020,13(5):473-478.
[63] QIAO Q, XU X, SONG Y, et al. Semaphorin 3A promotes osteogenic differentiation of BMSC from type 2 diabetes mellitus rats. J Mol Histol. 2018;49(4):369-376.
[64] LIU L, WANG J, SONG X, et al. Semaphorin 3A promotes osteogenic differentiation in human alveolar bone marrow mesenchymal stem cells. Exp Ther Med. 2018;15(4):3489-3494.
[65] SUN Z, YAN K, LIU S, et al. Semaphorin 3A promotes the osteogenic differentiation of rat bone marrow-derived mesenchymal stem cells in inflammatory environments by suppressing the Wnt/β-catenin signaling pathway. J Mol Histol. 2021;52(6):1245-1255.
[66] LIU X, TAN N, ZHOU Y, et al. Semaphorin 3A shifts adipose mesenchymal stem cells towards osteogenic phenotype and promotes bone regeneration in vivo. Stem Cells Int. 2016;2016:2545214.
[67] FANG K, SONG W, WANG L, et al. Semaphorin 3A-modified adipose-derived stem cell sheet may improve osseointegration in a type 2 diabetes mellitus rat model. Mol Med Rep. 2016;14(3):2449-2456.
[68] FUKUDA T, TAKEDA S, XU R, et al. Sema3A regulates bone-mass accrual through sensory innervations. Nature. 2013;497(7450):490-493.
[69] HAYASHI M, NAKASHIMA T, YOSHIMURA N, et al. Autoregulation of osteocyte Sema3A orchestrates estrogen action and counteracts bone aging. Cell Metab. 2019;29(3):627-637.e625.
[70] ŞEN S, LUX CJ, ERBER R. A Potential Role of Semaphorin 3A during Orthodontic Tooth Movement. Int J Mol Sci. 2021;22(15):8297.
|