中国组织工程研究 ›› 2018, Vol. 22 ›› Issue (17): 2761-2768.doi: 10.3969/j.issn.2095-4344.0501
• 干细胞综述 stem cell review • 上一篇 下一篇
力学因素对间充质干细胞神经向分化的影响
江静怡1,2,樊瑜波1,2,郑丽沙1,2
- 1北京航空航天大学生物与医学工程学院,生物力学与力生物学教育部重点实验室,北京市 100083;2北京航空航天大学生物医学工程高精尖创新中心,北京市 100083
-
修回日期:2018-01-13出版日期:2018-06-18发布日期:2018-06-18 -
通讯作者:郑丽沙,博士,副教授,硕士生导师,北京航空航天大学生物与医学工程学院,生物力学与力生物学教育部重点实验室,北京市 100083;北京航空航天大学生物医学工程高精尖创新中心,北京市 100083 -
作者简介:江静怡,女,1993年生,江苏省南通市人,汉族,北京航空航天大学生物与医学工程学院在读硕士,主要从事力学生物学、组织工程基础研究。 -
基金资助:国家自然科学基金(11572030,61227902,11120101001,11421202);科技部国家重点研发计划(2017YFC0108505,2017YFC0108500)
Effects of mechanical stresses on neural differentiation of mesenchymal stem cells
Jiang Jing-yi1, 2, Fan Yu-bo1, 2, Zheng Li-sha1, 2
- 1Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China; 2Beijing Advanced Innovation Centre for Biomedical Engineering, Beihang University, Beijing 100083, China
-
Revised:2018-01-13Online:2018-06-18Published:2018-06-18 -
Contact:Zheng Li-sha, Ph.D., Associate professor, Master’s supervisor, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China; Beijing Advanced Innovation Centre for Biomedical Engineering, Beihang University, Beijing 100083, China -
About author:Jiang Jing-yi, Master candidate, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China; Beijing Advanced Innovation Centre for Biomedical Engineering, Beihang University, Beijing 100083, China -
Supported by:the National Natural Science Foundation of China, No. 11572030, 61227902, 11120101001, 11421202; the National Key R&D Program of China, No. 2017YFC0108505, 2017YFC0108500
摘要:
文章快速阅读:
.jpg)
文题释义: 细胞外基质调控间充质干细胞分化:在生物体内,细胞并不是孤立存在的,而是存在于其周围一定的微环境中,这个微环境包括其周围的基质细胞、细胞间质、体液以及各种细胞因子和物理因素等。细胞的微环境也被称为细胞生存的细胞外基质,越来越多的证据证明这些物理和化学因素共同影响细胞的命运。因此,通过改变细胞外基质的物理环境来调控干细胞的生物学行为,已经成为组织工程和再生医学研究的热点领域。 力学因素:是调节间充质干细胞生物学行为的一种重要因素,存在于一切细胞微环境中。学者们利用生物反应器在体外模拟体内多种力学环境,研究力学刺激对干细胞生物学行为的影响。如:利用平行平板流动腔和径向流装置等模拟流动剪切力、Flexer-cell加载系统模拟牵张力、水凝胶和聚二甲基硅氧烷作为基底模拟细胞外基质的不同硬度和拓扑结构等。适当的力学因素结合各种化学分化诱导因子等可以影响干细胞的分化方向,对组织工程具有重要的意义。
中图分类号:
引用本文
江静怡,樊瑜波,郑丽沙. 力学因素对间充质干细胞神经向分化的影响[J]. 中国组织工程研究, 2018, 22(17): 2761-2768.
Jiang Jing-yi, Fan Yu-bo, Zheng Li-sha. Effects of mechanical stresses on neural differentiation of mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(17): 2761-2768.
2.2 细胞外基质调控间充质干细胞神经向分化 在生物体内,细胞并不是孤立存在的,而是存在于其周围一定的微环境中,这个微环境包括其周围的基质细胞、细胞间质、体液以及各种细胞因子和物理因素等[45-46]。细胞的微环境也被称为细胞生存的细胞外基质[47],越来越多的证据证明这些物理和化学因素共同影响细胞的命运[48-52]。因此,通过改变细胞外基质的物理环境来调控干细胞的生物学行为,已经成为组织工程和再生医学研究的热点领域。细胞在体内和体外的微环境条件,见图1[53]。
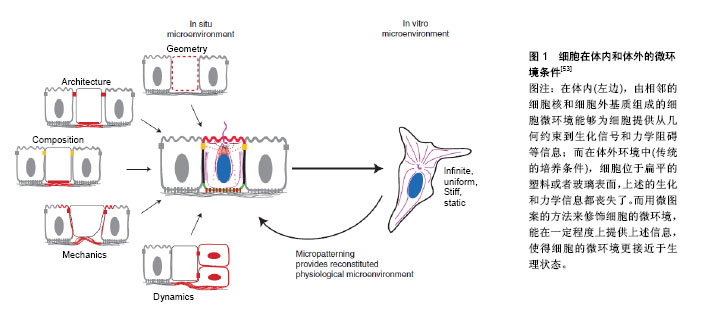
2.2.1 细胞外基质的硬度 在生物体内,每一种组织和器官都有自己独特的“硬度特征”,因此机体内的细胞都处于不同的细胞外基质硬度条件下[54]。细胞外基质的硬度作为一种力学性质,对细胞的多种生物学行为如黏附、形态、增殖、分化、凋亡和迁移等都有重要的影响[55-59]。特别是干细胞在不同硬度的基底上生长时对应不同的分化方向,见表1。研究表明,细胞在与不同组织相对应硬度的基底上生长时趋向于向相应组织方向分化[60-61]。

| [1] Manchineella S, Thrivikraman G, Basu B, et al. Surface-Functionalized Silk Fibroin Films as a Platform To Guide Neuron-like Differentiation of Human Mesenchymal Stem Cells. ACS Appl Mater Interfaces. 2016;8(35):22849-22859.[2] Zhang J, Li D, Shen A, et al. Expression of RBMX after spinal cord injury in rats. J Mol Neurosci. 2013;49(2):417-429.[3] Burdick JA, Ward M, Liang E, et al. Stimulation of neurite outgrowth by neurotrophins delivered from degradable hydrogels. Biomaterials. 2006;27(3):452-459.[4] Shi W, Nie D, Jin G, et al. BDNF blended chitosan scaffolds for human umbilical cord MSC transplants in traumatic brain injury therapy. Biomaterials. 2012;33(11):3119-3126.[5] Wichterle H, Lieberam I, Porter JA, et al. Directed differentiation of embryonic stem cells into motor neurons. Cell. 2002;110(3): 385-397.[6] Nishikawa S, Jakt LM, Era T. Embryonic stem-cell culture as a tool for developmental cell biology. Nat Rev Mol Cell Biol. 2007;8(6):502-507.[7] Wang TT, Tio M, Lee W, et al. Neural differentiation of mesenchymal-like stem cells from cord blood is mediated by PKA. Biochem Biophys Res Commun. 2007;357(4):1021-1027.[8] Caiazzo M, Dell'Anno MT, Dvoretskova E, et al. Direct generation of functional dopaminergic neurons from mouse and human fibroblasts. Nature. 2011;476(7359):224-227.[9] Han DW, Tapia N, Hermann A, et al. Direct reprogramming of fibroblasts into neural stem cells by defined factors. Cell Stem Cell. 2012;10(4):465-472.[10] Zuba-Surma EK, Kucia M, Ratajczak J, et al. "Small stem cells" in adult tissues: very small embryonic-like stem cells stand up. Cytometry A. 2009;75(1):4-13.[11] Pereira Lda V. The importance of the use of stem cells for public health. Cien Saude Colet. 2008;13(1):7-14.[12] Lu P, Tuszynski MH. Can bone marrow-derived stem cells differentiate into functional neurons. Exp Neurol. 2005;193(2):273-278.[13] Woodbury D, Schwarz EJ, Prockop DJ, et al. Adult rat and human bone marrow stromal cells differentiate into neurons. J Neurosci Res. 2000;61(4):364-370.[14] Prockop DJ, Azizi SA, Colter D, et al. Potential use of stem cells from bone marrow to repair the extracellular matrix and the central nervous system. Biochem Soc Trans. 2000;28(4):341-345.[15] Zhong W, Tian K, Zheng X, et al. Mesenchymal stem cell and chondrocyte fates in a multishear microdevice are regulated by Yes-associated protein. Stem Cells Dev. 2013;22(14):2083-2093.[16] Ward DF Jr, Salasznyk RM, Klees RF, et al. Mechanical strain enhances extracellular matrix-induced gene focusing and promotes osteogenic differentiation of human mesenchymal stem cells through an extracellular-related kinase-dependent pathway. Stem Cells Dev. 2007;16(3):467-480.[17] Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663-676.[18] Engler AJ, Sen S, Sweeney HL, et al. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126(4):677-689.[19] Lee HJ, Ewere A, Diaz MF, et al. TAZ responds to fluid shear stress to regulate the cell cycle. Cell Cycle. 2017:1-21.[20] Sera T, Sumii T, Fujita R, et al. Effect of shear stress on the migration of hepatic stellate cells. In Vitro Cell Dev Biol Anim. 2018;54(1):11-22.[21] Nishii K, Brodin E, Renshaw T, et al. Shear stress upregulates regeneration-related immediate early genes in liver progenitors in 3D ECM-like microenvironments. J Cell Physiol. 2017 Oct 20. doi: 10.1002/jcp.26246. [Epub ahead of print][22] Kapur S, Baylink DJ, Lau KH. et al. Fluid flow shear stress stimulates human osteoblast proliferation and differentiation through multiple interacting and competing signal transduction pathways. Bone. 2003;32(3):241-251.[23] Jeong SI, Kwon JH, Lim JI, et al. Mechano-active tissue engineering of vascular smooth muscle using pulsatile perfusion bioreactors and elastic PLCL scaffolds. Biomaterials. 2005;26(12): 1405-1411.[24] Kim MS, Yeon JH, Park JK. A microfluidic platform for 3-dimensional cell culture and cell-based assays. Biomed Microdevices. 2007;9(1):25-34.[25] Gronthos S, Franklin DM, Leddy HA, et al. Surface protein characterization of human adipose tissue-derived stromal cells. J Cell Physiol. 2001;189(1):54-63.[26] Bunnell BA, Flaat M, Gagliardi C, et al. Adipose-derived stem cells: isolation, expansion and differentiation. Methods. 2008; 45(2):115-120.[27] Choi J, Kim S, Jung J, et al. Wnt5a-mediating neurogenesis of human adipose tissue-derived stem cells in a 3D microfluidic cell culture system. Biomaterials. 2011;32(29):7013-7022.[28] Jeon KJ, Park SH, Shin JW, et al. Combined effects of flow-induced shear stress and micropatterned surface morphology on neuronal differentiation of human mesenchymal stem cells. J Biosci Bioeng. 2014;117(2):242-247. [29] Mascotte-Cruz JU, Ríos A, Escalante B. Combined effects of flow-induced shear stress and electromagnetic field on neural differentiation of mesenchymal stem cells. Electromagn Biol Med. 2016;35(2):161-166.[30] Basso N, Bellows CG, Heersche JN. Effect of simulated weightlessness on osteoprogenitor cell number and proliferation in young and adult rats. Bone. 2005;36(1):173-183.[31] Grimm D, Bauer J, Kossmehl P, et al. Simulated microgravity alters differentiation and increases apoptosis in human follicular thyroid carcinoma cells. FASEB J. 2002;16(6):604-606.[32] Dai ZQ, Wang R, Ling SK, et al. Simulated microgravity inhibits the proliferation and osteogenesis of rat bone marrow mesenchymal stem cells. Cell Prolif. 2007;40(5):671-684.[33] Hochleitner B, Hengster P, Duo L, et al. A novel bioartificial liver with culture of porcine hepatocyte aggregates under simulated microgravity. Artif Organs. 2005;29(1):58-66.[34] Marquette ML, Byerly D, Sognier M. A novel in vitro three-dimensional skeletal muscle model. In Vitro Cell Dev Biol Anim. 2007;43(7):255-263.[35] Wang N, Wang H, Chen J, et al. The simulated microgravity enhances multipotential differentiation capacity of bone marrow mesenchymal stem cells. Cytotechnology. 2014;66(1):119-131.[36] Xue L, Li Y, Chen J. Duration of simulated microgravity affects the differentiation of mesenchymal stem cells. Mol Med Rep. 2017; 15(5):3011-3018.[37] Chen J, Liu R, Yang Y, et al. The simulated microgravity enhances the differentiation of mesenchymal stem cells into neurons. Neurosci Lett. 2011;505(2):171-175.[38] Zarrinpour V, Hajebrahimi Z, Jafarinia M. Expression pattern of neurotrophins and their receptors during neuronal differentiation of adipose-derived stem cells in simulated microgravity condition. Iran J Basic Med Sci. 2017;20(2):178-186.[39] Park JS, Chu JS, Cheng C, et al. Differential effects of equiaxial and uniaxial strain on mesenchymal stem cells. Biotechnol Bioeng. 2004;88(3):359-368.[40] Chen YJ, Huang CH, Lee IC, et al. Effects of cyclic mechanical stretching on the mRNA expression of tendon/ligament-related and osteoblast-specific genes in human mesenchymal stem cells. Connect Tissue Res. 2008;49(1):7-14.[41] Huang CH, Chen MH, Young TH, et al. Interactive effects of mechanical stretching and extracellular matrix proteins on initiating osteogenic differentiation of human mesenchymal stem cells. J Cell Biochem. 2009;108(6):1263-1273.[42] Leong WS, Wu SC, Pal M, et al. Cyclic tensile loading regulates human mesenchymal stem cell differentiation into neuron-like phenotype. J Tissue Eng Regen Med. 2012;6 Suppl 3:s68-79.[43] Cho H, Seo YK, Jeon S, et al. Neural differentiation of umbilical cord mesenchymal stem cells by sub-sonic vibration. Life Sci. 2012;90(15-16):591-599.[44] Choi YK, Cho H, Seo YK, et al. Stimulation of sub-sonic vibration promotes the differentiation of adipose tissue-derived mesenchymal stem cells into neural cells. Life Sci. 2012;91(9-10):329-337.[45] Discher DE, Janmey P, Wang YL. Tissue cells feel and respond to the stiffness of their substrate. Science. 2005;310(5751):1139-1143.[46] Yim EK, Pang SW, Leong KW. Synthetic nanostructures inducing differentiation of human mesenchymal stem cells into neuronal lineage. Exp Cell Res. 2007;313(9):1820-1829.[47] Hynes RO. The extracellular matrix: not just pretty fibrils. Science. 2009;326(5957):1216-1219.[48] Geiger B, Spatz JP, Bershadsky AD. Environmental sensing through focal adhesions. Nat Rev Mol Cell Biol. 2009;10(1):21-33.[49] Wang N, Tytell JD, Ingber DE. Mechanotransduction at a distance: mechanically coupling the extracellular matrix with the nucleus. Nat Rev Mol Cell Biol. 2009;10(1):75-82.[50] Vogel V, Sheetz M. Local force and geometry sensing regulate cell functions. Nat Rev Mol Cell Biol. 2006;7(4):265-275.[51] Dalby MJ, Gadegaard N, Tare R, et al. The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder. Nat Mater. 2007;6(12):997-1003.[52] Janmey PA, Winer JP, Murray ME, et al. The hard life of soft cells. Cell Motil Cytoskeleton. 2009;66(8):597-605.[53] Théry M. Micropatterning as a tool to decipher cell morphogenesis and functions. J Cell Sci. 2010;123(Pt 24):4201-4213.[54] Higuchi A, Ling QD, Chang Y, et al. Physical cues of biomaterials guide stem cell differentiation fate. Chem Rev. 2013;113(5): 3297-3328.[55] Seiler C, Davuluri G, Abrams J, et al. Smooth muscle tension induces invasive remodeling of the zebrafish intestine. PLoS Biol. 2012;10(9):e1001386.[56] Butcher DT, Alliston T, Weaver VM. A tense situation: forcing tumour progression. Nat Rev Cancer. 2009;9(2):108-122.[57] Klein EA, Yin L, Kothapalli D, et al. Cell-cycle control by physiological matrix elasticity and in vivo tissue stiffening. Curr Biol. 2009;19(18):1511-1518.[58] Zaman MH, Trapani LM, Sieminski AL, et al. Migration of tumor cells in 3D matrices is governed by matrix stiffness along with cell-matrix adhesion and proteolysis. Proc Natl Acad Sci U S A. 2006;103(29):10889-10894.[59] Raab M, Swift J, Dingal PC, et al. Crawling from soft to stiff matrix polarizes the cytoskeleton and phosphoregulates myosin-II heavy chain. J Cell Biol. 2012;199(4):669-683.[60] Flanagan LA, Ju YE, Marg B, et al. Neurite branching on deformable substrates. Neuroreport. 2002;13(18):2411-2415.[61] Discher DE, Mooney DJ, Zandstra PW. Growth factors, matrices, and forces combine and control stem cells. Science. 2009;324 (5935):1673-1677.[62] Kong HJ, Polte TR, Alsberg E, et al. FRET measurements of cell-traction forces and nano-scale clustering of adhesion ligands varied by substrate stiffness. Proc Natl Acad Sci U S A. 2005; 102(12):4300-4305.[63] Rowlands AS, George PA, Cooper-White JJ. Directing osteogenic and myogenic differentiation of MSCs: interplay of stiffness and adhesive ligand presentation. Am J Physiol Cell Physiol. 2008; 295(4):C1037-1044.[64] Saha K, Keung AJ, Irwin EF, et al. Substrate modulus directs neural stem cell behavior. Biophys J. 2008;95(9):4426-4438.[65] Engler AJ, Griffin MA, Sen S, et al. Myotubes differentiate optimally on substrates with tissue-like stiffness: pathological implications for soft or stiff microenvironments. J Cell Biol. 2004; 166(6):877-887.[66] Wang LS, Chung JE, Chan PP, et al. Injectable biodegradable hydrogels with tunable mechanical properties for the stimulation of neurogenesic differentiation of human mesenchymal stem cells in 3D culture. Biomaterials. 2010;31(6):1148-1157. [67] Zhang QY, Zhang YY, Xie J, et al. Stiff substrates enhance cultured neuronal network activity. Sci Rep. 2014;4:6215.[68] Kostic A, Sap J, Sheetz MP. RPTPalpha is required for rigidity-dependent inhibition of extension and differentiation of hippocampal neurons. J Cell Sci. 2007;120(Pt 21):3895-3904.[69] Sun Y, Yong KM, Villa-Diaz LG, et al. Hippo/YAP-mediated rigidity-dependent motor neuron differentiation of human pluripotent stem cells. Nat Mater. 2014;13(6):599-604.[70] Leong WS, Tay CY, Yu H, et al. Thickness sensing of hMSCs on collagen gel directs stem cell fate. Biochem Biophys Res Commun. 2010;401(2):287-292.[71] Jiang L, Sun Z, Chen X, et al. Cells Sensing Mechanical Cues: Stiffness Influences the Lifetime of Cell-Extracellular Matrix Interactions by Affecting the Loading Rate. ACS Nano. 2016;10(1): 207-217.[72] Du J, Chen X, Liang X, et al. Integrin activation and internalization on soft ECM as a mechanism of induction of stem cell differentiation by ECM elasticity. Proc Natl Acad Sci U S A. 2011; 108(23):9466-9471.[73] Shih YR, Tseng KF, Lai HY, et al. Matrix stiffness regulation of integrin-mediated mechanotransduction during osteogenic differentiation of human mesenchymal stem cells. J Bone Miner Res. 2011;26(4):730-738.[74] Provenzano PP, Inman DR, Eliceiri KW, et al. Matrix density-induced mechanoregulation of breast cell phenotype, signaling and gene expression through a FAK-ERK linkage. Oncogene. 2009;28(49):4326-4343.[75] Alexanian AR, Liu QS, Zhang Z. Enhancing the efficiency of direct reprogramming of human mesenchymal stem cells into mature neuronal-like cells with the combination of small molecule modulators of chromatin modifying enzymes, SMAD signaling and cyclic adenosine monophosphate levels. Int J Biochem Cell Biol. 2013;45(8):1633-1638.[76] Zouani OF, Kalisky J, Ibarboure E, et al. Effect of BMP-2 from matrices of different stiffnesses for the modulation of stem cell fate. Biomaterials. 2013;34(9):2157-2166.[77] Lim SH, Liu XY, Song H, et al. The effect of nanofiber-guided cell alignment on the preferential differentiation of neural stem cells. Biomaterials. 2010;31(34):9031-9039.[78] Georgiou M, Golding JP, Loughlin AJ, et al. Engineered neural tissue with aligned, differentiated adipose-derived stem cells promotes peripheral nerve regeneration across a critical sized defect in rat sciatic nerve. Biomaterials. 2015;37:242-251.[79] Dalby MJ, McCloy D, Robertson M, et al. Osteoprogenitor response to semi-ordered and random nanotopographies. Biomaterials. 2006;27(15):2980-2987.[80] Silva GA, Czeisler C, Niece KL, et al. Selective differentiation of neural progenitor cells by high-epitope density nanofibers. Science. 2004;303(5662):1352-1355.[81] Curtis A, Wilkinson C. Nantotechniques and approaches in biotechnology. Trends Biotechnol. 2001;19(3):97-101.[82] McBeath R, Pirone DM, Nelson CM, et al. Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev Cell. 2004;6(4):483-495.[83] Kilian KA, Bugarija B, Lahn BT, et al. Geometric cues for directing the differentiation of mesenchymal stem cells. Proc Natl Acad Sci U S A. 2010;107(11):4872-4877.[84] Watari S, Hayashi K, Wood JA, et al. Modulation of osteogenic differentiation in hMSCs cells by submicron topographically-patterned ridges and grooves. Biomaterials. 2012; 33(1):128-136.[85] Gobaa S, Hoehnel S, Roccio M, et al. Artificial niche microarrays for probing single stem cell fate in high throughput. Nat Methods. 2011;8(11):949-955.[86] Mirzaei E, Ai J, Ebrahimi-Barough S, et al. The Differentiation of Human Endometrial Stem Cells into Neuron-Like Cells on Electrospun PAN-Derived Carbon Nanofibers with Random and Aligned Topographies. Mol Neurobiol. 2016;53(7):4798-4808.[87] Javazon EH, Beggs KJ, Flake AW. Mesenchymal stem cells: paradoxes of passaging. Exp Hematol. 2004;32(5):414-425.[88] Alavi A, Stupack DG. Cell survival in a three-dimensional matrix. Methods Enzymol. 2007;426:85-101.[89] Nakajima M, Ishimuro T, Kato K, et al. Combinatorial protein display for the cell-based screening of biomaterials that direct neural stem cell differentiation. Biomaterials. 2007;28(6): 1048-1060.[90] Liu H, Lin J, Roy K. Effect of 3D scaffold and dynamic culture condition on the global gene expression profile of mouse embryonic stem cells. Biomaterials. 2006;27(36):5978-5989.[91] Zhao F, Grayson WL, Ma T, et al. Effects of hydroxyapatite in 3-D chitosan-gelatin polymer network on human mesenchymal stem cell construct development. Biomaterials. 2006;27(9):1859-1867.[92] Markusen JF, Mason C, Hull DA, et al. Behavior of adult human mesenchymal stem cells entrapped in alginate-GRGDY beads. Tissue Eng. 2006;12(4):821-830.[93] Lutolf MP. Biomaterials: Spotlight on hydrogels. Nat Mater. 2009; 8(6):451-453.[94] Lee JH, Yu HS, Lee GS, et al. Collagen gel three-dimensional matrices combined with adhesive proteins stimulate neuronal differentiation of mesenchymal stem cells. J R Soc Interface. 2011;8(60):998-1010.[95] Gu H, Yue Z, Leong WS, et al. Control of in vitro neural differentiation of mesenchymal stem cells in 3D macroporous, cellulosic hydrogels. Regen Med. 2010;5(2):245-253.[96] Lee JH, Lee JY, Yang SH, et al. Carbon nanotube-collagen three-dimensional culture of mesenchymal stem cells promotes expression of neural phenotypes and secretion of neurotrophic factors. Acta Biomater. 2014;10(10):4425-4436.[97] Khosravizadeh Z, Razavi S, Bahramian H, et al. The beneficial effect of encapsulated human adipose-derived stem cells in alginate hydrogel on neural differentiation. J Biomed Mater Res B Appl Biomater. 2014;102(4):749-755.[98] Rao SR, Subbarayan R, Dinesh MG, et al. Differentiation of human gingival mesenchymal stem cells into neuronal lineages in 3D bioconjugated injectable protein hydrogel construct for the management of neuronal disorder. Exp Mol Med. 2016;48:e209.[99] Roozafzoon R, Lashay A, Vasei M, et al. Dental pulp stem cells differentiation into retinal ganglion-like cells in a three dimensional network. Biochem Biophys Res Commun. 2015;457(2):154-160.[100] Gugliandolo A, Diomede F, Cardelli P, et al. Transcriptomic analysis of gingival mesenchymal stem cells cultured on 3D bioprinted scaffold: A promising strategy for neuroregeneration. J Biomed Mater Res A. 2018;106(1):126-137.[101] Feng X, Lu X, Huang D, et al. 3D porous chitosan scaffolds suit survival and neural differentiation of dental pulp stem cells. Cell Mol Neurobiol. 2014;34(6):859-870.[102] Zhang J, Lu X, Feng G, et al. Chitosan scaffolds induce human dental pulp stem cells to neural differentiation: potential roles for spinal cord injury therapy. Cell Tissue Res. 2016;366(1):129-142.[103] Ghasemi-Mobarakeh L, Prabhakaran MP, Morshed M, et al. Electrospun poly(epsilon-caprolactone)/gelatin nanofibrous scaffolds for nerve tissue engineering. Biomaterials. 2008;29(34): 4532-4539.[104] Jahani H, Jalilian FA, Wu CY, et al. Controlled surface morphology and hydrophilicity of polycaprolactone toward selective differentiation of mesenchymal stem cells to neural like cells. J Biomed Mater Res A. 2015;103(5):1875-1881. |
| [1] | 蒲 锐, 陈子扬, 袁凌燕. 不同细胞来源外泌体保护心脏的特点与效应[J]. 中国组织工程研究, 2021, 25(在线): 1-. |
| [2] | 林清凡, 解一新, 陈婉清, 叶振忠, 陈幼芳. 人胎盘源间充质干细胞条件培养液可上调缺氧状态下BeWo细胞活力和紧密连接因子的表达[J]. 中国组织工程研究, 2021, 25(在线): 4970-4975. |
| [3] | 张秀梅, 翟运开, 赵 杰, 赵 萌. 类器官模型国内外数据库近10年文献研究热点分析[J]. 中国组织工程研究, 2021, 25(8): 1249-1255. |
| [4] | 王正东, 黄 娜, 陈婧娴, 郑作兵, 胡鑫宇, 李 梅, 苏 晓, 苏学森, 颜 南. 丁酸钠抑制氟中毒可诱导小胶质细胞活化及炎症因子表达增多[J]. 中国组织工程研究, 2021, 25(7): 1075-1080. |
| [5] | 汪显耀, 关亚琳, 刘忠山. 提高间充质干细胞治疗难愈性创面的策略[J]. 中国组织工程研究, 2021, 25(7): 1081-1087. |
| [6] | 万 然, 史 旭, 刘京松, 王岩松. 间充质干细胞分泌组治疗脊髓损伤的研究进展[J]. 中国组织工程研究, 2021, 25(7): 1088-1095. |
| [7] | 廖成成, 安家兴, 谭张雪, 王 倩, 刘建国. 口腔鳞状细胞癌干细胞的治疗靶点及应用前景[J]. 中国组织工程研究, 2021, 25(7): 1096-1103. |
| [8] | 谢文佳, 夏天娇, 周卿云, 刘羽佳, 顾小萍. 小胶质细胞介导神经元损伤在神经退行性疾病中的作用[J]. 中国组织工程研究, 2021, 25(7): 1109-1115. |
| [9] | 李珊珊, 郭笑霄, 尤 冉, 杨秀芬, 赵 露, 陈 曦, 王艳玲. 感光细胞替代治疗视网膜变性疾病[J]. 中国组织工程研究, 2021, 25(7): 1116-1121. |
| [10] | 焦 慧, 张一宁, 宋雨晴, 林 宇, 王秀丽. 乳腺癌类器官研究进展及临床应用前景[J]. 中国组织工程研究, 2021, 25(7): 1122-1128. |
| [11] | 王诗琦, 张金生. 中医药调控缺血缺氧微环境对骨髓间充质干细胞增殖、分化及衰老的影响[J]. 中国组织工程研究, 2021, 25(7): 1129-1134. |
| [12] | 曾燕华, 郝延磊. 许旺细胞体外培养及纯化的系统性综述[J]. 中国组织工程研究, 2021, 25(7): 1135-1141. |
| [13] | 孔德胜, 何晶晶, 冯宝峰, 郭瑞云, Asiamah Ernest Amponsah, 吕 飞, 张舒涵, 张晓琳, 马 隽, 崔慧先. 间充质干细胞修复大动物模型脊髓损伤疗效评价的Meta分析[J]. 中国组织工程研究, 2021, 25(7): 1142-1148. |
| [14] | 侯婧瑛, 于萌蕾, 郭天柱, 龙会宝, 吴 浩. 缺氧预处理激活HIF-1α/MALAT1/VEGFA通路促进骨髓间充质干细胞生存和血管再生[J]. 中国组织工程研究, 2021, 25(7): 985-990. |
| [15] | 史洋洋, 秦英飞, 吴福玲, 何 潇, 张雪静. 胎盘间充质干细胞预处理预防小鼠毛细支气管炎[J]. 中国组织工程研究, 2021, 25(7): 991-995. |
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
.jpg)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||