Design
Comparative observation of the experiment.
Time and setting
Between April 2014 and November 2014, completed in Yunnan Province Key Laboratory of Lung Cancer Research
Materials
Staphylococcus epidermidis reference stain RP62A (ATCC35984, biofilm-positive strains), purchased from Culture Collection Center, Guangdong Institute of Microbiology; PVC (Guangdong Province Dongguan Kewei Medical Instrument Co., Ltd., Lot No. 20050903);Furanone 1: 3,4-dibromo-5-hydroxyl-furanone (Fluka Company); Furanone 2: 4-bromo-5-(4-methoxypheny)-3- (methylamino)-furanone (Aldrich company); Furanone 3: 3,4-dibromo-5,5-dimethoxypheny-2 (5H)-furanone (Aldrich company); Live/Dead® Baclight™ Bacterial Viability Kit (Invitrogen); MRC-1 024ES type confocal laser scanning microscope (CLSM, BIO-RAD); XL30ESEM type scanning electron microscope (SEM, PHILIPS)
Methods
Preparation of bacterial suspension
SE frozen stock was inoculated in sterilized LB broth and incubated for 16 hours at 37 ℃ and then plated on LB plate and incubated for 24 hours (37 ℃) to obtain colonies. Single colony was selected and inoculated in LB broth for 12-16 hours (37 ℃) to obtain SE suspension. The concentration of SE in the suspension was adjusted to 1×105 CFU/mL for the experiment by pour plate method.
Modification of PVC surface and experimental groups
There were four groups of treatment: control, furanone 1, furanone 2, and furanone 3 groups. PVC was squared into 1×1 cm2 small pieces by the customized simulator and sterilized by fumigation with ethylene oxide. Brominated furanones, which are insoluble in water, were solubilized in ethanol and made up to 1 mg/mL solution. In furanone 1-3 groups, PVC was dipped in the corresponding ethanolic solution of furanone for 5 minutes, in order to modify the surface of PVC piece. Then the PVC piece was taken out of the solution and air-dried. In control group, PVC piece was dipped in 75% ethanol solution for 5 minutes.
Formation of bacterial biofilm on the surface of PVC
In five sterile flasks in each group, 20 mL LB broth, 5 μL bacterial suspension and four corresponding PVC pieces were placed and the flasks were incubated at 37 ℃. PVC pieces were taken out one after another, following incubations for 6, 12, 18 and 24 hours (four pieces for the observation by CLSM, another piece for the observation by SEM).
Observation of CLSM
According to manufacturer’s instructions, the fluorescent stains for bacterial viability on the BF was identified using Live/Dead® Baclight™ Bacterial Viability Kit; PVC pieces which taken out of culture flasks were washed 3 times with distilled water and put into the fluorescent stains, stained for 20 minutes at room temperature. CLSM observation was conducted with argon laser (514/488 nm), one visual field was randomly selected to observe the quantity of bacterial colony, 10×10 per field for each PVC piece observed. One bacterial biofilm captured randomly in each visual field was scanned from internal to external to measure its thickness.
Observation of SEM
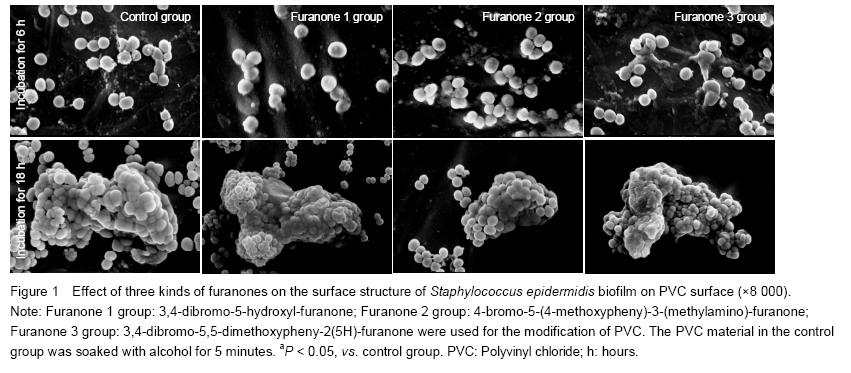
The PVC pieces were washed for three times by HEPES buffer, pH=7, fixed on the object stage of SEM, dried in the critical CO2. The surface of PVC turned to the golden color due to the ions sputtering surface fixture deposition. The surface structure of bacterial biofilm was observed.
Main outcome measures
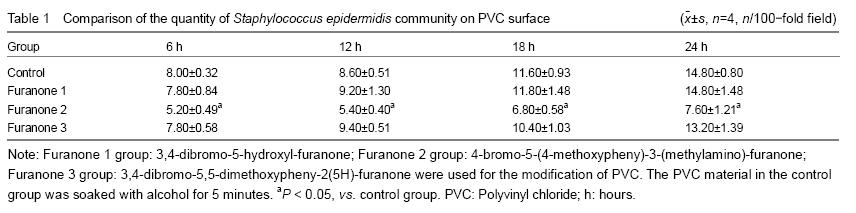
After the surface coating modification and co-cultivation with three kinds of brominated furanones with representative chemical structure, the thickness of bacterium BF, bacterium community quantity unit area and surface structure of biofilm formation of SE on the surface of PVC materials were observed.
Statistical analysis
The data was analyzed by SPSS12.0 software; all the values were expressed as mean SD; the data were analyzed by repeated analysis of variance, P < 0.05 was defined statistically significant.
.jpg)