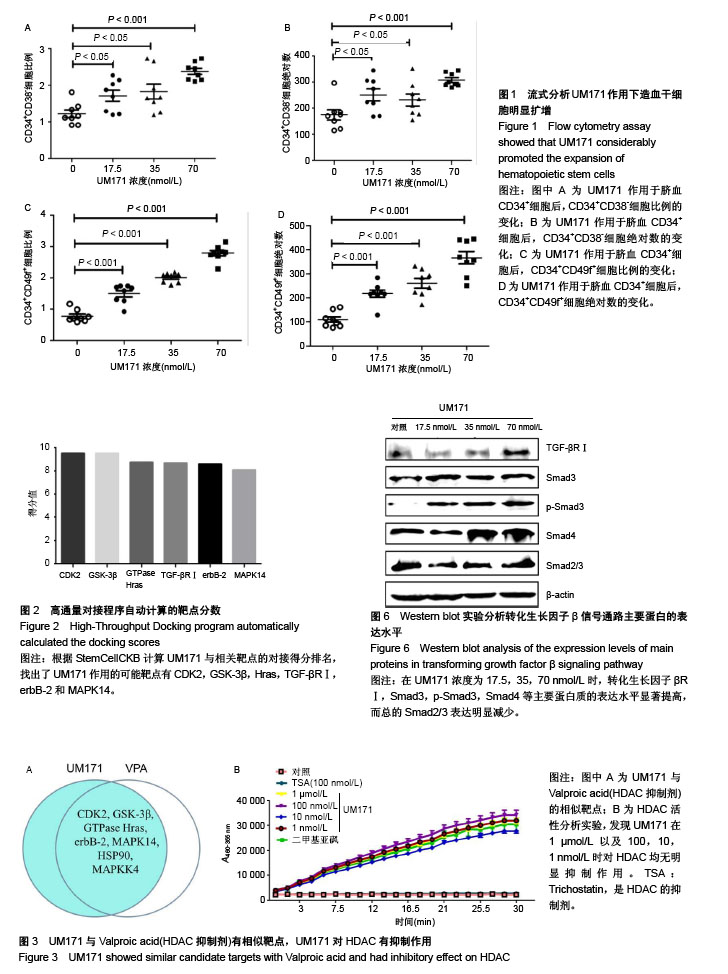
| [1] Bozda? SC, Yüksel MK, Demirer T. Adult Stem Cells and Medicine. Adv Exp Med Biol. 2018 Mar 20. doi: 10. 1007/5584_2018_184. [Epub ahead of print][2] Peccatori J, Ciceri F. Allogeneic stem cell transplantation for acute myeloid leukemia. Haematologica. 2010;95(6): 857-859. [3] Gragert L, Eapen M, Williams E, et al. HLA match likelihoods for hematopoietic stem-cell grafts in the U. S. registry. N Engl J Med. 2014;371(4):339-348. [4] Fares I, Chagraoui J, Gareau Y, et al. Cord blood expansion. Pyrimidoindole derivatives are agonists of human hematopoietic stem cell self-renewal. Science. 2014;345 (6203):1509-1512. [5] Ramírez P, Brunstein CG, Miller B, et al. Delayed platelet recovery after allogeneic transplantation: a predictor of increased treatment-related mortality and poorer survival. Bone Marrow Transplant. 2011;46(7):981-986. [6] Rocha V, Labopin M, Sanz G, et al. Transplants of umbilical-cord blood or bone marrow from unrelated donors in adults with acute leukemia. N Engl J Med. 2004;351(22): 2276-2285. [7] Miller PH, Knapp DJ, Eaves CJ. Heterogeneity in hematopoietic stem cell populations: implications for transplantation. Curr Opin Hematol. 2013;20(4):257-264. [8] Morin V. Stem-cell replication to treat blood diseases. CMAJ. 2014;186(17):E648. [9] Brunstein CG, Gutman JA, Weisdorf DJ, et al. Allogeneic hematopoietic cell transplantation for hematologic malignancy: relative risks and benefits of double umbilical cord blood. Blood. 2010;116(22):4693-4699. [10] Ali S, Yu SM, Yao ZJ. Gram-Scale Laboratory Synthesis of UM171, a Potent Agonist of Human Hematopoietic Stem Cell Self-Renewal. J Org Chem. 2016;81(21):10236-10241. [11] Zhang Y, Wang L, Feng Z, et al. StemCellCKB: An Integrated Stem Cell-Specific Chemogenomics KnowledgeBase for Target Identification and Systems-Pharmacology Research. J Chem Inf Model. 2016;56(10):1995-2004. [12] Blank U, Karlsson S. TGF-β signaling in the control of hematopoietic stem cells. Blood. 2015;125(23):3542-3550. [13] Batard P, Monier MN, Fortunel N, et al. TGF-(beta)1 maintains hematopoietic immaturity by a reversible negative control of cell cycle and induces CD34 antigen up-modulation. J Cell Sci. 2000;113(Pt 3):383-390. [14] Fortunel NO, Hatzfeld A, Hatzfeld JA. Transforming growth factor-beta: pleiotropic role in the regulation of hematopoiesis. Blood. 2000;96(6):2022-2036. [15] Garbe A, Spyridonidis A, Möbest D, et al. Transforming growth factor-beta 1 delays formation of granulocyte- macrophage colony-forming cells, but spares more primitive progenitors during ex vivo expansion of CD34+ haemopoietic progenitor cells. Br J Haematol. 1997;99(4):951-958. [16] Yamazaki S, Iwama A, Takayanagi S, et al. Cytokine signals modulated via lipid rafts mimic niche signals and induce hibernation in hematopoietic stem cells. EMBO J. 2006;25(15): 3515-3523. [17] Yamazaki S, Iwama A, Takayanagi S, et al. TGF-beta as a candidate bone marrow niche signal to induce hematopoietic stem cell hibernation. Blood. 2009;113(6):1250-1256. [18] Cheshier SH, Morrison SJ, Liao X, et al. In vivo proliferation and cell cycle kinetics of long-term self-renewing hematopoietic stem cells. Proc Natl Acad Sci U S A. 1999; 96(6):3120-3125. [19] Wilson A, Laurenti E, Oser G, et al. Hematopoietic stem cells reversibly switch from dormancy to self-renewal during homeostasis and repair. Cell. 2008;135(6):1118-1129. [20] Heldin CH, Landström M, Moustakas A. Mechanism of TGF-beta signaling to growth arrest, apoptosis, and epithelial-mesenchymal transition. Curr Opin Cell Biol. 2009;21(2):166-176. [21] Zuo W, Huang F, Chiang YJ, et al. c-Cbl-mediated neddylation antagonizes ubiquitination and degradation of the TGF-β type II receptor. Mol Cell. 2013;49(3):499-510. [22] Xie WL, Jiang R, Shen XL, et al. Diosgenin attenuates hepatic stellate cell activation through transforming growth factor-β/Smad signaling pathway. Int J Clin Exp Med. 2015; 8(11):20323-20329. [23] Wang Z, Fei S, Suo C, et al. Antifibrotic Effects of Hepatocyte Growth Factor on Endothelial-to-Mesenchymal Transition via Transforming Growth Factor-Beta1 (TGF-β1)/Smad and Akt/mTOR/P70S6K Signaling Pathways. Ann Transplant. 2018;23:1-10. [24] You SP, Ma L, Zhao J, et al. Phenylethanol Glycosides from Cistanche tubulosa Suppress Hepatic Stellate Cell Activation and Block the Conduction of Signaling Pathways in TGF-β1/smad as Potential Anti-Hepatic Fibrosis Agents. Molecules. 2016;21(1):102. [25] Feng XH, Derynck R. Specificity and versatility in tgf-beta signaling through Smads. Annu Rev Cell Dev Biol. 2005;21: 659-693. [26] Massagué J, Seoane J, Wotton D. Smad transcription factors. Genes Dev. 2005;19(23):2783-2810. [27] Ikushima H, Miyazono K. TGFbeta signalling: a complex web in cancer progression. Nat Rev Cancer. 2010;10(6):415-424. [28] Carli C, Giroux M, Delisle JS. Roles of transforming growth factor-β in graft-versus-host and graft-versus-tumor effects. Biol Blood Marrow Transplant. 2012;18(9):1329-1340. [29] Lee MKS, Al-Sharea A, Dragoljevic D, et al. Hand of FATe: lipid metabolism in hematopoietic stem cells. Curr Opin Lipidol. 2018;29(3):240-245. [30] Sugimura R. Bioengineering Hematopoietic Stem Cell Niche toward Regenerative Medicine. Adv Drug Deliv Rev.2016; 99(Pt B):212-220. [31] Bari S, Seah KK, Poon Z, et al. Expansion and homing of umbilical cord blood hematopoietic stem and progenitor cells for clinical transplantation. Biol Blood Marrow Transplant. 2015;21(6):1008-1019. [32] Fares I, Rivest-Khan L, Cohen S, et al. Small molecule regulation of normal and leukemic stem cells. Curr Opin Hematol. 2015;22(4):309-316. [33] Sitnicka E, Ruscetti FW, Priestley GV, et al. Transforming growth factor beta 1 directly and reversibly inhibits the initial cell divisions of long-term repopulating hematopoietic stem cells. Blood. 1996;88(1):82-88. [34] Hao S, Chen C, Cheng T. Cell cycle regulation of hematopoietic stem or progenitor cells. Int J Hematol. 2016; 103(5):487-497. |
.jpg)
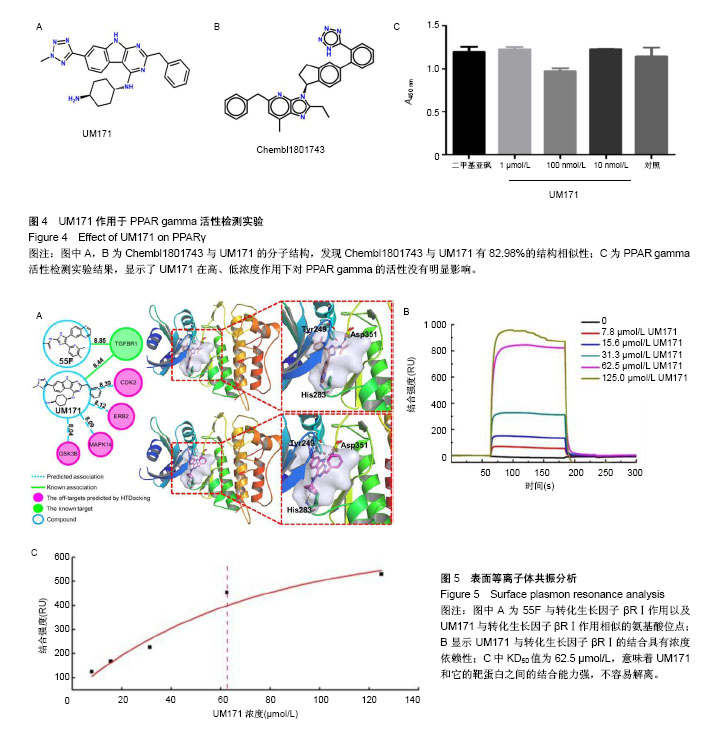
.jpg)