中国组织工程研究 ›› 2014, Vol. 18 ›› Issue (1): 7-13.doi: 10.3969/j.issn.2095-4344.2014.01.002
• 骨髓干细胞 bone marrow stem cells • 上一篇 下一篇
骨髓间充质干细胞增殖能力与皮质类固醇性骨坏死
王佰亮,李铁军,岳德波,孙 伟
- 中日友好医院骨关节外科,北京市 100029
-
修回日期:2013-10-24出版日期:2014-01-01发布日期:2014-01-01 -
通讯作者:王佰亮,博士,副主任医师,中日友好医院骨关节外科,北京市 100029 -
作者简介:王佰亮,男,汉族,1978年生,山东省潍坊市人,2007年北京协和医学院毕业,博士,副主任医师。
Proliferation ability of bone marrow mesenchymal stem cells in corticosteroid-induced osteonecrosis of femoral head
Wang Bai-liang, Li Tie-jun, Yue De-bo, Sun Wei
- Department of Joint Surgery, China-Japan Friendship Hospital, Beijing 100029, China
-
Revised:2013-10-24Online:2014-01-01Published:2014-01-01 -
Contact:Wang Bai-liang, M.D., Associate chief physician, Department of Joint Surgery, China-Japan Friendship Hospital, Beijing 100029, China -
About author:Wang Bai-liang, M.D., Associate chief physician, Department of Joint Surgery, China-Japan Friendship Hospital, Beijing 100029, China
摘要:
背景:皮质类固醇激素性骨坏死是造成髋关节功能丧失的主要病因之一。近年研究表明,激素性股骨头坏死可能与激素引起的骨髓间充质干细胞增殖能力有关。 目的:检测皮质类固醇性骨坏死患者骨髓间充质干细胞的增殖活性,为建立自体骨髓干细胞移植治疗股骨头坏死的合理性寻求证据。 方法:选取皮质类固醇性股骨头坏死病例设为股骨头坏死组,按取材部位不同再分为股骨头坏死股骨头组、股骨头坏死髂骨组,同时选取无股骨头坏死、无激素应用的拟行人工关节置换的股骨颈骨折患者设为对照组。用密度梯度离心法分离各组骨髓间充质干细胞,再经贴壁筛选法筛选,选取第3代细胞进行实验。 结果与结论:MTT结果显示,股骨头坏死股骨头组增殖能力明显弱于其他2组,其骨髓间充质干细胞在培养后1-7 d为生长滞留期,第8天达到对数生长期,以后进入到平台期,而其他2组较病例组生长曲线明显前移,并且峰值增高。流式细胞仪测定的细胞周期结果显示,股骨头坏死股骨头组中G0/G1细胞比例明显增高,而S+G2/M期细胞比例降低,细胞增殖指数较其他2组降低(P < 0.05),而股骨头坏死髂骨组的细胞增殖活性最强。结果证实,皮质类固醇性骨坏死患者股骨头来源的骨髓间充质干细胞增殖活性较低,髂骨来源的骨髓间充质干细胞活性增殖活性较高。
中图分类号:
引用本文
王佰亮,李铁军,岳德波,孙 伟. 骨髓间充质干细胞增殖能力与皮质类固醇性骨坏死[J]. 中国组织工程研究, 2014, 18(1): 7-13.
Wang Bai-liang, Li Tie-jun, Yue De-bo, Sun Wei. Proliferation ability of bone marrow mesenchymal stem cells in corticosteroid-induced osteonecrosis of femoral head[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(1): 7-13.
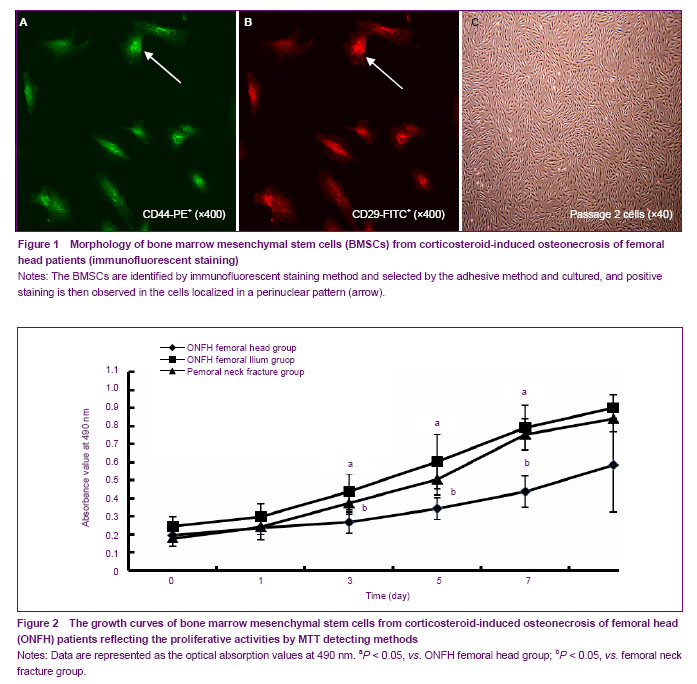
There was marked difference in growth state of BMSCs between corticosteroid-induced ONFH femoral head group and the other two groups under an inverted microscope 24 hours after primary culture, and BMSCs from all groups adhered and extended in spindle or polygonal shape. Forty-eight hours later, the clone formed and adherent cells increased. However, the number of clones in the corticosteroid-induced ONFH femoral head group was much less than that in the other two groups. BMSCs resembled typical features of fibroblasts 3 days later in the other two groups and 5 days later in the corticosteroid-induced ONFH femoral head group. Clones enlarged and fused into monolayer 7 days later in the femoral neck fracture group and the corticosteroid-induced ONFH ilium group and 10 days later in corticosteroid-induced ONFH femoral head group. In the femoral neck fracture group and the corticosteroid-induced ONFH ilium group, BMSCs proliferated and distributed evenly after passage and adhered partially 2 hours later and completely in funicular shape 24 hours later. In contrast, BMSCs from corticosteroid-induced ONFH femoral head group proliferated very slowly and overgrew the bottle till 10-12 days later. The immunofluorescent staining showed that the cultured cells expressed typical BMSCs markers such as CD29 and CD44, but not typical haematopoietic cell markers including CD34, CD14 and CD133 (Figures 1 A-C).
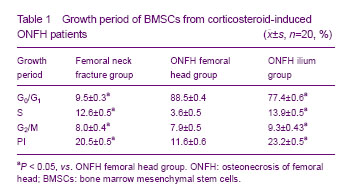
The proliferative activities of BMSCs using MTT methods were obtained from the three groups and drawn into the growth curves. As shown in the figures, the proliferative activity of BMSCs in the femoral neck fracture group and the corticosteroid-induced ONFH ilium group were remarkably stronger than that in the corticosteroid-induced ONFH femoral head group. For the corticosteroid-induced ONFH femoral head group, BMSCs growth fell into the stagnant stage within 1-7 days after seeding, turned into the logarithmic stage at 8 days, and came to the plateau stage thereafter. However, the growth curve in the femoral neck fracture group and the corticosteroid-induced ONFH ilium group moved left obviously and the peak increased (Figure 2).
For FCAS, the percentage of cells in S stage or G2/M+S stages was usually taken as PI to indicate the proliferation of cells. Compared with the other two groups, the percentage of cells in G0/G1 stages in the corticosteroid-induced ONFH femoral head group was increased significantly while the percentage in G2/M+S stages (PI) was decreased significantly (P < 0.05). The percentage of cells in G2 stage in the corticosteroid-induced ONFH ilium group was the highest in the three groups. These were in consistent with results revealed by MTT method (Table 1).
| [1] Jones JP Jr. Fat embolism, intravascular coagulation,and osteonecrosis. Clin Orthop Relat Res. 1993;(292):294-308. [2] Mirzai R, Chang C, Greenspan A, et al. The pathogenesis of osteonecrosis and the relationships to corticosteroids. J Asthma.1999;36(1):77-95. [3] Pittenger MF, Mackay AM, Jaiswal SC, et al. Multilineage potential of adult human mesenchamal stem cells. Science. 1999;284(5411):143-147. [4] Hernigou P, Beaujean F, Lambotte JC. Decrease in the mesenchymal stem cell pool in the proximal femur in corticosteroid-induced osteonecrosis. J Bone Joint Surg (Br). 1999;81(2):349-355. [5] Gangji V, Hauzeur JP, Schoutens A, et al. Abnormalities in the replicative capacity of osteoblastic cells in the proximal femur of patients with osteonecrosis of the femoral head. J Rheumatol. 2003;30(2):348-351. [6] Wang GJ, Cui Q, Balian G. The pathogenesis and prevention of steroid-induced osteonecrosis. Clin Orthop. 2000;(370):295-310. [7] State Council of the People’s Republic of China. Administrative Regulations on Medical Institution. 1994-09-01.[8] Suh KT, Kim SW, Roh HL, et al. Decreased osteogenic differentiation of mesenchymal stem cells in alcohol-induced osteonecrosis. Clin Orthop Relat Res. 2005;(431):220-225. [9] Beyer Nardi N, da Silva Meirelles L. Mesenchymal stem cells: isolation, in vitro expansion and characterization. Handb Exp Pharmacol. 2006;(174):249-282. [10] Drescher W, Weigert KP, Bunger MH, et al. Fermoral head blood flow reduction and hypercoagulablitity under 24 h megadose steroid treatment in pigs. J Orthop Res. 2004; 22(3):501-508. [11] Asano T, Takahashi KA, Fujioka M, et al. Genetic analysis of steroid-induced osteonecrosis of the femoral head. J Orthop Sci. 2003;8(3):329-333. [12] Tektonidou MG, Moutsopoulos HM. Immunologic factor in the pathogenesis of osteonecrosis. Orthop Clin North Am. 2004;35(3):259-263. [13] Wu Z, Ji C, Li H, et al. Elevated level of membrane microparticles in the disease of steroid-inducedvascular osteonecrosis. J Craniofac Surg. 2013;24(4):1252-1256. [14] Li X, Jin L, Cui Q, et al. Steroid effects on osteogenesis through mesenchymal cell gene expression. Osteoporos Int. 2005;16(1):101-108. [15] Cui Q, Wang GJ, Balian G. Steroid-induced adipogenesis in a pluripotential cell line from bone marrow. J Bone Joint Surg Am. 1997;79(7):1054-1063. [16] Lee JS, Lee JS, Roh HL, et al. Alterations in the differentiation ability of mesenchymal stem cells in patients with nontraumatic osteonecrosis of the femoral head: comparative analysis according to the risk factor. J Orthop Res. 2006;24(4):604-609. [17] Yin L, Li YB, Wang YS. Dexamethasone-induced adipogenesis in primary marrow stromal cell cultures: mechanism of steroid-induced osteonecrosis. Chin Med J (Engl). 2006;119(7):581-588. [18] Castro-Malaspina H, Gay RE, Resnick G, et al. Characterization of human bone marrow fibroblast colony-forming cells (CFU-F) and their progeny. Blood. 1980;56(2):289-301. [19] Egrise D, Martin D, Vienne A, et al. The number of fibroblastic colonies formed from bone marrow is decreased and the in vitro proliferation rate of trabecular bone cells increased in aged rats. Bone. 1992;13(5): 355-361. [20] Fazeli PK, Horowitz MC, MacDougald OA, et al. Marrow fat and bone--new perspectives. J Clin Endocrinol Metab. 2013;8(3):935-945. [21] Penolazzi L, Mazzitelli S, Vecchiatini R, et al. Human mesenchymal stem cells seeded on extracellular matrix-scaffold: viability and osteogenic potential. J Cell Physiol. 2012;27(2):857-866. [22] Muschler GF, Nitto H, Boehm CA, et al. Age- and gender-related changes in the cellularity of human bone marrow and the prevalence of osteoblastic progenitors. J Orthop Res. 2001;19(1):117-125. [23] Glimcher MJ, Kenzora JE. The biology of osteonecrosis of the human femoral head and its clinical implications: III. Discussion of the etiology and genesis of the pathological sequelae: Comments on treatment. Clin Orthop. 1979;(140): 273-312. [24] Helbig L, Simank HG, Kroeber M, et al. Core decompression combined with implantation of a demineralised bone matrix for non-traumatic osteonecrosis of the femoral head. Arch Orthop Trauma Surg. 2012;132(8):1095-1103. [25] Youm YS, Lee SY, Lee SH. Apoptosis in the osteonecrosis of the femoral head. Clin Orthop Surg. 2010;2(4):250-255.[26] Wang BL, Sun W, Shi ZC, et al. Decreased proliferation of mesenchymal stem cells in corticosteroid-induced osteonecrosis of femoral head. Orthopedics. 2008;31(5): 444. [27] Hernigou P, Beaujean F. Abnormalities in the bone marrow of the iliac crest in patients who have osteonecrosis secondary to corticosteroid therapy or alcohol abuse. J Bone Joint Surg (Am). 1997;79(7):1047-1053. [28] Gangji V, De Maertelaer V, Hauzeur JP. Autologous bone marrow cell implantation in the treatment of non-traumatic osteonecrosis of the femoral head: five year follow-up of a prospective controlled study. Bone. 2011;9(5):1005-1009. [29] Weinstein RS, Nicholas RW, Manolagas SC. Apoptosis of osteocytes in glucocorticoid-induced osteonecrosis of the hip. J Clin Endocrinol Metab. 2000;85(8):2907-2912. [30] Weinstein RS, Jilka RL, Parfitt AM, et al. Inhibition of osteoblastogenesis and promotion of apoptosis of osteoblasts and osteocytes by glucocorticoids.Potential mechanisms of their deleterious effects on bone. J Clin Invest. 1998;102(2):274-282. [31] Hofbauer LC, Khosla S, Dunstan CR, et al. The roles of osteoprotegerin and osteoprotegerin ligand in the paracrine regulation of bone resorption. J Bone Mner Res. 2000;15: 2-12. [32] Wang BL, Sun W, Shi ZC, et al. Treatment of nontraumatic osteonecrosis of the femoral head with the implantation of core decompression and concentrated autologous bone marrow containing mononuclear cells. Arch Orthop Trauma Surg. 2010;130(7):859-865.[33] Zhao D, Cui D, Wang B, et al. Treatment of early stage osteonecrosis of the femoral head with autologous implantation of bone marrow-derived and cultured mesenchymal stem cells. Bone. 2012;50(1):325-330. [34] Chotivichit A, Korwutthikulrangsri E, Auewarakul C, et al. Core decompression and concentrated autologous bone marrow injection for treatment of osteonecrosis of the femoral head. J Med Assoc Thai. 2012;95(Suppl 9):S14-S20. [35] Ji WF, Ding WH, Ma ZC, et al. Three-tunnels core decompression with implantation of bone marrow stromal cells (bMSCs) and decalcified bone matrix (DBM) for the treatment of early femoral head necrosis. Zhongguo Gu Shang. 2008;21(10):776-778. [36] Yan ZQ, Chen YS, Li WJ, et al. Treatment of osteonecrosis of the femoral head by percutaneous decompression and autologous bone marrow mononuclear cell infusion. Chin J Traumatol. 2006;9(1):3-7. [37] Liu Y, Liu S, Su X. Core decompression and implantation of bone marrow mononuclear cells with porous hydroxylapatite composite filler for the treatment of osteonecrosis of the femoral head. Arch Orthop Trauma Surg. 2013;133(1):125-133. [38] Chang T, Tang K, Tao X, et al. Treatment of early avascular necrosis of femoral head by core decompression combined with autologous bone marrow mesenchymal stem cells transplantation. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2010;24(6):739-743. |
| [1] | 林清凡, 解一新, 陈婉清, 叶振忠, 陈幼芳. 人胎盘源间充质干细胞条件培养液可上调缺氧状态下BeWo细胞活力和紧密连接因子的表达[J]. 中国组织工程研究, 2021, 25(在线): 4970-4975. |
| [2] | 蒲 锐, 陈子扬, 袁凌燕. 不同细胞来源外泌体保护心脏的特点与效应[J]. 中国组织工程研究, 2021, 25(在线): 1-. |
| [3] | 韦 玮, 李 剑, 黄林海, 兰敏东, 卢显威, 黄绍东. 全膝或全髋关节置换后老年人首次活动时跌倒恐惧的影响因素[J]. 中国组织工程研究, 2021, 25(9): 1351-1355. |
| [4] | 张 冲, 刘志昂, 姚帅辉, 高军胜, 姜 岩, 张 陆. 局部应用氨甲环酸减少老年股骨颈骨折全髋关节置换后引流的安全和有效性[J]. 中国组织工程研究, 2021, 25(9): 1381-1386. |
| [5] | 陈俊名, 岳 辰, 何沛霖, 张俊涛, 孙墨渊, 刘又文. 髋关节置换与股骨近端防旋髓内钉内固定修复高龄股骨转子间骨折效果的Meta分析[J]. 中国组织工程研究, 2021, 25(9): 1452-1457. |
| [6] | 张秀梅, 翟运开, 赵 杰, 赵 萌. 类器官模型国内外数据库近10年文献研究热点分析[J]. 中国组织工程研究, 2021, 25(8): 1249-1255. |
| [7] | 李中峰, 陈明海, 凡一诺, 魏秋实, 何 伟, 陈镇秋. 右归饮治疗激素性股骨头坏死作用机制的网络药理学分析[J]. 中国组织工程研究, 2021, 25(8): 1256-1263. |
| [8] | 刘 聪, 刘 肃. miR-17-5p调控低氧诱导因子1α介导脂肪细胞分化及血管生成的分子机制[J]. 中国组织工程研究, 2021, 25(7): 1069-1074. |
| [9] | 王正东, 黄 娜, 陈婧娴, 郑作兵, 胡鑫宇, 李 梅, 苏 晓, 苏学森, 颜 南. 丁酸钠抑制氟中毒可诱导小胶质细胞活化及炎症因子表达增多[J]. 中国组织工程研究, 2021, 25(7): 1075-1080. |
| [10] | 汪显耀, 关亚琳, 刘忠山. 提高间充质干细胞治疗难愈性创面的策略[J]. 中国组织工程研究, 2021, 25(7): 1081-1087. |
| [11] | 万 然, 史 旭, 刘京松, 王岩松. 间充质干细胞分泌组治疗脊髓损伤的研究进展[J]. 中国组织工程研究, 2021, 25(7): 1088-1095. |
| [12] | 廖成成, 安家兴, 谭张雪, 王 倩, 刘建国. 口腔鳞状细胞癌干细胞的治疗靶点及应用前景[J]. 中国组织工程研究, 2021, 25(7): 1096-1103. |
| [13] | 谢文佳, 夏天娇, 周卿云, 刘羽佳, 顾小萍. 小胶质细胞介导神经元损伤在神经退行性疾病中的作用[J]. 中国组织工程研究, 2021, 25(7): 1109-1115. |
| [14] | 李珊珊, 郭笑霄, 尤 冉, 杨秀芬, 赵 露, 陈 曦, 王艳玲. 感光细胞替代治疗视网膜变性疾病[J]. 中国组织工程研究, 2021, 25(7): 1116-1121. |
| [15] | 焦 慧, 张一宁, 宋雨晴, 林 宇, 王秀丽. 乳腺癌类器官研究进展及临床应用前景[J]. 中国组织工程研究, 2021, 25(7): 1122-1128. |
Before the study, enrolled patients were informed of the program and risk of study, and the written informed consent was obtained for all patients for research.
.jpg)
Methods
All statistical analyses were completed using SPSS Statistical Software 10.0 (SPSS, Chicago, IL, USA). Mean±SD and frequencies were calculated for general demographic and routine clinical data. The Mann-Whitney U test was used to compare the non-parametric data between two independent samples. P < 0.05 was considered to be statistically significant.
皮质类固醇性股骨头坏死的自行修复能力有限可能与此类患者股骨头内骨髓间充质干细胞增殖活性下降有关。实验通过检测股骨头坏死患者股骨头内和髂骨处来源的的骨髓间充质干细胞的增殖活性,并与股骨颈骨折的患者做对照,发现股骨头坏死患者股骨头内的骨髓间充质干细胞增殖活性明显下降,明显低于股骨颈骨折患者和其髂骨来源的骨髓间充质干细胞,而其髂骨来源的骨髓间充质干细胞活性增殖活性最高,这就为移植含有骨髓间充质干细胞的浓缩骨髓血治疗股骨头坏死提供了理论依据。
皮质类固醇性股骨头坏死的自行修复能力有限可能与这些患者股骨头内骨髓间充质干细胞增殖活性下降有关。我们假设皮质类固醇性股骨头坏死患者的体内的骨髓间充质干细胞增殖活性有关改变,检测来源于皮质类固醇性股骨头坏死患者的股骨头内和髂骨骨髓间充质干细胞的增殖活性,并与股骨颈骨折患者股骨头内部的骨髓间充质干细胞做对照,为建立自体骨髓干细胞移植治疗股骨头坏死的合理性寻求证据。研究发现与股骨颈骨折的患者相比较,股骨头坏死患者股骨头内的骨髓间充质干细胞增殖活性明显下降,要明显低于股骨颈骨折患者和其髂骨来源的骨髓间充质干细胞,而其髂骨来源的骨髓间充质干细胞活性增殖活性最高。实验不足之处在于未设置非骨坏死患者的髂骨来源的骨髓间充质干细胞做对照,而不能发现激素性骨坏死患者髂骨来源的骨髓间充质干细胞活性是否也受到了激素的影响。 皮质类固醇性股骨头坏死的自行修复能力有限可能与这些患者股骨头内骨髓间充质干细胞增殖活性下降有关。实验通过检测股骨头坏死患者股骨头内和髂骨处来源的的骨髓间充质干细胞的增殖活性,并与股骨颈骨折的患者做对照,发现股骨头坏死患者股骨头内的骨髓间充质干细胞增殖活性明显下降,要明显低于股骨颈骨折患者和其髂骨来源的骨髓间充质干细胞,而其髂骨来源的骨髓间充质干细胞活性增殖活性最高,这就为移植含有骨髓间充质干细胞的浓缩骨髓血治疗股骨头坏死提供了理论依据。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||