Design
A randomized controlled animal experiment.
Time and setting
This experiment was completed at the Stem Cell and Animal Laboratory of Xinjiang Medical University, China from March to September in 2014.
Materials
Twenty-seven New Zealand white rabbits, 2 months old, weighing 2.7-3.2 kg, were provided by the Experimental Animal Center of the First Affiliated Hospital to Xinjiang Medical University (license No. IACUC-20140127004).
Methods
PRF preparation
10 mL blood sample was collected from the ear vein of each rabbit. Then, the blood sample was centrifuged for 15 minutes at 3 000 r/min. After centrifugation, the blood sample was divided into three layers and the middle layer was PRF.
Isolation, cultivation and identification of autologous BMSCs in vitro
Under aseptic conditions, 5 mL bone marrow was taken from the bilateral femurs of rabbits and then rinsed with DMED solution (Thermo Fisher Biochemical Products Co., Ltd., Beijing, China) and centrifuged at 1 500 r/min for 5 minutes followed by DMEM culture (10% fetal bovine serum, 100 U/mL penicillin, streptomycin 100 mg/mL) in 37 ℃, 5% CO2 incubation under saturated humidity conditions. After 48 hours, the medium was exchanged first, the cell suspension was discarded. Then, the culture medium was changed every 2 days. Cell growth and morphology were observed using inverted microscope (Leica Microsystems IR GmbH, Germany). The cells were subcultured and expanded at the proportion of 1:2. In this study, passages 2-3 cells were used.
In vitro culture of autologous BMSCs with PRF
Passages 2-3 BMSCs were labeled with 4,6-acetyl 2-2-phenyl indole (0.02 g/L; Santa Cruz Biotech, Dallas, Texas, USA) overnight, washed with 10×PBS, digested and resuspended. BMSCs and PRF were co-cultured at an identical density of 6×107/mL cells onto 24-well plates. Nutrient solution (2 mL) was added and incubated at 37 ℃, 5% CO2. Then, the medium was changed every 48 hours. BMSCs and PRF were mixed in culture bottles and observed under electron microscopy (Leica Microsystems IR GmbH).
Modeling and intervention
Twenty-seven rabbits were randomly divided into three groups: BMSCs/PRF group, PRF group and model group (n=9 per group). Under general anesthesia induced by 3% pentobarbital sodium injection (1 mL/kg), the left mandibular incisor was extracted aseptically from each rabbit. Width and height of the alveolar socket were measured. Adjacent incisor was cut as reference plane. The BMSCs/PRF group was implanted with autologous BMSCs (1.2×106, 20 μL) and PRF (4 mm× 6 mm). The PRF group was filled with PRF(4 mm×6 mm) and the model group filled with nothing. The wound was closed by suture with 4-0 silk. After surgery, each rabbit was injected gentamicin sulfate (1 mg/kg, Anyang, Henan, China), three times a day, totally for 3 days. Each rabbit was caged individually and had free access to food and water. Three animals of each group were killed under overdose 3% pentobarbital anesthesia at 4, 8 and 12 weeks.
Morphological observation
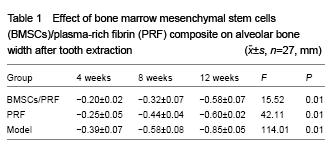
The width and height of the alveolar bone at extracted sites were measured.
Radiographic observation
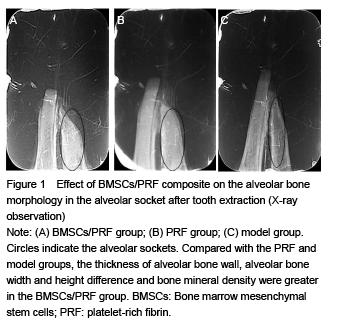
Rabbit's mandible was fixed in 10% formalin as specimens and underwent computerized tomography. Alveolar bone density was observed after X-ray imaging.
Histological evaluation
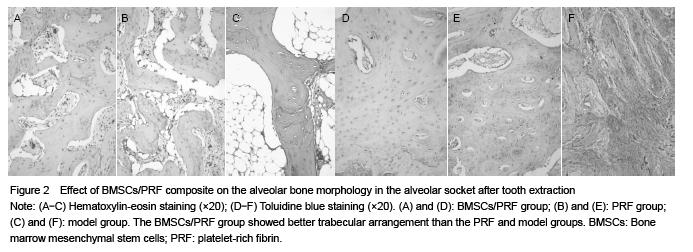
The specimen was observed under inverted microscope after decalcification, embedding, sectioning. Hematoxylin-eosin and toluidine blue staining were used to observe osteoblast and trabecular structure.
Main outcome measures
The alveolar bone width, height and mineral density were measured. Osteoblast and trabecular structure were observed.
Statistical analysis
All data are expressed as mean±SD. Intergroup comparison was determined by analysis of variance, followed by Fisher post hoc test and paired Student t-test, when appropriate. A value of P < 0.01 was regarded as statistically significant.
.jpg)
.jpg)