| [1] Myer GD, Ford KR, Hewett TE. Rationale and Clinical Techniques for Anterior Cruciate Ligament Injury Prevention Among Female Athletes. J Athl Train. 2004;39(4):352-364.[2] Weiss K, Whatman C. Biomechanics Associated with Patellofemoral Pain and ACL Injuries in Sports. Sports Med. 2015;45(9):1325-1337.[3] Yasen SK, Borton ZM, Eyre-Brook AI, et al. Clinical outcomes of anatomic, all-inside, anterior cruciate ligament (ACL) reconstruction. Knee. 2017;24(1):55-62.[4] Soon MY, Hassan A, Hui JH, et al. An analysis of soft tissue allograft anterior cruciate ligament reconstruction in a rabbit model: a short-term study of the use of mesenchymal stem cells to enhance tendon osteointegration. Am J Sports Med. 2007;35(6):962-971.[5] Li F, Jia H, Yu C. ACL reconstruction in a rabbit model using irradiated Achilles allograft seeded with mesenchymal stem cells or PDGF-B gene-transfected mesenchymal stem cells. Knee Surg Sports Traumatol Arthrosc. 2007;15(10): 1219-1227.[6] Bilic G, Zeisberger SM, Mallik AS, et al. Comparative characterization of cultured human term amnion epithelial and mesenchymal stromal cells for application in cell therapy. Cell Transplant. 2008;17(8):955-968.[7] Pan W, Wei Y, Zhou L, et al. Comparative in vivo study of injectable biomaterials combined with BMP for enhancing tendon graft osteointegration for anterior cruciate ligament reconstruction. J Orthop Res. 2011;29(7):1015-1021.[8] Engler AJ, Sen S, Sweeney HL, et al. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126(4):677-689.[9] Tedder ME, Simionescu A, Chen J, et al. Assembly and testing of stem cell-seeded layered collagen constructs for heart valve tissue engineering. Tissue Eng Part A. 2011; 17(1-2):25-36.[10] McFetridge PS, Daniel JW, Bodamyali T, et al. Preparation of porcine carotid arteries for vascular tissue engineering applications. J Biomed Mater Res A. 2004;70(2):224-234.[11] Funamoto S, Nam K, Kimura T, et al. The use of high-hydrostatic pressure treatment to decellularize blood vessels. Biomaterials. 2010;31(13):3590-3595.[12] Uygun BE, Soto-Gutierrez A, Yagi H, et al. Organ reengineering through development of a transplantable recellularized liver graft using decellularized liver matrix. Nat Med. 2010;16(7):814-820.[13] Chehelcheraghi F, Eimani H, Homayoonsadraie S, et al. Effects of Acellular Amniotic Membrane Matrix and Bone Marrow-Derived Mesenchymal Stem Cells in Improving Random Skin Flap Survival in Rats. Iran Red Crescent Med J. 2016;18(6):e25588.[14] Liu PF, Guo L, Zhao DW, et al. Study of human acellular amniotic membrane loading bone marrow mesenchymal stem cells in repair of articular cartilage defect in rabbits. Genet Mol Res. 2014;13(3):7992-8001.[15] Huang G, Ji S, Luo P, et al. Accelerated expansion of epidermal keratinocyte and improved dermal reconstruction achieved by engineered amniotic membrane. Cell Transplant. 2013;22(10):1831-1844.[16] Zhang L, Zou D, Li S, et al. An Ultra-thin Amniotic Membrane as Carrier in Corneal Epithelium Tissue-Engineering. Sci Rep. 2016;6:21021.[17] 袁丁,罗晗,黄斌,等. 人脱细胞羊膜治疗下肢静脉性溃疡的初步报告[J]. 中国修复重建外科杂志,2014,28(11):1449-1450.[18] 邹刚,李豫皖,金瑛,等. TGF-β1联合VEGF对人羊膜间充质干细胞向韧带成纤维细胞体外分化作用的研究[J]. 中国修复重建外科杂志, 2017,31(5): 582-593.[19] 方宁,张路,宋秀军,等.人羊膜间充质干细胞的分离、培养及鉴定[J]. 遵义医学院学报, 2009,32(3):234-236.[20] Bonomi A, Silini A, Vertua E, et al. Human amniotic mesenchymal stromal cells (hAMSCs) as potential vehicles for drug delivery in cancer therapy: an in vitro study. Stem Cell Res Ther. 2015;6:155.[21] 金瑛,李豫皖,张承昊,等.体外诱导人羊膜间充质干细胞向韧带细胞分化的实验研究[J]. 中国修复重建外科杂志, 2016,30(2): 237-244.[22] Miranda-Sayago JM, Fernandez-Arcas N, Reyes-Engel A, et al. Changes in CDKN2D , TP53, and miR125a expression: potential role in the evaluation of human amniotic fluid-derived mesenchymal stromal cell fitness. Genes Cells. 2012;17(8):673-687.[23] Rutka JT. Expression of Concern: Dual regeneration of muscle and nerve by intravenous administration of human amniotic fluid-derived mesenchymal stem cells regulated by stromal cell-derived factor-1α in a sciatic nerve injury model. J Neurosurg. 2015;123(6):1605.[24] Mano JF, Silva GA, Azevedo HS, et al. Natural origin biodegradable systems in tissue engineering and regenerative medicine: present status and some moving trends. J R Soc Interface. 2007;4(17):999-1030.[25] Riau AK, Beuerman RW, Lim LS, et al. Preservation, sterilization and de-epithelialization of human amniotic membrane for use in ocular surface reconstruction. Biomaterials. 2010;31(2):216-225.[26] 鞠晓军,潘锋,柏树令,等. 人脱细胞羊膜复合脂肪源性干细胞修复大鼠全层皮肤缺损的实验研究[J]. 中国修复重建外科杂志, 2010,24(2): 143-149.[27] Mobini S, Solati-Hashjin M, Peirovi H, et al. Bioactivity and Biocompatibility Studies on Silk-Based Scaffold for Bone Tissue Engineering. J Med Biol Eng. 2013;33(2):207-213.[28] Nigam R, Mahanta B. An Overview of Various Biomimetic Scaffolds: Challenges and Applications in Tissue Engineering. J Tissue Sci Eng.2014;5(2):137.[29] Chua KH, Raduan F, Wan Safwani WK, et al. Effects of serum reduction and VEGF supplementation on angiogenic potential of human adipose stromal cells in vitro. Cell Prolif. 2013;46(3): 300-311.[30] Koizumi N, Fullwood NJ, Bairaktaris G, et al. Cultivation of corneal epithelial cells on intact and denuded human amniotic membrane. Invest Ophthalmol Vis Sci. 2000;41(9): 2506-2513. |
.jpg)
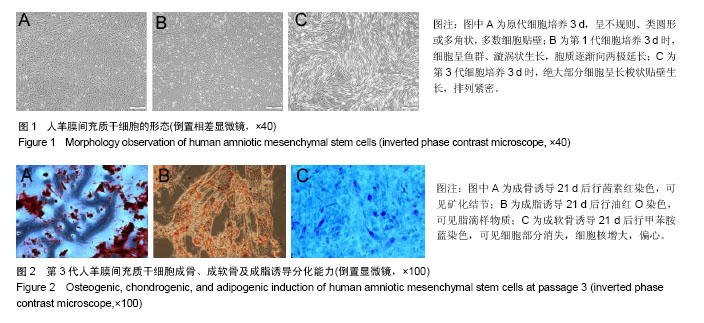

.jpg)