中国组织工程研究 ›› 2015, Vol. 19 ›› Issue (21): 3302-3308.doi: 10.3969/j.issn.2095-4344.2015.21.005
• 膜生物材料 membrane biomaterials • 上一篇 下一篇
胶原海绵人工硬脑膜结合臭氧修复颅脑损伤
曲 虹,赵明光,梁 英,赵丽萍,李晓红,王 莹
- 解放军沈阳军区总医院神经外科,辽宁省沈阳市 110016
-
出版日期:2015-05-21发布日期:2015-05-21 -
通讯作者:赵明光,博士,副主任医师,解放军沈阳军区总医院神经外科,辽宁省沈阳市 110016 -
作者简介:曲虹,女,1971年生,山东省黄县人,汉族,2011年解放军第四军医大学毕业,硕士,主要从事临床神经外科方面的研究。 -
基金资助:辽宁省科技攻关项目(2012225019)
Collagen sponge as an artificial dura mater combined with ozone to repair brain injury
Qu Hong, Zhao Ming-guang, Liang Ying, Zhao Li-ping, Li Xiao-hong, Wang Ying
- Department of Neurology, General Hospital of Shenyang Military Region, Shenyang 110016, Liaoning Province, China
-
Online:2015-05-21Published:2015-05-21 -
Contact:Corresponding author: Zhao Guang-ming, M.D., Associate chief physician, Department of Neurology, General Hospital of Shenyang Military Region, Shenyang 110016, Liaoning Province, China -
About author:Qu Hong, Master, Department of Neurology, General Hospital of Shenyang Military Region, Shenyang 110016, Liaoning Province, China -
Supported by:Liaoning Science and Technology Project, No. 2012225019
摘要:
背景:硬膜的完整性对于颅脑损伤患者手术的预后十分重要。人工硬脑膜是目前常见的硬脑膜修补材料,寻找理想的人工硬脑膜是神经外科探索的方向。 目的:观察并分析胶原海绵人工硬脑膜和臭氧治疗颅脑损伤患者的临床资料,探讨并评价其使用价值。 方法:回顾性分析60例颅脑损伤患者应用胶原海绵人工硬脑膜修补和臭氧治疗后的疗效及并发症随访结果。 结果与结论:患者中2例死于重型颅脑损伤术后弥漫性脑肿胀,1例死于脑损伤合并多器官衰竭,2例因广泛脑挫裂伤合并脑疝术后植物生存,其余55例患者进入结果分析。术后2例出现切口局部皮下积液,予穿刺抽吸及弹性绷带加压包扎后好转;1例术后出现术侧少量硬膜下积液,未行特殊处理,动态复查头颅CT显示积液逐步吸收减少。头颅CT 检查未见与人工脑膜有关的异常反应和表现。28例患者3-6个月进行颅骨修补时发现人工硬脑膜与正常硬脑膜融合恢复较好,无粘连及炎症反应发生。表明胶原海绵人工硬脑膜和臭氧在颅脑损伤患者治疗中充分发挥减压作用,维持脑功能,缩短手术时间,并发症少,相容性好,能够较好的与正常硬脑膜融合,保护脑皮质,为后期颅骨修补创造有利条件。
中图分类号:
引用本文
曲 虹,赵明光,梁 英,赵丽萍,李晓红,王 莹. 胶原海绵人工硬脑膜结合臭氧修复颅脑损伤[J]. 中国组织工程研究, 2015, 19(21): 3302-3308.
Qu Hong, Zhao Ming-guang, Liang Ying, Zhao Li-ping, Li Xiao-hong, Wang Ying. Collagen sponge as an artificial dura mater combined with ozone to repair brain injury[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(21): 3302-3308.
Two cases died of severe brain injury after diffuse brain swelling, one died of brain injury with multiple organ failure, and two cases of extensive brain injury accompanied by cerebral herniation were in vegetative state. Patients were followed for 1 year, and the remaining 55 patients were enrolled in the final analysis (Table 1).
There were no cerebrospinal fluid leakage from the incision, subcutaneous fluid, intracranial infections, epilepsy and other complications, and skin incision healed well in all patients followed by regular removal of stitches. Before discharge, head CT examinations showed no obvious abnormalities at the repair site. Patients recovered well postoperatively with normal body temperature and normal results of blood and biochemical tests (Table 1). Of the 60 patients, 2 patients died of postoperative diffuse brain swelling, one died of brain injury with multiple organ failures, and two cases of extensive brain injury accompanied by cerebral herniation were in vegetative state after surgery. Another 55 cases showed obvious midline shift on CT images at 1, 7, 14 days after surgery (P < 0.05). There was also a significant difference in the intracranial pressure at 1, 7, 14 days after surgery (P < 0.05; Table 2), indicating the treatment achieved the purpose of decompression.

Head CT results
Head CT results showed that there were no abnormal radiographic changes at 1 week after surgery and at discharge (Figure 1).
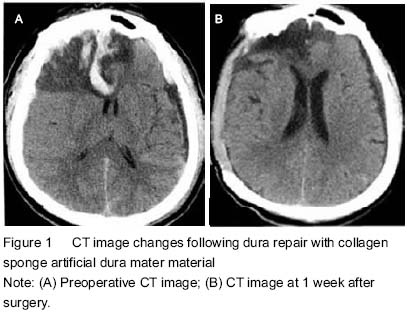
Follow-up results
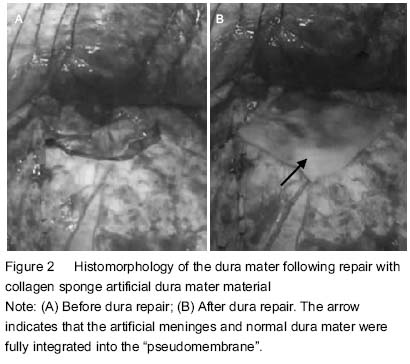
According to Glasgow coma scale score, complete recovery was in 12 cases (20%), mild disability in 16 cases (27%), moderate disability in 21 cases (35%), severe disability in 7 cases (12%), vegetative state in 2 cases (3%), and death in 2 cases (3%). At 3-6 months after dura repair, 28 cases were subject to skull repair, and during the skull repair, the anatomical structures of the dura mater were found basically clear and less wound bleeding. There was shorter operative time. The dura mater separated was intact with no damage to brain tissues, and the artificial meninges and normal dura mater were fully integrated into the “pseudomembrane” that had a complete smooth surface and medial surface. No meningeal adhesions and severe inflammatory reactions in the repair area were found (Figure 2).
After surgery, two patients appeared with postoperative subcutaneous fluid and their conditions improved following puncture aspiration and pressure dressing with elastic bandage; another patient showed a small amount of subdural effusion, but with no special treatment, and dynamic head CT showed the effusion was gradually reduced.
During the follow-up, there was no advanced incision leakage of cerebrospinal fluid, brain incarceration and epilepsy. All skin incisions were healed at stage I, with regular removal of stitches. Repair materials had no response to the host in the blood, tissue, and immunological fields.

| [1] Tsuji M, Inoue Y, Sugaya C, et al. Higher toxicity of dibutyltin and poly-L-lactide with a large amount of tin but lower toxicity of poly-L-lactide of synthetic artificial dura mater exhibited on murine astrocyte cell line. Yakugaku Zasshi. 2010;130(6):847-855. [2] Zhang YL, Xin GH, Zeng YL. Pig peritonium as a donor transplant to repair the defect in human cerebral dura mater. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2008;12(23):4489-4492. [3] Chen J, Shi SS, Zhang GL, et al. Bio-artificial dura mater in repair of dural defects. Zhongguo Zuzhi Gongheng Yanjiu. 2013;17(51):8914-8919. [4] Esposito F, Grimod G, Cavallo LM, et al. Collagen-only biomatrix as dural substitute: What happened after a 5-year observational follow-up study. Clin Neurol Neurosurg. 2013;115(9):1735-1737. [5] Parlato C, di Nuzzo G, Luongo M, et al. Use of a collagen biomatrix (TissuDura) for dura repair: a long-term neuroradiological and neuropathological evaluation. Acta Neurochir (Wien). 2011;153(1):142-147. [6] Papakostas JC, Avgos S, Arnaoutoglou E, et al. Use of the vascu-guard bovine pericardium patch for arteriotomy closure in carotid endarterectomy. Early and long-term results. Ann Vasc Surg. 2014;28(5):1213-1218. [7] Coleman S, Kerr H, Krishnamurthi V, et al. The use of bovine pericardium for complex urologic venous reconstruction. Urology. 2014;83(2):495-497. [8] Costa BS, Cavalcanti-Mendes Gde A, Abreu MS, et al. Clinical experience with a novel bovine collagen dura mater substitute. Arq Neuropsiquiatr. 2011;69(2A):217-220. [9] Chen X, Liu J, Yang GY.The clinical application of collagen dressing. Linchuang yu Shiyan Yixue Zazhi. 2010;17(23):1078-1079. [10] Sartori C, Lepori M, Scherrer U. Interaction between nitric oxide and the cholinergic and sympathetic nervous system in cardiovascular control in humans. Pharmacol Ther. 2005;106(2):209-220. [11] Bocci V, Paulesu L. Studies on the biological effects of ozone 1. Induction of interferon gamma on human leucocytes. Haematologica. 1990;75(6):510-515. [12] Larini A, Bocci V. Effects of ozone on isolated peripheral blood mononuclear cells. Toxicol In Vitro. 2005;19(1): 55-61. [13] Zhou NB, Fu ZJ, Sun T. Effects of different concentrations of oxygen-ozone on rats' astrocytes in vitro. Neurosci Lett. 2008;441(2):178-182. [14] Parízek J, Husek Z, M?ricka P, et al. Ovine pericardium: a new material for duraplasty. J Neurosurg. 1996;84(3): 508-513. [15] Chen YG, Zhao MM, Ning TY, et al. The application of artificial dura in emergency craniocerebral surgery. Hebei Yiyao. 2010;16(6):688-689. [16] Roberts I, Sydenham E. Barbiturates for acute traumatic brain injury. Cochrane Database Syst Rev. 2012;12: CD000033. [17] Timofeev I, Carpenter KL, Nortje J, et al. Cerebral extracellular chemistry and outcome following traumatic brain injury: a microdialysis study of 223 patients. Brain. 2011;134(Pt 2):484-494. [18] Wakai A, McCabe A, Roberts I, et al. Mannitol for acute traumatic brain injury. Cochrane Database Syst Rev. 2013;8:CD001049. [19] Tong WS, Zheng P, Xu JF, et al. Early CT signs of progressive hemorrhagic injury following acute traumatic brain injury. Neuroradiology. 2011;53(5):305-309. [20] Walcott BP, Kahle KT, Simard JM. The DECRA trial and decompressive craniectomy in diffuse traumatic brain injury: is decompression really ineffective? World Neurosurg. 2013;79(1):80-81. [21] Bor-Seng-Shu E, Figueiredo EG, Fonoff ET, et al. Decompressive craniectomy and head injury: brain morphometry, ICP, cerebral hemodynamics, cerebral microvascular reactivity, and neurochemistry. Neurosurg Rev. 2013;36(3):361-370. [22] Timofeev I, Santarius T, Kolias AG, et al. Decompressive craniectomy - operative technique and perioperative care. Adv Tech Stand Neurosurg. 2012;38:115-136. [23] Wen L, Li QC, Wang SC, et al. Contralateral haematoma secondary to decompressive craniectomy performed for severe head trauma: a descriptive study of 15 cases. Brain Inj. 2013;27(3):286-292. [24] Sakas DE, Charnvises K, Borges LF, et al. Biologically inert synthetic dural substitutes. Appraisal of a medical-grade aliphatic polyurethane and a polysiloxane-carbonate block copolymer. J Neurosurg. 1990;73(6):936-941. [25] Andreula CF, Simonetti L, De Santis F, et al. Minimally invasive oxygen-ozone therapy for lumbar disk herniation. AJNR Am J Neuroradiol. 2003;24(5):996-1000. [26] Bocci V. Ozone as a bioregulator. Pharmacology and toxicology of ozonetherapy today. J Biol Regul Homeost Agents. 1996;10(2-3):31-53. [27] Leone S, Noera G, Bertolini A. Melanocortins as innovative drugs for ischemic diseases and neurodegenerative disorders: established data and perspectives. Curr Med Chem. 2013;20(6):735-750. [28] Dzieran J, Fabian J, Feng T, et al. Comparative analysis of TGF-β/Smad signaling dependent cytostasis in human hepatocellular carcinoma cell lines. PLoS One. 2013;8(8): e72252. [29] Timmermann J, Hahn M, Krueger K. Short-term follow-up: micro-invasive therapy of the cervical herniated disk by percutaneous nucleotomy. J Back Musculoskelet Rehabil. 2011;24(2):89-93. [30] Steppan J, Meaders T, Muto M, et al. A metaanalysis of the effectiveness and safety of ozone treatments for herniated lumbar discs. J Vasc Interv Radiol. 2010;21(4):534-548. [31] Zhang Y, Ma Y, Jiang J, et al. Treatment of the lumbar disc herniation with intradiscal and intraforaminal injection of oxygen-ozone. J Back Musculoskelet Rehabil. 2013;26(3):317-322. [32] Zhou JQ, Chen H, Chen ZY, et al. Effects of ozone oxidative preconditioning on apoptosis induced by renal ischemia/reperfusion injury in rats. Zhonghua Qiguan Yizhi Zazhi. 2012;33(2):113-117. [33] Xin ZG, Liu B, Gao Y, et al. Small doses of triamcinolone aacetonide for heel pain. Zhongguo Xiandai Yisheng. 2011;49(1):130-131. [34] Zeng Y, Xiong M, Yu HL, et al. Efficacy comparison of medical ozone with sodium hyaluronate in the treatment of knee osteoarthritis. Linchuang Waike Zazhi. 2010;18(8):565-567. [35] Gautam S, Rastogi V, Jain A, et al. Comparative evaluation of oxygen-ozone therapy and combined use of oxygen-ozone therapy with percutaneous intradiscal radiofrequency thermocoagulation for the treatment of lumbar disc herniation. Pain Pract. 2011;11(2):160-166. [36] Zamora ZB, Borrego A, López OY, et al. Effects of ozone oxidative preconditioning on TNF-alpha release and antioxidant-prooxidant intracellular balance in mice during endotoxic shock. Mediators Inflamm. 2005;2005(1):16-22. [37] Lehnert T, Naguib NN, Wutzler S, et al. Analysis of disk volume before and after CT-guided intradiscal and periganglionic ozone-oxygen injection for the treatment of lumbar disk herniation. J Vasc Interv Radiol. 2012;23(11): 1430-1436. [38] Torossian A, Ruehlmann S, Eberhart L, et al. Pre-treatment with ozonized oxygen (O3) aggravates inflammation in septic rats. Inflamm Res. 2004;53 Suppl 2:S122-125. |
| [1] | 周继辉, 李新志, 周 游, 黄 卫, 陈文瑶. 髌骨骨折修复内植物选择的多重问题[J]. 中国组织工程研究, 2021, 25(9): 1440-1445. |
| [2] | 李 黎, 马 力. 磁性壳聚糖微球固定化乳糖酶及其酶学性质[J]. 中国组织工程研究, 2021, 25(4): 576-581. |
| [3] | 向飞帆, 叶俊武, 张喜海, 葛建华, 唐 炼, 阳运康. 股骨干合并同侧股骨颈骨折3种内固定方式的比较[J]. 中国组织工程研究, 2021, 25(3): 403-408. |
| [4] | 周安琪, 唐渝菲, 吴秉峰, 向 琳. 骨膜组织工程设计:共性与个性的结合[J]. 中国组织工程研究, 2021, 25(22): 3551-3557. |
| [5] | 郎丽敏, 何 生, 姜增誉, 胡奕奕, 张智星, 梁敏茜. 导电复合材料在心肌梗死组织工程治疗领域的应用进展[J]. 中国组织工程研究, 2021, 25(22): 3584-3590. |
| [6] | 刘 鋆, 杨 龙, 王伟宇, 周玉虎, 吴 颖, 卢 涛, 舒莉萍, 马敏先, 叶 川. 聚3-羟基丁酸酯4-羟基丁酸酯/聚乙二醇/氧化石墨烯组织工程支架的制备和性能评价[J]. 中国组织工程研究, 2021, 25(22): 3466-3472. |
| [7] | 付攀峰, 商 炜, 亢 哲, 邓 宇, 祝少博 . 全髋关节置换中前外侧微创入路与后外侧入路疗效差异的Meta分析[J]. 中国组织工程研究, 2021, 25(21): 3409-3415. |
| [8] | 任兴宇, 张 一, 徐浩然, 范 宾, 戴士峰, 梁春雨. 机器人辅助单髁置换与传统置换效果比较的Meta分析[J]. 中国组织工程研究, 2021, 25(21): 3416-3422. |
| [9] | 李海峰, 刘 宇, 殷渠东, 孙振中, 芮永军, 顾三军. 股骨颈关节内骨折内固定后早期负重的并发症风险:2年随访[J]. 中国组织工程研究, 2021, 25(18): 2875-2880. |
| [10] | 袁家钦, 栾富钧, 陈杨帆, 邓 怡, 李 波. 双极与单极半髋置换修复老年移位型股骨颈骨折的Meta分析[J]. 中国组织工程研究, 2021, 25(18): 2915-2922. |
| [11] | 李尚志, 郑得志, 刘 军. 加速康复外科模式下鸡尾酒疗法对全膝关节置换后的早期镇痛[J]. 中国组织工程研究, 2021, 25(18): 2794-2798. |
| [12] | 宋凯凯, 杜刚强, 李 朋, 蒋昇源, 宫智浩, 张志伟, 张 锴. 头颈比法在人工股骨头置换治疗老年股骨颈骨折中的应用[J]. 中国组织工程研究, 2021, 25(18): 2805-2809. |
| [13] | 解 健, 苏俭生. 静电纺丝取向纳米纤维作为组织工程生物支架的优势与特征[J]. 中国组织工程研究, 2021, 25(16): 2575-2581. |
| [14] | 纪 琦, 喻正文, 张 剑. 3D打印金属基生物材料工艺和临床应用的问题与趋势[J]. 中国组织工程研究, 2021, 25(16): 2597-2604. |
| [15] | 高丰禾, 陈桐莹, 林杰彬, 梁祖建. 氨甲环酸联合利伐沙班在原发性全膝及全髋关节置换中的有效性和安全性:一项Meta分析[J]. 中国组织工程研究, 2021, 25(15): 2453-2460. |
have indicating it has good histocompatibility[9-10]. Ozone efficacy in the treatment of brain trauma and cerebrovascular diseases has been gradually recognized by the medical mobile features[11-13].
A retrospective case analysis.
The case analysis was completed at the Department of Neurosurgery, General Hospital of Shenyang Military Region from June 2012 to June 2014.
Subjects
A retrospective analysis was done in patients with brain injury undergoing dura mater repair.
Materials
Collagen sponge artificial dura mater was selected and used intraoperatively according to dural defect size and shape, which stuck to the dural defect and was fixed with biomedical glue without sutures. Collagen sponge artificial dura mater was purchased from Beijing TianXinFu Medical Appliance Co., Ltd. The artificial dura mater was mainly made of bovine tendon as a raw material in China, which was collected from state-run large-scale meat production and processing enterprises using aseptic technique to ensure the material source in line with national standards. After collection, Achilles tendon that was healthy, fresh and safe was processed into sponge-like collagen biofilm scaffolds for repair of dura mater, nerve sheath, skin, cartilage as well as prevention of various postoperative adhesions. The product consisted of collagen foils with biocompatibility, toughness, various sizes which were soft and could be completed absorbed. Biomedical glue was purchased from BIOSEAL BIOTECH, Guangzhou, China, which was developed from bovine blood source[14-15].
Methods
Dural defect repair
Under general anesthesia, all patients underwent tracheal intubation. A large (reversed) question mark-shaped skin incision based at the ear (1 cm anterior to the antilobium) was made to expose the frontal, temporal and parietal region. Then, five or six holes were drilled on the skull, and a bone flap including the frontal, temporal squama, and parietal bone was removed. When the bone window was above the middle cranial fossa, the parts from the temporal squama to the base of the middle cranial fossa was cut using a rongeur to remove epidural hematoma, subdural hematoma, intracerebral hematoma, brain contusion lesions. The dura could be cut in radial, arc-shaped, cruciform manners, which was determined according to surgical sites and requirements. The dura edge was cut as smooth as possible to clear inactive brain tissues and intracranial hematoma. The dura should be fulgurized as little as possible to avoid dural retraction. The smooth surface of the artificial dura mater faced to tissue defects. After repair, the dura mater could even be lifted via four points at a height of 2.5-3.0 cm. Generally, the brain tissue was allowed to bulge outwards in a length of at least 2.5 cm, so there was a certain region allowing the floating of the dura, which could help to buffer the intracranial pressure. If there was no high intracranial pressure and no encephalocele, the bone flap was fixed; otherwise, the bone flap was removed to reduce the intracranial pressure. Subdural catheter was connected to a drainage bag, and the epidural catheter was attached to negative pressure. Temporalis muscle and subgaleal were tightly sutured.
Postoperative process
All patients undergoing dural repair were enrolled at the neurosurgical intensive care unit for postoperative care and treatment. As appropriate, mechanical ventilation and tracheotomy were performed to maintain the airway patency as well as dehydration and reduing intracranial pressure. Routine treatment against infection and gastrointestinal bleeding was done, and in some patients, hypothermia and sedation therapy was given to relieve brain swelling and brain edema. Active treatments for nerve nutrition and improvement of microcirculation of the central nervous system were preferred. For coma patients, enteral and parenteral nutrition support and early rehabilitation were necessary to promote the recovery of neurological function.
Ozone-enriched autologous blood transfusion
In the supine, blood samples were extracted from the cubital vein. First, the blood transfusion apparatus was inserted into saline and exhausted. Then, the short tube of the apparatus was inserted into the blood transfusion bag, and full of anticoagulant agents from the blood transfusion bag via the open access. Blood samples from the vein, 100 mL, were collected via puncture followed by shutting the blood transfusion path, and the saline pathway was open for intravenous infusion. Ozone sample, 47 mg/g, was extracted and injected into the blood bag following gently shaking until the blood was fully ozonized. Then, the saline pathway was closed, and the transfusion path was open. The ozonized blood was transferred back to the patients. After blood transfusion, the saline pathway was open until the blood was completely into the body.
Complications and follow-up
Patients were subject to periodic head CT examination after surgery, and underwent routine cranioplasty at postoperative 3-6 months. After repair, complications associated with the artificial meninges were observed and recorded, and all patients were followed up for 1 year.
Complication evaluation: The observation indexes included (1) inflammation, (2) adhesion, and (3) subflap effusion.
Main outcome measures
Head CT examination results and complications.
Data were expressed as mean±SD and analyzed statistically using the SPSS 19.0 (IBM, USA). Paired t test was done for evaluation of intracranial pressure and midline shift level. A value of P < 0.05 was considered statistically significant.
试验通过观察60例联合应用胶原海绵人工硬脑膜修补和臭氧治疗颅脑损伤患者,证实胶原海绵人工硬脑膜和臭氧联合治疗能有效减轻颅脑损伤颅内压,且胶原海绵人工硬脑膜生物相容性好,能够较好的与正常硬脑膜融合,为后期颅骨修补创造有利条件。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||