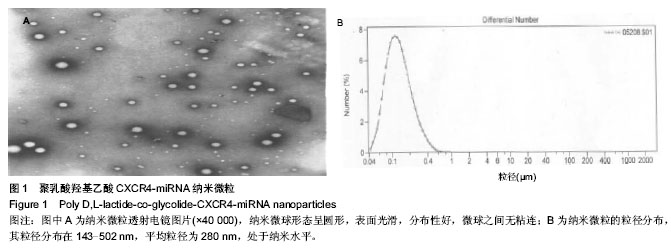
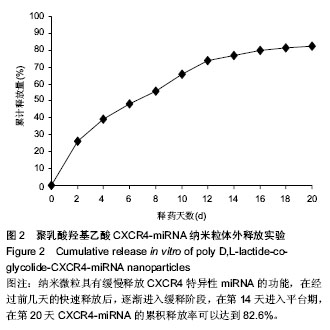
| [1] Wei F,Moore DC,Li Y,et al.Attenuation of osteoarthritis via blockade of the SDF-1/CXCR4 signaling pathway.Arthritis Res Ther.2012;14(4):R177.
[2] Bikash D,Shili X,Fedora G,et al.Small Molecule Inhibitors of CXCR4.Theranostics.2013;3(1):47-75.
[3] Jin F,Brockmeier U,Otterbach F,et al.New insight into the SDF-1/CXCR4 axis in a breast carcinoma model: hypoxia-induced endothelial SDF-1 and tumor cell CXCR4 are required for tumor cell intravasation.Mol Cancer Res. 2012;10(8):1021-1031.
[4] Heckmann D,Maier P,Laufs S,et al.The disparate twins: a comparative study of CXCR4 and CXCR7 in SDF-1α-induced gene expression, invasion and chemosensitivity of colon cancer.Clin Cancer Res.2014;20(3):604-616.
[5] Seku?a M,Miekus K,Majka M.Downregulation of the CXCR4 receptor inhibits cervical carcinoma metastatic behavior in vitro and in vivo.Int J Oncol.2014;44(6):1853-1860.
[6] Zhao Y,Guo J,Gu S,et al.SDF-1/CXCR4 signal is involved in decreased expression of p57kip2 in de novo MDS patients. Hematology.2012;17(4):220-228.
[7] Wang L, Huang T, Chen W, et al. Silencing of CXCR4 by RNA interference inhibits cell growth and metastasis in human renal cancer cells.Oncol Rep. 2012;28(6): 2043-2048.
[8] D'Alterio C,Consales C,Polimeno M,et al.Concomitant CXCR4 and CXCR7 expression predicts poor prognosis in renal cancer.Curr Cancer Drug Targets. 2010;10(7):772-781.
[9] Bhardwaj A,Singh S,Singh AP.MicroRNA-based Cancer Therapeutics: Big Hope from Small RNAs.Mol Cell Pharmacol. 2010;2(5):213-219.
[10] Zhang G,Wang Q,Xu R.Therapeutics Based on microRNA: A New Approach for Liver Cancer.Curr Genomics.2010;11(5): 311-325.
[11] Ranganath SH,Fu Y,Arifin DY,et al.The use of submicron/nanoscale PLGA implants to deliver paclitaxel with enhanced pharmacokinetics and therapeutic efficacy in intracranial glioblastoma in mice.Biomaterials. 2010;31(19): 5199-5207.
[12] de Martimprey H,Bertrand JR,Malvy C,et al.New core-shell nanoparticules for the intravenous delivery of siRNA to experimental thyroid papillary carcinoma. Pharm Res. 2010; 27(3):498-509.
[13] 董勤,崔杰,高烽,等.STAT3反义寡核苷酸纳米复合微粒的制备及特性研究[J].武警医学院学报,2011,20(9):723-726.
[14] Liu JW,Zhao Q,Wan CX.Research progresses on degradation mechanism in vivo and medical applications of polylactic acid.Space Med Eng.2001;14(4):308-312.
[15] Sahoo SK,Ma Labhasetwar V.Efficacy of transferrin- conjugated paclitaxel-loaded nanoparticles in a murine model of prostate cancer.Int J Cancer.2004;112(2): 335-340.
[16] Rivera PA,Martinez-Oharriz MC,Rubio M,et al.Fluconazole encapsulation in PLGA microspheres by spray-drying.J Microencaspsul.2004;21(2):203-211.
[17] Cheng FY,Wang SP,Su CH,et al. Stabilizer-free poly(lactide-co-glycolide)nanoparticles for multimodal biomedical probes.Biomaterials.2008;29(13):2014-2112.
[18] Amjadi I,Rabice M,Hosseini MS,et al.Synthesis and characterization of doxorubicin-loaded poly(lactide-co- glycolide)nanoparticles as a sustained-release anticancer drug delivery system.Appl Biochem Biotechnol.2012;168(6): 1434-1447.
[19] Nair KL,Thulasidasan AKT,Deepa G,et al.Purely aqueous PLGA nanoparticulate formulations of curcumin exhibit enhanced anticancer activity with dependence on the combination of the carrier.Int J Pharm.2012;425(1/2):44-52.
[20] Anand P,Nair HB,Sung B,et al.Design of Curcumin Loaded PLGA Nanoparticles Formulation with Enhanced Cellular Uptake, and Increased Bioactivity in vitro and Superior Bioavailabilityin vivo.Biochem Pharmacol.2010;79(3): 330-338.
[21] Wang YF,Liu PF,Qiu LH,et al.Toxicity and therapy of cisplatin- loaded EGF modified mPEG-PLGA-PLL nanoparticles for SKOV3 cancer in mice.Biomaterials. 2013;34(16):4068-4077.
[22] Chen J,Li S,Shen Q.Folic acid and cell-penetrating peptide conjugated PLGA-PEG bifunctional nanoparticles for vincristine sulfate delivery.Eur J Pharm Sci. 2012;47(2): 430-443.
[23] Graf N,Bielenberg DR,Kolishetti N,et al.alpha(V)beta(3) Integrin- Targeted PLGA-PEG Nanoparticles for Enhanced Anti-tumor Efficacy of a Pt(IV) Prodrug.ACS Nano. 2012;6(5): 4530-4539.
[24] Wang G,Yu B,Wu Y,et al.Controlled preparation and antitumor efficacy of vitamin E TPGS-functionalized PLGA nanoparticles for delivery of paclitaxel.Int J Pharm. 2013;446(1/2):24-33.
[25] Chakravarthi SS, Robinson DH.Enhanced cellular association of paclitaxel delivered in chitosan-PLGA particles. Int J Pharm. 2011;409(1-2):111-120.
[26] 马立坤,叶鹏,黄文良,等.骨形态发生蛋白2/聚乳酸缓释微球的制备及表征[J].中国组织工程研究,2014,18(3):395-400.
[27] Dangi R,Hurkat P,Jain A,et al.Targeting liver cancer via ASGP receptor using 5-FU-loaded surface-modified PLGA nanoparticles.J Microencapsul.2014;31 (5):479-487.
[28] Zanin H,Hollanda LM,Ceragioli HJ,et al.Carbon nanoparticles for gene transfection in eukaryotic cell lines. Mater Sci Eng C Mater Biol Appl.2014;39:359-370.
[29] Apaolaza PS,Delgado D,del Pozo-Rodríguez A,et al.A novel gene therapy vector based on hyaluronic acid and solid lipid nanoparticles for ocular diseases. Int J Pharm.2014;465(1-2): 413-426.
[30] Ji XT,Huang L,Huang HQ.Construction of nanometer cisplatin core-ferritin (NCC-F) and proteomic analysis of gastric cancer cell apoptosis induced with cisplatin released from the NCC-F. J Proteomics.2012;75(11):3145-3157.
[31] Liu J,Jiang Z,Zhou J,et al.Enzyme-synthesized poly (amine-co-esters) as nonviral vectors for gene delivery.J Biomed Mater Res A.2011;96(2):456-465.
[32] Chen M,Ouyang H, Zhou S,et al.PLGA-nanoparticle mediated delivery of anti-OX40 monoclonal antibody enhances anti-tumor cytotoxic T cell responses. Cell Immunol. 2014;287(2):91-99.
[33] Feng L,Ward JA,Li SK,et al.Assessment of PLGA-PEG-PLGA copolymer hydrogel for sustained drug delivery in the ear.Curr Drug Deliv,.2014;11(2):279-286.
[34] Jagani HV,Josyula VR,Palanimuthu VR,et al.Improvement of therapeutic efficacy of PLGA nanoformulation of siRNA targeting anti-apoptotic Bcl-2 through chitosan coating. Eur J Pharm Sci.2013;48(4-5):611-618.
[35] Chen YS,Alany RG,Young SA,et al.In vitro release characteristics and cellular uptake of poly (D,L-lactic-co-glycolic acid) nanoparticles for topical delivery of antisense oligodeoxynucleotides.Drug Deliv.2011;18(7):493-501.
[36] Lane ME,Brennan FS,Corrigan OI.Comparison of post-emulsification freeze drying or spray drying processes for the microencapsulation of plasmid DNA. J Pharm Pharmacol. 2005;57(7):831-838.
[37] Qi F,Wu J,Fan Q,et al.Preparation of uniform-sized exenatide-loaded PLGA microspheres as long-effective release system with high encapsulation efficiency and bio-stability.Colloids Surf B Biointerfaces.2013;112:492-498.
[38] 郑延波,刘胜,李丽君,等.包载缺氧诱导因子-1ɑ反义寡核苷酸纳米粒子的制备和特性评价[J].生物医学工程与临床,2011, 15(6): 579-563.
[39] Matsumoto A,Matsukawa Y,Suzuki T,et al.Drug release characteristics of multi-reservoir type microspheres with poly(dl-lactide-co-glycolide) and poly(dl-lactide). J Control Release.2005;106(1-2):172-180.
[40] 叶向阳,孙湘,贾会文,等.利福平/聚乳酸-聚羟基乙酸缓释微球的制备及特性[J].中国组织工程研究与临床康复,2011,15(51): 9608-9612.
[41] Lee YS, Lowe JP,Gilby E,et al.The initial release of cisplatin from poly(lactide-co-glycolide) microspheres.Int J Pharm. 2010;383(1-2):244-254 |