中国组织工程研究 ›› 2024, Vol. 28 ›› Issue (10): 1599-1605.doi: 10.12307/2024.314
• 生物材料综述 biomaterial review • 上一篇 下一篇
人工生物材料在关节软骨损伤修复中的应用
张本妥,杨 昕
- 北京大学第一医院,北京市 100034
-
收稿日期:2023-03-16接受日期:2023-05-27出版日期:2024-04-08发布日期:2023-08-21 -
通讯作者:杨昕,副主任医师,副教授,硕士生导师,北京大学第一医院,北京市 100034 -
作者简介:张本妥,男,2001年生,江西省上饶市人,汉族,北京大学医学部本科在读,主要从事软骨损伤修复研究。
Application of synthetic and biological materials in articular cartilage repair
Zhang Bentuo, Yang Xin
- First Hospital, Peking University, Beijing 100034, China
-
Received:2023-03-16Accepted:2023-05-27Online:2024-04-08Published:2023-08-21 -
Contact:Yang Xin, Associate chief physician, Associate professor, Master’s supervisor, First Hospital, Peking University, Beijing 100034, China -
About author:Zhang Bentuo, First Hospital, Peking University, Beijing 100034, China
摘要:
文题释义:
软骨组织工程:是指将种子细胞植入模拟软骨细胞外基质结构的凝胶/支架中,并将支架材料移植在缺损软骨部位,在生长因子作用下使种子细胞增殖分化、合成并分泌细胞外基质,以达到软骨修复的目的。交联:是指通过物理和化学(如光、热、超声波、加入交联剂等)方法使支架中线性高分子聚合物通过共价键连接形成网状高分子聚合物,从而改善支架的机械性能。
背景:关节软骨损伤修复是临床上亟待解决的难题,利用人工生物组织工程材料促进软骨再生修复是目前的研究热点之一。
目的:对人工生物材料在关节软骨损伤修复中应用的研究进展进行综述。方法:应用计算机检索 PubMed 数据库和中国知网收录的有关人工生物材料在关节软骨损伤修复中应用的研究,英文检索词为“collagen,gelatin,silk,chitosan,alginate,PEG,PCL,PLA,cartilage tissue engineering,cartilage tissue engineering materials”,中文检索词为“胶原,明胶,丝素蛋白,壳聚糖,海藻酸,聚乙二醇,聚已内酯,聚乳酸,软骨组织工程,软骨组织工程材料”。根据纳入与排除标准对所有文章进行初筛后,保留质量和相关性较高的98篇文献进行综述。
结果与结论:天然材料包括胶原、明胶、丝素蛋白、壳聚糖、海藻酸等,具有良好的生物相容性、降解性;合成材料包括聚乙二醇、聚已内酯、聚乳酸等,具有良好的机械性能。材料的改性与复合能够弥补材料本身的缺陷,表现出更优秀的软骨修复能力。基于分层结构合成的多层支架在研究中相对少见,且该支架更多针对骨软骨损伤修复而非单纯的软骨损伤修复。目前支架研究集中在合成研发阶段,相应的临床试验较少,未来需要注重临床应用转化。
https://orcid.org/0000-0003-3781-2742(张本妥)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
中图分类号:
引用本文
张本妥, 杨 昕. 人工生物材料在关节软骨损伤修复中的应用[J]. 中国组织工程研究, 2024, 28(10): 1599-1605.
Zhang Bentuo, Yang Xin. Application of synthetic and biological materials in articular cartilage repair[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(10): 1599-1605.
2.1.1 蛋白质
(1)胶原:作为细胞外基质的骨架结构,胶原是一种不溶性纤维蛋白,占人体蛋白质总量的25%以上。根据氨基酸序列及空间结构将胶原可分为原纤维性胶原、原纤维相关胶原、成网胶原、短链胶原以及其他类胶原,原纤维性胶原中的Ⅰ、Ⅱ型胶原作为关节软骨外基质的主要成分,是理想的软骨修复材料[9]。
Ⅱ型胶原是软骨细胞外基质中胶原的主要成分,更能模拟天然软骨细胞外基质成分。相较于Ⅰ型胶原纤维,Ⅱ型胶原纤维细小、具有更多的带电和疏水残基,这些残基更多参与分子内相互作用,缺乏分子间相互作用,在形成物理交联水凝胶时表现出较差的力学特性[10]。但单纯Ⅰ型胶原凝胶诱导软骨损伤修复的效果并不理想,在Ⅰ型胶原凝胶中加入Ⅱ型胶原会改变凝胶的特性,相较于Ⅰ型胶原∶Ⅱ型胶原=1∶1,在Ⅰ型胶原∶Ⅱ型胶原=3∶1时能够结合并保留较多的硫酸软骨素、模拟软骨成分,使得关节软骨能够承受更大的压缩力,呈现出更低的孔隙率以及更高的储存、损耗和复合模量[11]。Ⅰ/Ⅱ型胶原凝胶能诱导并维持骨髓间充质干细胞向软骨细胞分化,使糖胺聚糖合成增多,在兔软骨损伤模型具有更好的软骨修复效果[10]。在胶原凝胶中加入相关生长因子能够增强细胞的增殖分化能力,但植入细胞常倾向于分化为成熟的肥大软骨细胞,并非形成正常软骨组织。QI等[12]在在胶原水凝胶中加入Wnt5a,在促进骨髓间充质干细胞和软骨细胞的增殖、迁移和软骨分化的同时抑制软骨细胞肥大。以上研究大多针对单纯的软骨损伤,而针对骨软骨损伤,MATSUSHITA等[13]设计了胶原-软骨碎片混合凝胶,体外实验显示其表面的软骨细胞具有良好的迁移和增殖能力,体内兔模型实验显示其具有较好的骨软骨修复能力。此外,CAO等[14]根据骨软骨分层结构构建了表层为Ⅱ型胶原和硫酸软骨素、中间层为Ⅱ型胶原和羟基磷灰石、底层为Ⅰ型胶原和羟基磷灰石的3层支架,用于骨软骨损伤的修复。
目前胶原已被广泛应用于软骨损伤修复中,在基质诱导的自体软骨细胞移植术中,通常使用Ⅰ/Ⅲ型胶原(猪源性)为原料构建生物膜接种自体软骨细胞,然后植入到软骨缺损处,多项临床试验发现其临床预后优于微骨折术[15-16]。一项多中心随机临床试验发现,基于Ⅰ型胶原凝胶的自体软骨细胞移植术能够改善患者术后功能、减轻术后疼痛[17]。KON等[18]使用Ⅰ型胶原和羟基磷灰石纳米颗粒构成的软骨层(Ⅰ型胶原)、中间层(60%Ⅰ型胶原和40%羟基磷灰石)和底层(30%Ⅰ型胶原和70%羟基磷灰石)多层复合支架治疗30例膝关节软骨及骨软骨损伤患者,2年随访的MRI成像及临床评分显示该支架具有较好的临床获益及安全性。
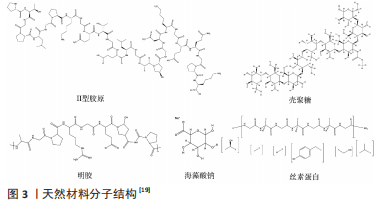
Ⅱ型胶原及其他天然材料分子结构,见图3。
(2)明胶:是胶原蛋白通过热变性或物理化学降解得到的蛋白质。不同于胶原,明胶在生物体内基本不表达抗原性、不会引发免疫反应,具有更好的生物相容性。同时,明胶保留了胶原中许多细胞黏附和信号转导的天然表位,在诱导干细胞向软骨细胞分化或维持软骨细胞表型方面起重要作用[20]。由明胶制作而成的水凝胶可作为软骨组织工程修复的支架材料,但由于明胶的凝胶温度低于体温,其在体内的稳定性不好[21],组织工程中应用的明胶支架主要是由甲基丙烯酸和明胶反应得到的甲基丙烯酰明胶。
尽管甲基丙烯酰明胶在保留明胶生物特性的基础上增强了稳定性,但在实际应用中降解速度快、高溶胀率、机械性能差仍是制约明胶作为软骨修复材料的一大难题。LIU等[22]通过在甲基丙烯酰明胶凝胶中加入甲基丙烯酸酐修饰的聚氨基胺制备甲基丙烯酰明胶/甲基丙烯酸酐修饰的聚氨基胺凝胶,显著增强了凝胶的机械稳定性。与之类似,在甲基丙烯酰明胶凝胶中混合透明质酸[20]、细菌纤维素[23]、壳聚糖[24]、聚乙二醇二丙烯酸酯等物质均能改善甲基丙烯酰明胶凝胶的高溶胀率和力学性能[25],见表1。

除在甲基丙烯酰明胶基础上进行改造外,也有研究提出其他明胶凝胶制作方法。LI等[26]利用3D打印技术,通过辣根过氧化物酶介导丝素蛋白和酪胺明胶的交联制备了大孔水凝胶支架。HAGHIGHI等[27]以壳聚糖、明胶、丝素蛋白为原料制作新型3D支架,发现当三者质量比为2∶3∶3时支架孔径大小适宜,并且支架吸水率和降解率符合软骨修复特性。
同时,明胶支架也能作为药物载体作用于软骨损伤区域。SUO等[21]将葡萄糖胺丙烯酸化为丙烯酰氨基葡萄糖,与甲基丙烯酰明胶复合形成的丙烯酰氨基葡萄糖-甲基丙烯酰明胶水凝胶,相较于纯葡萄糖胺或甲基丙烯酰明胶软骨修复效果更优异。HU等[28]研究人脐带间充质干细胞衍生小细胞外囊泡在软骨损伤修复中的作用与机制,并在此基础上研发了小细胞外囊泡缓释纳米甲基丙烯酰明胶。JIANG等[29]设计含转化生长因子β的羟磷灰石/钆纳米颗粒,将其固定在明胶海绵上构建羟磷灰石/钆纳米颗粒/明胶复合支架。YANG等[30]利用丙氨酰谷氨酰胺修饰甲基丙烯酰明胶制备甲基丙烯酰明胶-丙氨酰谷氨酰胺,在体内可降解释放谷氨酰胺激活软骨细胞能量代谢,促进软骨修复。
(3)丝素蛋白:是在蚕丝或蛛丝中分离的一种天然高分子纤维蛋白,因其良好的生物相容性、高机械性能和生物降解性可作为组织工程中常见的天然支架材料[31-32]。组织工程中使用的丝素蛋白主要来自蚕丝,蚕丝由丝素蛋白和丝胶蛋白组成,为获得纯丝素蛋白需要对蚕丝进行脱胶、冲洗、干燥等加工[33]。丝素蛋白是由390 kD重链和26 kD轻链通过二硫键连接形成的重链-轻链复合物,富含甘氨酸、丙氨酸和丝氨酸等氨基酸[34],在体内降解产生多种氨基酸供给细胞生长,也可通过氨基酸侧链修饰获得不同的性能,形成多种复合产品。
丝素蛋白凝胶的力学及降解性能由凝胶中β-折叠的含量控制[35],可使用乙醇或京尼平等交联剂或涡流和超声波等物理交联方法诱导丝素凝胶形成β-折叠结构[32,36]。ZHANG等[37]使用酶交联法将丝素蛋白与纳米硅胶盐交联,增强了凝胶的机械性能。YUAN等[32]研究出新型超声交联方法,相较于传统超声交联方法,该方法构建的丝素蛋白凝胶物理化学和生物力学性能更加优越。除交联方法外,3D打印技术也是常见的组织工程应用技术。NI等[38]利用3D打印技术构建丝素蛋白和羟丙基甲基纤维素联合双网络凝胶,丝素蛋白分子间形成的β-折叠为第一网络,丝素蛋白与羟丙基甲基纤维素交联形成第二网络,相较于单网络凝胶,该网络凝胶断裂强度、断裂伸长率和压缩再现性更高。YUAN等[32]基于3D打印技术构建甲基丙烯酰明胶+丝素蛋白-甲状旁腺素/甲基丙烯酰明胶+丝素蛋白-甲基丙烯酸酐双相支架,体内实验显示其能够促进骨软骨损伤的再生、维持透明软骨表型。此外,静电纺丝、冷冻干燥和盐浸法等方法均可用于丝素蛋白凝胶或支架的制作[39]。SHARAFAT-VAZIRI等[40]混合丝素蛋白与人皮肤源性胶原溶液设计出丝素蛋白/胶原复合支架,其溶胀率与吸水性高于单纯丝素蛋白支架,在湿环境中压缩模量下降较少,在2例软骨损伤患者中发现该复合支架治疗能够恢复膝关节软骨功能,MRI提示该支架与软骨整合良好,软骨水肿减少及软骨形成信号增强。
丝素蛋白凝胶或支架中也可加入相关药物或生长因子,增强骨髓间充质干细胞等细胞的软骨再生修复能力。CHEN等[41]构建的丹参酮ⅡA递送丝素蛋白支架可减少细胞凋亡与氧化应激。LI等[31]在混合骨形态发生蛋白2的丝素蛋白凝胶中加入包被转化生长因子β1的壳聚糖纳米颗粒,形成转化生长因子β1-壳聚糖纳米颗粒/骨形态发生蛋白2-丝素蛋白凝胶,该凝胶可增强骨髓间充质干细胞的成软骨能力。SHEN等[42]在丝素蛋白凝胶中加入骨髓间充质干细胞的缺氧预处理外泌体,发现其能够增强关节软骨细胞的增殖、迁移和抗炎能力。此外在3D打印技术中,向丝素蛋白生物油墨中加入富含多种生长因子的富血小板血浆或者脱细胞外基质[36,43],可增强支架的生物活性。
2.1.2 多糖
(1)壳聚糖:是甲壳素N-脱乙酰基的产物,和纤维素具有相似的化学结构(纤维素C2位上的羟基被氨基代替)。壳聚糖具有生物降解性、生物相容性、无毒、抑菌、降脂、增强免疫等多种生理功能,是一种具备良好应用前景的功能性生物材料[44]。壳聚糖的基本组成单位是氨基葡萄糖,其侧链上的氨基基团反应活性强,能够进行多种化学修饰形成不同的壳聚糖衍生产品以满足不同的需要[45]。
作为组织工程支架,带正电荷的壳聚糖能够和软骨细胞分泌的带负电荷的糖胺聚糖结合形成离子复合物将其固定在支架上,促进细胞的黏附和正常软骨组织的生成[46]。尽管壳聚糖在体内外实验中表现出促进软骨细胞增殖、维持正常软骨细胞表型、诱导脂肪及关节滑膜来源干细胞向软骨细胞分化能力[47-49],但存在天然材料低机械强度、生物降解快的固有特性,软骨细胞接种后易介导凝胶/支架收缩,最终会影响软骨细胞的增殖和导致软骨细胞的去分化等[50]。可使用物理/化学交联方法(如水热法,紫外线照射法、戊二醛和吉尼平交联法)等改善壳聚糖支架的物理和力学性能。YU等[51]使用邻羧甲基壳聚糖和交联剂(聚醚胺和苯-1,3,5-三羧醛反应生成)交联形成以亚氨键交联的动态水凝胶,储存模量可达到1 400 Pa,能够在不同pH值条件下能呈现出不同的溶胀率,具备自动修复性能。SHAMEKHI等[50]在水热交联制备壳聚糖支架的基础上,加入氧化石墨烯纳米颗粒使其与壳聚糖官能团发生反应,随着纳米颗粒的加入,支架的弹性模量增强,同时空隙大小和孔隙率轻微增加,而降解率不受影响,在细胞实验中发现其能促进软骨细胞增殖、维持软骨细胞形态。此外,纳米羟基磷灰石二次交联形成的壳聚糖水凝胶[48]、加入聚乳酸羟基乙酸短纤维和脱细胞外基质的壳聚糖基支架等[52],均能够改善天然生物材料的力学性能,并具备良好的生物活性。
与前面天然材料类似,在壳聚糖凝胶或支架中可以添加药物或生物活性物质增强软骨诱导能力。LI等[53]利用低聚壳聚糖包埋脂肪间充质干细胞来源细胞外囊泡,在体内外实验中发现其可逆转软骨损伤相关基因的表达。DEHGHAN-BANIANI等[54]研发小分子物质Kartogenin(KGN)和双氯芬酸钠纳米颗粒缓释的壳聚糖热凝胶,具有良好的热强度,能发挥抗炎和诱导软骨生成双重作用。此外,壳聚糖材料本身也可被设计为纳米颗粒或胶囊装载药物[55]。
目前有临床试验表明,壳聚糖-磷酸甘油凝胶支架不会引起马软骨炎症,允许植入软骨细胞合成Ⅱ型胶原与蛋白聚糖,具备良好的生物相容性[56]。此外,将壳聚糖-磷酸甘油凝胶与患者自体外周血混合,辅助微骨折术治疗软骨缺损患者,与单纯微骨折术相比,MRI显示该治疗方法软骨缺损再生好、透明软骨特征组织信号强[57],且经济评价优于单纯微骨折术[58]。
(2)海藻酸:是一类从褐藻类植物中提取出来的天然多糖产物,结构上由β-D-甘露糖醛酸和α-L-古洛糖醛酸按1,4共价键连接而成,被广泛应用于食品、药物制剂、组织工程等领域[59]。海藻酸富含羧基,在水溶液中带负电荷,具有良好的吸水性。海藻酸可与Ca2+和Sr2+等二价阳离子发生离子交换反应,构成海藻酸盐交联网络,但该海藻酸盐水凝胶易与组织中Na+、K+等单价阳离子发生交换反应,凝胶正常交联网络被破坏[60]。因此,与前文介绍的大部分天然材料类似,在支架设计时通常会对海藻酸进行化学修饰或与其他材料形成复合支架。
海藻酸盐支架具有良好的生物相容性,FILARDO等[61]制备由骨相(1.25%海藻酸盐和4%透明质酸)和软骨相(1%海藻酸盐和0.5%透明质酸)组成的双相凝胶,在骨软骨缺损修复实验中发现其不会引起在损伤组织周围的炎症细胞浸润。?ZTüRK等[62]使用硫酸化修饰海藻酸模拟天然软骨基质的高糖胺聚糖含量,诱导软骨基质沉积;使用酪氨酸酶介导与损伤软骨周围基质胶原中酪氨基团交联,增强凝胶黏附性。此外研究发现,在支架中加入纳米材料能够增强支架的机械强度。ARJMANDI等[63]在海藻酸-聚丙烯酰胺聚合网络支架中加入二氧化硅纳米颗粒,二氧化硅纳米颗粒使凝胶具备高耐磨性及低摩擦系数。与之类似,YUAN等[64]在牛血清白蛋白/海藻酸盐凝胶中掺杂羟基磷灰石纳米丝,使材料具备良好的抗压强度、韧性和抗疲劳性能。同时,海藻酸盐材料也可装载相关生长因子,SAYGILI等[65]在海藻酸盐-聚丙烯酰胺水凝胶加入转化生长因子β3纳米颗粒,体外实验显示软骨细胞活力增强,兔软骨缺损模型显示软骨修复效果增强。XU等[66]利用二甲双胍的抗衰老和锶的抗炎作用设计了二甲双胍-海藻酸锶凝胶,体外实验中发现其可以抑制软骨细胞衰老、凋亡和炎症等相关基因的表达。
一项临床试验使用同种异体软骨细胞联合海藻酸盐支架治疗21例膝关节软骨和骨软骨缺损患者,临床随访中发现,术后6个月患者的临床症状改善,术后12个月在13例患者组织活检中发现部分可再生出透明软骨[67],但12例患者的MRI成像显示部分患者出现骨髓水肿及积液[68],与患者的临床症状改善结果不一致。
2.2 合成材料
2.2.1 聚乙二醇 聚乙二醇是一种常见的高分子聚合物,化学式为HO(CH2CH2O)nH,在工业生产中主要由环氧乙烷和水或由乙二醇逐步聚合而成。聚乙二醇安全无毒,并与其他有机物具备良好的相容性,具备优良的润滑性和保湿性,是软骨组织工程最常用的聚合物之一[69]。
聚乙二醇凝胶制备过程中通常使用化学交联或光交联等方法。在化学交联中存在聚乙二醇二甲基丙烯酸酯的链式聚合和多结合位点交联剂参与的多臂聚乙二醇的非链式聚合,前者由于反应的快速动力学以及交联的随机性,所得的凝胶结构不规则、机械强度弱[70]。而根据交联密度,聚乙二醇可分为四臂和八臂、短链和长链[71]。WANG等[72]提出基于四臂聚乙二醇和乙烯砜和短二硫醇作为交联剂的4+2体系,在较低浓度下合成与天然软骨强度相当的可注射四臂聚乙二醇水凝胶,在体内实验中其可形成典型的软骨腔隙结构,并与缺损区域周围软骨组织融合。与之类似,以丙烯酸酯和降冰片烯修饰的八臂聚乙二醇和聚乙二硫醇为原料,在光引发剂和紫外光的诱导下合成可降解光点击聚乙二醇水凝胶,在软骨细胞接种培养中发现,随时间推移,聚乙二醇水凝胶降解,同时凝胶中硫酸化糖胺聚糖和胶原沉积增加、压缩模量增强[73]。
采用光或化学交联方法制备的聚乙二醇凝胶支架具备良好的性能,在体内外实验中也呈现出优秀的修复效果,而聚乙二醇与其他天然或人工材料混合形成的复合支架具备较好的生物相容性,为软骨修复提供合适的环境[74]。FU等[75]设计了聚己内酯-聚乙二醇-聚己内酯复合支架,相较于纯聚己内酯支架,该复合支架具备更好的降解率、亲水性及更低的酸性。KO等[76]使用软骨细胞和骨髓间充质干细胞作为聚己内酯-聚乙二醇-聚己内酯复合支架的种子细胞,软骨细胞诱导骨髓间充质干细胞软骨分化,骨髓间充质干细胞维持细胞增殖,在软骨细胞∶骨髓间充质干细胞=1∶4时软骨修复效果最佳。此外,ZHANG等[77]设计了一种基于丝素蛋白/聚乙二醇的可注射骨髓间充质干细胞双网络水凝胶,在整合聚乙二醇支架的弹性和丝素支架的硬度的同时,为骨髓间充质干细胞的体外增殖分化提供了良好的微环境。KIM等[78]模仿软骨正常结构,以丝素蛋白/聚乙二醇双层凝胶与低交联四臂聚乙二醇包被丝素纤维组成底层,用于生物降解和软骨细胞分化;以高交联八臂聚乙二醇组成顶层,防护机械应力,在制备凝胶过程中增加交联程度能够有效改善支架的机械强度,但高交联度会导致较低的营养交换和细胞迁移。ZHANG等[79]使用聚乙二醇和胶原形成高交联网络的同时,利用氯化钠盐析方法形成孔隙结构,改善细胞附着、增殖和细胞外基质积累。
2.2.2 聚已内酯 聚已内酯又称聚ε-己内酯,化学式为(C6H10O2)n,是一种生物相容性较好的合成高分子材料。作为组织工程支架备选材料,聚已内酯在体内与生物细胞的相容性好,细胞在支架上能够正常生长,同时具有良好的机械性能、生物降解性[80],能够在体内保留约2年,适用于软骨这种需要长期再生的组织[81]。聚已内酯也具有良好的有机高聚物溶解性,能够与多种材料结合形成多种复合物质,也是一种良好的药物载体[82]。
聚已内酯支架可由3D打印和静电纺丝技术制成。在3D打印技术中,聚已内酯的分子质量、孔几何形状及孔隙大小均对支架机械性能产生影响[83],OLUBAMIJI等[84]基于此制备出了模拟软骨结构的仿生聚已内酯支架。此外,孔径大小还影响营养渗入、氧气供给和细胞黏附等,影响最终软骨修复效果[85]:小孔径(150 μm)支架可以为骨髓间充质干细胞分化提供良好的机械刺激,而大孔径(750 μm)支架则可以通过血管生成促进营养物质的运输[85]。SUN等[86]构建一个孔径异性的3D打印支架,由浅表层(150 μm)至深层(750 μm)其孔径大小呈梯度变化,诱导区域特异性软骨及骨化、促进深层血管生成,形成类似于天然软骨基质梯度,该支架在兔软骨损伤模型中修复效果优于孔径同性聚已内酯支架。而静电纺丝技术制成的聚已内酯支架平均纤维直径在纳米范围,具备多孔隙结构,利于细胞的黏附和外基质的扩散。建立在静电纺丝技术之上的同轴静电纺丝技术,能够结合多种材料改善支架性能,同时纺丝内芯也能包裹生物因子或药物,是制作药物缓释支架的优良策略[87]。
聚已内酯材料存在低亲水性,缺乏官能团结合生长因子,细胞亲和力较差,不利于软骨修复。DABASINSKAITE等[88]在静电纺丝制备聚已内酯支架基础上,在水相中使用O3对支架进行表面改性,使支架亲水性提高,并出现能结合生长因子(如胰岛素样生长因子1)的含氧官能团。LI等[89-90]在聚已内酯支架中加入聚多巴胺进行表面改性,提高了支架的亲水性和细胞亲和力,混合负载胰岛素样生长因子1的聚乳酸-羟基乙酸共聚纳米颗粒助于细胞的增殖分化,该支架组的糖胺聚糖含量显著高于其他组别,软骨分化相关基因表达量增高。除前文所提到的聚多巴胺表面改性外,也可通过与天然材料混合来改善聚已内酯的生物性能,如加入胶原、明胶和硫酸软骨素等成分能够增强支架的机械性能和黏附力[81,91-92]。
2.2.3 聚乳酸 聚乳酸又称聚丙交酯,化学式为(C3H4O2)n,是以乳酸为原料聚合而成的聚酯类聚合物。作为软骨组织工程支架,聚乳酸的机械性能与热稳定好,但与前文的聚已内酯类似,疏水性及缺乏细胞识别信号使其在细胞植入培养方面不具备天然材料的优势。此外,聚乳酸在体内的降解速率略快于软骨再生速率,同时其酸性降解产物可能会诱发炎症[93],因此,聚乳酸材料常与天然材料结合形成复合支架以改善合成材料本身所带来的固有缺陷。
种子细胞作为组织工程中的基石之一,是软骨修复的重要因素。DING等[94]发现单纯骨髓间充质干细胞接种聚乳酸支架相较于软骨细胞接种及二者混合接种能够改善猪实验模型体内因聚乳酸降解导致的炎症反应,通过免疫荧光、基因表达分析等验证其可能是通过诱导巨噬细胞的M2极化来改善炎症反应。WAYNE等[95]报道,单纯的聚乳酸-海藻盐支架在进行犬软骨缺损修复时能够诱导缺损部位的软骨组织填充,接种骨髓间充质干细胞只能略微增强软骨修复效果,无法与正常软骨组织相媲美。因此,种子细胞和聚乳酸支架的组合不足以完成软骨组织的修复,在支架中可加入生长因子或药物成分促进细胞的增殖分化,以达到更优的软骨修复效果。有文献报道,在聚乳酸纳米纤维支架中加入基质蛋白3能够抑制软骨细胞的肥大、调节软骨基质稳态,体内外实验中均显示出其可促进骨髓间充质干细胞的软骨分化及抑制细胞肥大[96]。与之类似,在聚乳酸/明胶支架中加入具有抗衰老、抗炎、减少细胞外基质损伤的天然多酚(白藜芦醇),极大促进了软骨的再生[97]。除作为支架主体,聚乳酸材料可也作为微粒添加入其他支架中以增强支架的力学性能和稳定性,如在丝素蛋白/明胶支架中加入聚乳酸多孔微球可显著提高支架的机械强度,并实现多孔结构的多级重构,为细胞附着提供更大的表面积[98]。

| [1] HUBER M, TRATTNIG S, LINTNER F. Anatomy, biochemistry, and physiology of articular cartilage. Invest Radiol. 2000;35(10):573-580. [2] NEWMAN AP. Articular cartilage repair. Am J Sports Med. 1998;26(2):309-324. [3] ULRICH-VINTHER M, MALONEY MD, SCHWARZ EM, et al. Articular cartilage biology. J Am Acad Orthop Surg. 2003;11(6):421-430. [4] SIMON TM, JACKSON DW. Articular Cartilage: Injury Pathways and Treatment Options. Sports Med Arthrosc Rev. 2018;26(1):31-39. [5] JONES KJ, CASH BM. Matrix-Induced Autologous Chondrocyte Implantation With Autologous Bone Grafting for Osteochondral Lesions of the Femoral Trochlea. Arthrosc Tech. 2019;8(3):e259-e266. [6] DEKKER TJ, AMAN ZS, DEPHILLIPO NN, et al. Chondral Lesions of the Knee: An Evidence-Based Approach. J Bone Joint Surg Am. 2021;103(7):629-645. [7] RICHTER DL, SCHENCK RC JR, WASCHER DC, et al. Knee Articular Cartilage Repair and Restoration Techniques: A Review of the Literature. Sports Health. 2016;8(2):153-160. [8] HUNZIKER EB, LIPPUNER K, KEEL MJ, et al. An educational review of cartilage repair: precepts & practice--myths & misconceptions--progress & prospects. Osteoarthritis Cartilage. 2015;23(3):334-350. [9] RICARD-BLUM S. The collagen family. Cold Spring Harb Perspect Biol. 2011; 3(1):a004978. [10] KILMER CE, BATTISTONI CM, COX A, et al. Collagen Type I and II Blend Hydrogel with Autologous Mesenchymal Stem Cells as a Scaffold for Articular Cartilage Defect Repair. ACS Biomater Sci Eng. 2020;6(6):3464-3476. [11] VÁZQUEZ-PORTALATI NN, KILMER CE, PANITCH A, et al. Characterization of Collagen Type I and II Blended Hydrogels for Articular Cartilage Tissue Engineering. Biomacromolecules. 2016;17(10):3145-3152. [12] QI Y, ZHANG W, LI G, et al. An oriented-collagen scaffold including Wnt5a promotes osteochondral regeneration and cartilage interface integration in a rabbit model. Faseb j. 2020;34(8):11115-11132. [13] MATSUSHITA R, NAKASA T, ISHIKAWA M, et al. Repair of an Osteochondral Defect With Minced Cartilage Embedded in Atelocollagen Gel: A Rabbit Model. Am J Sports Med. 2019;47(9):2216-2224. [14] CAO R, XU Y, XU Y, et al. Development of Tri-Layered Biomimetic Atelocollagen Scaffolds with Interfaces for Osteochondral Tissue Engineering. Adv Healthc Mater. 2022;11(11):e2101643. [15] VOLZ M, SCHAUMBURGER J, FRICK H, et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop. 2017;41(4): 797-804. [16] BRITTBERG M, RECKER D, ILGENFRITZ J, et al. Matrix-Applied Characterized Autologous Cultured Chondrocytes Versus Microfracture: Five-Year Follow-up of a Prospective Randomized Trial. Am J Sports Med. 2018;46(6): 1343-1351. [17] SCHNEIDER U, RACKWITZ L, ANDEREYA S, et al. A prospective multicenter study on the outcome of type I collagen hydrogel-based autologous chondrocyte implantation (CaReS) for the repair of articular cartilage defects in the knee. Am J Sports Med. 2011;39(12):2558-2565. [18] KON E, DELCOGLIANO M, FILARDO G, et al. Novel nano-composite multilayered biomaterial for osteochondral regeneration: a pilot clinical trial. Am J Sports Med. 2011;39(6):1180-1190. [19] WEI W, MA Y, YAO X, et al. Advanced hydrogels for the repair of cartilage defects and regeneration. Bioact Mater. 2021;6(4):998-1011. [20] LIN H, BECK AM, SHIMOMURA K, et al. Optimization of photocrosslinked gelatin/hyaluronic acid hybrid scaffold for the repair of cartilage defect. J Tissue Eng Regen Med. 2019;13(8):1418-1429. [21] SUO H, LI L, ZHANG C, et al. Glucosamine-grafted methacrylated gelatin hydrogels as potential biomaterials for cartilage repair. J Biomed Mater Res B Appl Biomater. 2020;108(3):990-999. [22] LIU F, WANG X, LI Y, et al. Dendrimer-modified gelatin methacrylate hydrogels carrying adipose-derived stromal/stem cells promote cartilage regeneration. Stem Cell Res Ther. 2022;13(1):26. [23] GU L, LI T, SONG X, et al. Preparation and characterization of methacrylated gelatin/bacterial cellulose composite hydrogels for cartilage tissue engineering. Regen Biomater. 2020;7(2):195-202. [24] ZHANG W, CHEN R, XU X, et al. Construction of Biocompatible Hydrogel Scaffolds With a Long-Term Drug Release for Facilitating Cartilage Repair. Front Pharmacol. 2022;13:922032. [25] YU H, FENG M, MAO G, et al. Implementation of Photosensitive, Injectable, Interpenetrating, and Kartogenin-Modified GELMA/PEDGA Biomimetic Scaffolds to Restore Cartilage Integrity in a Full-Thickness Osteochondral Defect Model. ACS Biomater Sci Eng. 2022;8(10):4474-4485. [26] LI Q, XU S, FENG Q, et al. 3D printed silk-gelatin hydrogel scaffold with different porous structure and cell seeding strategy for cartilage regeneration. Bioact Mater. 2021;6(10):3396-3410. [27] HAGHIGHI P, SHAMLOO A. Fabrication of a novel 3D scaffold for cartilage tissue repair: In-vitro and in-vivo study. Mater Sci Eng C Mater Biol Appl. 2021;128:112285. [28] HU H, DONG L, BU Z, et al. miR-23a-3p-abundant small extracellular vesicles released from Gelma/nanoclay hydrogel for cartilage regeneration. J Extracell Vesicles. 2020;9(1):1778883. [29] JIANG X, XIU J, SHEN F, et al. Repairing of Subchondral Defect and Articular Cartilage of Knee Joint of Rabbit by Gadolinium Containing Bio-Nanocomposites. J Biomed Nanotechnol. 2021;17(8):1584-1597. [30] YANG R, ZHANG X, LIU J, et al. Functional gelatin hydrogel scaffold with degraded-release of glutamine to enhance cellular energy metabolism for cartilage repair. Int J Biol Macromol. 2022;221:923-933. [31] LI Y, LIU Y, GUO Q. Silk fibroin hydrogel scaffolds incorporated with chitosan nanoparticles repair articular cartilage defects by regulating TGF-β1 and BMP-2. Arthritis Res Ther. 2021;23(1):50. [32] YUAN T, LI Z, ZHANG Y, et al. Injectable Ultrasonication-Induced Silk Fibroin Hydrogel for Cartilage Repair and Regeneration. Tissue Eng Part A. 2021;27(17-18):1213-1224. [33] GHOLIPOURMALEKABADI M, SAPRU S, SAMADIKUCHAKSARAEI A, et al. Silk fibroin for skin injury repair: Where do things stand? Adv Drug Deliv Rev. 2020;153:28-53. [34] SUN W, GREGORY DA, TOMEH MA, et al. Silk Fibroin as a Functional Biomaterial for Tissue Engineering. Int J Mol Sci. 2021;22(3):1499. [35] RODRIGUEZ MJ, BROWN J, GIORDANO J, et al. Silk based bioinks for soft tissue reconstruction using 3-dimensional (3D) printing with in vitro and in vivo assessments. Biomaterials. 2017;117:105-115. [36] ZHANG X, LIU Y, LUO C, et al. Crosslinker-free silk/decellularized extracellular matrix porous bioink for 3D bioprinting-based cartilage tissue engineering. Mater Sci Eng C Mater Biol Appl. 2021;118:111388. [37] ZHANG W, ZHANG Y, ZHANG A, et al. Enzymatically crosslinked silk-nanosilicate reinforced hydrogel with dual-lineage bioactivity for osteochondral tissue engineering. Mater Sci Eng C Mater Biol Appl. 2021; 127:112215. [38] NI T, LIU M, ZHANG Y, et al. 3D Bioprinting of Bone Marrow Mesenchymal Stem Cell-Laden Silk Fibroin Double Network Scaffolds for Cartilage Tissue Repair. Bioconjug Chem. 2020;31(8):1938-1947. [39] ZHOU Z, CUI J, WU S, et al. Silk fibroin-based biomaterials for cartilage/osteochondral repair. Theranostics. 2022;12(11):5103-5124. [40] SHARAFAT-VAZIRI A, KHORASANI S, DARZI M, et al. Safety and efficacy of engineered tissue composed of silk fibroin/collagen and autologous chondrocytes in two patients with cartilage defects: A pilot clinical trial study. Knee. 2020;27(5):1300-1309. [41] CHEN W, XU Y, LI H, et al. Tanshinone IIA Delivery Silk Fibroin Scaffolds Significantly Enhance Articular Cartilage Defect Repairing via Promoting Cartilage Regeneration. ACS Appl Mater Interfaces. 2020;12(19):21470-21480. [42] SHEN K, DUAN A, CHENG J, et al. Exosomes derived from hypoxia preconditioned mesenchymal stem cells laden in a silk hydrogel promote cartilage regeneration via the miR-205-5p/PTEN/AKT pathway. Acta Biomater. 2022;143:173-188. [43] LI Z, ZHANG X, YUAN T, et al. Addition of Platelet-Rich Plasma to Silk Fibroin Hydrogel Bioprinting for Cartilage Regeneration. Tissue Eng Part A. 2020;26(15-16): 886-895. [44] ZHANG M, ZHANG F, LI C, et al. Application of Chitosan and Its Derivative Polymers in Clinical Medicine and Agriculture. Polymers (Basel). 2022; 14(5):958. [45] SULTANKULOV B, BERILLO D, SULTANKULOVA K, et al. Progress in the Development of Chitosan-Based Biomaterials for Tissue Engineering and Regenerative Medicine. Biomolecules. 2019;9(9):470. [46] LIN IC, WANG TJ, WU CL, et al. Chitosan-cartilage extracellular matrix hybrid scaffold induces chondrogenic differentiation to adipose-derived stem cells. Regen Ther. 2020;14:238-244. [47] ZUBILLAGA V, ALONSO-VARONA A, FERNANDES SCM, et al. Adipose-Derived Mesenchymal Stem Cell Chondrospheroids Cultured in Hypoxia and a 3D Porous Chitosan/Chitin Nanocrystal Scaffold as a Platform for Cartilage Tissue Engineering. Int J Mol Sci. 2020;21(3):1004. [48] KUMAR BYS, ISLOOR AM, KUMAR GCM, et al. Nanohydroxyapatite Reinforced Chitosan Composite Hydrogel with Tunable Mechanical and Biological Properties for Cartilage Regeneration. Sci Rep. 2019;9(1):15957. [49] LI P, FU L, LIAO Z, et al. Chitosan hydrogel/3D-printed poly(ε-caprolactone) hybrid scaffold containing synovial mesenchymal stem cells for cartilage regeneration based on tetrahedral framework nucleic acid recruitment. Biomaterials. 2021;278:121131. [50] SHAMEKHI MA, MIRZADEH H, MAHDAVI H, et al. Graphene oxide containing chitosan scaffolds for cartilage tissue engineering. Int J Biol Macromol. 2019; 127:396-405. [51] YU R, ZHANG Y, BARBOIU M, et al. Biobased pH-responsive and self-healing hydrogels prepared from O-carboxymethyl chitosan and a 3-dimensional dynamer as cartilage engineering scaffold. Carbohydr Polym. 2020;244:116471. [52] SHEN Y, XU Y, YI B, et al. Engineering a Highly Biomimetic Chitosan-Based Cartilage Scaffold by Using Short Fibers and a Cartilage-Decellularized Matrix. Biomacromolecules. 2021;22(5):2284-2297. [53] LI S, LIU J, LIU S, et al. Chitosan oligosaccharides packaged into rat adipose mesenchymal stem cells-derived extracellular vesicles facilitating cartilage injury repair and alleviating osteoarthritis. J Nanobiotechnology. 2021;19(1):343. [54] DEHGHAN-BANIANI D, MEHRJOU B, WANG D, et al. A dual functional chondro-inductive chitosan thermogel with high shear modulus and sustained drug release for cartilage tissue engineering. Int J Biol Macromol. 2022;205:638-650. [55] SHOUEIR KR, EL-DESOUKY N, RASHAD MM, et al. Chitosan based-nanoparticles and nanocapsules: Overview, physicochemical features, applications of a nanofibrous scaffold, and bioprinting. Int J Biol Macromol. 2021;167:1176-1197. [56] MARTINS EA, MICHELACCI YM, BACCARIN RY, et al. Evaluation of chitosan-GP hydrogel biocompatibility in osteochondral defects: an experimental approach. BMC Vet Res. 2014;10:197. [57] STANISH WD, MCCORMACK R, FORRIOL F, et al. Novel scaffold-based BST-CarGel treatment results in superior cartilage repair compared with microfracture in a randomized controlled trial. J Bone Joint Surg Am. 2013; 95(18):1640-1650. [58] FRAPPIER J, STANISH W, BRITTBERG M, et al. Economic evaluation of BST-CarGel as an adjunct to microfracture vs microfracture alone in knee cartilage surgery. J Med Econ. 2014;17(4):266-278. [59] LIU W, MADRY H, CUCCHIARINI M. Application of Alginate Hydrogels for Next-Generation Articular Cartilage Regeneration. Int J Mol Sci. 2022; 23(3):1147. [60] LIU J, FANG Q, LIN H, et al. Alginate-poloxamer/silk fibroin hydrogels with covalently and physically cross-linked networks for cartilage tissue engineering. Carbohydr Polym. 2020;247:116593. [61] FILARDO G, PERDISA F, GELINSKY M, et al. Novel alginate biphasic scaffold for osteochondral regeneration: an in vivo evaluation in rabbit and sheep models. J Mater Sci Mater Med. 2018;29(6):74. [62] ÖZTÜRK E, STAUBER T, LEVINSON C, et al. Tyrosinase-crosslinked, tissue adhesive and biomimetic alginate sulfate hydrogels for cartilage repair. Biomed Mater. 2020;15(4):045019. [63] ARJMANDI M, RAMEZANI M. Mechanical and tribological assessment of silica nanoparticle-alginate-polyacrylamide nanocomposite hydrogels as a cartilage replacement. J Mech Behav Biomed Mater. 2019;95:196-204. [64] YUAN H, ZHENG X, LIU W, et al. A novel bovine serum albumin and sodium alginate hydrogel scaffold doped with hydroxyapatite nanowires for cartilage defects repair. Colloids Surf B Biointerfaces. 2020;192:111041. [65] SAYGILI E, KAYA E, ILHAN-AYISIGI E, et al. An alginate-poly(acrylamide) hydrogel with TGF-β3 loaded nanoparticles for cartilage repair: Biodegradability, biocompatibility and protein adsorption. Int J Biol Macromol. 2021;172:381-393. [66] XU L, MA F, HUANG J, et al. Metformin Hydrochloride Encapsulation by Alginate Strontium Hydrogel for Cartilage Regeneration by Reliving Cellular Senescence. Biomacromolecules. 2021;22(2):671-680. [67] ALMQVIST KF, DHOLLANDER AA, VERDONK PC, et al. Treatment of cartilage defects in the knee using alginate beads containing human mature allogenic chondrocytes. Am J Sports Med. 2009;37(10):1920-1929. [68] DHOLLANDER AA, HUYSSE WC, VERDONK PC, et al. MRI evaluation of a new scaffold-based allogenic chondrocyte implantation for cartilage repair. Eur J Radiol. 2010;75(1):72-81. [69] ZARRINTAJ P, KHODADADI YAZDI M, YOUSSEFI AZARFAM M, et al. Injectable Cell-Laden Hydrogels for Tissue Engineering: Recent Advances and Future Opportunities. Tissue Eng Part A. 2021;27(11-12):821-843. [70] JIN R, MOREIRA TEIXEIRA LS, KROUWELS A, et al. Synthesis and characterization of hyaluronic acid-poly(ethylene glycol) hydrogels via Michael addition: An injectable biomaterial for cartilage repair. Acta Biomater. 2010;6(6):1968-1977. [71] BORGES FTP, PAPAVASILIOU G, TEYMOUR F. Characterizing the Molecular Architecture of Hydrogels and Crosslinked Polymer Networks beyond Flory-Rehner-I. Theory. Biomacromolecules. 2020;21(12):5104-5118. [72] WANG J, ZHANG F, TSANG WP, et al. Fabrication of injectable high strength hydrogel based on 4-arm star PEG for cartilage tissue engineering. Biomaterials. 2017;120:11-21. [73] NEUMANN AJ, QUINN T, BRYANT SJ. Nondestructive evaluation of a new hydrolytically degradable and photo-clickable PEG hydrogel for cartilage tissue engineering. Acta Biomater. 2016;39:1-11. [74] ZHAO X, PAPADOPOULOS A, IBUSUKI S, et al. Articular cartilage generation applying PEG-LA-DM/PEGDM copolymer hydrogels. BMC Musculoskelet Disord. 2016;17:245. [75] FU N, LIAO J, LIN S, et al. PCL-PEG-PCL film promotes cartilage regeneration in vivo. Cell Prolif. 2016;49(6):729-739. [76] KO CY, KU KL, YANG SR, et al. In vitro and in vivo co-culture of chondrocytes and bone marrow stem cells in photocrosslinked PCL-PEG-PCL hydrogels enhances cartilage formation. J Tissue Eng Regen Med. 2016;10(10):E485-e496. [77] ZHANG Y, CAO Y, ZHANG L, et al. Fabrication of an injectable BMSC-laden double network hydrogel based on silk fibroin/PEG for cartilage repair. J Mater Chem B. 2020;8(27):5845-5848. [78] KIM JS, CHOI J, KI CS, et al. 3D Silk Fiber Construct Embedded Dual-Layer PEG Hydrogel for Articular Cartilage Repair - In vitro Assessment. Front Bioeng Biotechnol. 2021;9:653509. [79] ZHANG J, WANG J, ZHANG H, et al. Macroporous interpenetrating network of polyethylene glycol (PEG) and gelatin for cartilage regeneration. Biomed Mater. 2016;11(3):035014. [80] MALINAUSKAS M, JANKAUSKAITE L, AUKSTIKALNE L, et al. Cartilage regeneration using improved surface electrospun bilayer polycaprolactone scaffolds loaded with transforming growth factor-beta 3 and rabbit muscle-derived stem cells. Front Bioeng Biotechnol. 2022;10:971294. [81] KIM S, GWON Y, PARK S, et al. Synergistic effects of gelatin and nanotopographical patterns on biomedical PCL patches for enhanced mechanical and adhesion properties. J Mech Behav Biomed Mater. 2021; 114:104167. [82] VENUGOPAL E, SAHANAND KS, BHATTACHARYYA A, et al. Electrospun PCL nanofibers blended with Wattakaka volubilis active phytochemicals for bone and cartilage tissue engineering. Nanomedicine. 2019;21:102044. [83] MARTÍNEZ-MORENO D, JIMÉNEZ G, CHOCARRO-WRONA C, et al. Pore geometry influences growth and cell adhesion of infrapatellar mesenchymal stem cells in biofabricated 3D thermoplastic scaffolds useful for cartilage tissue engineering. Mater Sci Eng C Mater Biol Appl. 2021;122:111933. [84] OLUBAMIJI AD, IZADIFAR Z, SI JL, et al. Modulating mechanical behaviour of 3D-printed cartilage-mimetic PCL scaffolds: influence of molecular weight and pore geometry. Biofabrication. 2016;8(2):025020. [85] DI LUCA A, SZLAZAK K, LORENZO-MOLDERO I, et al. Influencing chondrogenic differentiation of human mesenchymal stromal cells in scaffolds displaying a structural gradient in pore size. Acta Biomater. 2016;36:210-219. [86] SUN Y, WU Q, ZHANG Y, et al. 3D-bioprinted gradient-structured scaffold generates anisotropic cartilage with vascularization by pore-size-dependent activation of HIF1α/FAK signaling axis. Nanomedicine. 2021;37:102426. [87] SILVA JC, UDANGAWA RN, CHEN J, et al. Kartogenin-loaded coaxial PGS/PCL aligned nanofibers for cartilage tissue engineering. Mater Sci Eng C Mater Biol Appl. 2020;107:110291. [88] DABASINSKAITE L, KRUGLY E, BANIUKAITIENE O, et al. The Effect of Ozone Treatment on the Physicochemical Properties and Biocompatibility of Electrospun Poly(ε)caprolactone Scaffolds. Pharmaceutics. 2021;13(8):1288. [89] LI J, YAO Q, XU Y, et al. Lithium Chloride-Releasing 3D Printed Scaffold for Enhanced Cartilage Regeneration. Med Sci Monit. 2019;25:4041-4050. [90] WEI P, XU Y, GU Y, et al. IGF-1-releasing PLGA nanoparticles modified 3D printed PCL scaffolds for cartilage tissue engineering. Drug Deliv. 2020; 27(1):1106-1114. [91] JIANG T, HENG S, HUANG X, et al. Biomimetic Poly(Poly(ε-caprolactone)-Polytetrahydrofuran urethane) Based Nanofibers Enhanced Chondrogenic Differentiation and Cartilage Regeneration. J Biomed Nanotechnol. 2019; 15(5):1005-1017. [92] JIANG T, KAI D, LIU S, et al. Mechanically cartilage-mimicking poly(PCL-PTHF urethane)/collagen nanofibers induce chondrogenesis by blocking NF-kappa B signaling pathway. Biomaterials. 2018;178:281-292. [93] LI Z, LIU P, YANG T, et al. Composite poly(l-lactic-acid)/silk fibroin scaffold prepared by electrospinning promotes chondrogenesis for cartilage tissue engineering. J Biomater Appl. 2016;30(10):1552-1565. [94] DING J, CHEN B, LV T, et al. Bone Marrow Mesenchymal Stem Cell-Based Engineered Cartilage Ameliorates Polyglycolic Acid/Polylactic Acid Scaffold-Induced Inflammation Through M2 Polarization of Macrophages in a Pig Model. Stem Cells Transl Med. 2016;5(8):1079-1089. [95] WAYNE JS, MCDOWELL CL, SHIELDS KJ, et al. In vivo response of polylactic acid-alginate scaffolds and bone marrow-derived cells for cartilage tissue engineering. Tissue Eng. 2005;11(5-6):953-963. [96] LIU Q, WANG J, CHEN Y, et al. Suppressing mesenchymal stem cell hypertrophy and endochondral ossification in 3D cartilage regeneration with nanofibrous poly(l-lactic acid) scaffold and matrilin-3. Acta Biomater. 2018;76:29-38. [97] MING L, ZHIPENG Y, FEI Y, et al. Microfluidic-based screening of resveratrol and drug-loading PLA/Gelatine nano-scaffold for the repair of cartilage defect. Artif Cells Nanomed Biotechnol. 2018;46(sup1):336-346. [98] LI T, LIU B, JIANG Y, et al. L-polylactic acid porous microspheres enhance the mechanical properties and in vivo stability of degummed silk/silk fibroin/gelatin scaffold. Biomed Mater. 2020;16(1):015025. |
| [1] | 莫 骏, 罗宗平. 静态拉伸对大鼠椎间盘髓核和纤维环的影响[J]. 中国组织工程研究, 2024, 28(8): 1180-1185. |
| [2] | 李 蕊, 张桂红, 王 涛, 樊 萍. 人参多糖干预创伤性骨关节炎模型大鼠前列腺素E2/6-酮-前列腺素F1α的表达[J]. 中国组织工程研究, 2024, 28(8): 1235-1240. |
| [3] | 卜献忠, 卜保献, 许 伟, 张 驰, 张翼升, 钟远鸣, 李智斐, 唐福波, 麦 威, 周劲衍. 急性期神经根型颈椎病患者的血清差异蛋白组学分析[J]. 中国组织工程研究, 2024, 28(4): 535-541. |
| [4] | 杨启航, 蒲 锐, 陈子扬, 冷思逸, 宋永晶, 刘 辉, 杜光友. 肠道菌群代谢物在肥胖调控中的作用与机制[J]. 中国组织工程研究, 2024, 28(2): 308-314. |
| [5] | 黄惠敏, 谢冰颖, 黄景文, 黄小彬, 谢丽华, 李生强, 葛继荣. 阿仑膦酸钠治疗骨质疏松模型大鼠机制的串联质量标签蛋白质组学分析[J]. 中国组织工程研究, 2024, 28(16): 2505-2511. |
| [6] | 艾芳芳, 肖红燕, 汪 芳, 朱永朝, 马丽君. 枸杞多糖联合奥沙利铂可逆转结肠癌干细胞的耐药[J]. 中国组织工程研究, 2024, 28(1): 74-79. |
| [7] | 张厚君, 邓博文, 蒋昇源, 赵 毅, 任敬佩, 徐 林, 穆晓红. 脑性瘫痪儿童脑脊液内外泌体的蛋白质组学分析[J]. 中国组织工程研究, 2023, 27(6): 903-908. |
| [8] | 高 婷, 马小红, 李晓荣. 三种不同来源卵巢颗粒细胞外泌体的提取及鉴定[J]. 中国组织工程研究, 2023, 27(6): 860-865. |
| [9] | 张 驰, 章晓云, 柴 源, 陈 锋. 金天格胶囊治疗维甲酸诱导骨质疏松大鼠的蛋白质组学分析[J]. 中国组织工程研究, 2023, 27(35): 5634-5641. |
| [10] | 秦祎雄, 袁子健, 谢山周, 祝英文, 陈明慧, 韩 标, 郭 勇. 茶多酚对脂多糖刺激下成骨细胞增殖分化的影响[J]. 中国组织工程研究, 2023, 27(35): 5665-5669. |
| [11] | 王梦抒, 张 宇, 郑周杭, 陈 龙, 尤冬春, 郭伟锋, 胡 飞, 陈 欢, 刘幸明, 吴荣海, 张 寅. 气虚血瘀型腰椎管狭窄症患者增生肥厚与正常黄韧带之间的蛋白质表达差异[J]. 中国组织工程研究, 2023, 27(35): 5589-5595. |
| [12] | 张慧宇, 于婧文, 白振军, 李 亮, 穆秉桃, 张金锋, 解佳伟. 雷公藤甲素调节小胶质细胞极化反应保护损伤的神经元[J]. 中国组织工程研究, 2023, 27(33): 5342-5347. |
| [13] | 郑靖杰, 黄金勇, 吐尔逊江·达地汗, 高彦华, 李志强, 马海蓉. 血清TMT标记定量蛋白质组学分析鹰嘴豆芽异黄酮对骨质疏松模型大鼠的作用[J]. 中国组织工程研究, 2023, 27(31): 5022-5027. |
| [14] | 陈景涛, 陈 有, 李玉静, 刘志刚. 黄芪多糖抑制Toll样受体4/核因子κB p65通路治疗大鼠膝骨关节炎[J]. 中国组织工程研究, 2023, 27(31): 5002-5008. |
| [15] | 吴沥豪, 邵安良, 许 林, 任 康, 王洪建, 陈 亮, 许 零. 复合大孔聚多糖可吸收止血材料免疫毒性反应的评价[J]. 中国组织工程研究, 2023, 27(3): 329-334. |
关节软骨损伤治疗策略分为非手术治疗和手术治疗。非手术治疗主要为药物治疗、体质量管理和运动治疗等,但只能改善症状或作为姑息治疗,对软骨损伤无明显修复效果[5]。现有软骨修复手术主要为微骨折术、骨软骨移植术、自体软骨细胞移植/基质诱导的自体软骨细胞移植等。目前认为,较小的(< 2 cm2)软骨损伤推荐使用自体骨软骨移植和微骨折术,而较大的(> 2 cm2)软骨损伤通常采用骨软骨异体移植和自体软骨细胞移植/基质诱导的自体软骨细胞移植等治疗[6]。
尽管软骨修复策略能够部分修复软骨结构和功能,改善患者预后,但均无法恢复正常的透明软骨结构,且存在相应局限性[7]。外科技术的局限促进了组织工程技术的发展,目前基于凝胶/支架的组织工程方法是修复软骨损伤的最有前景技术之一。以凝胶/支架材料、生长因子和种子细胞为基础,组织工程技术利用凝胶/支架模拟天然软骨组织细胞外基质作为种子细胞的培养环境,并提供物理力学刺激和生化因子刺激,诱导促进软骨组织修复[8]。
该综述在介绍了多种人工生物材料基础上关注材料的创新研究,展望人工生物材料在关节软骨修复中的应用前景。 中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
1.1.1 检索人及检索时间 由第一作者在2022年12月进行检索。
1.1.2 检索文献时限 1998年1月至2022年12月。
1.1.3 检索数据库 PubMed和中国知网数据库。
1.1.4 检索词 英文检索词为“collagen,gelatin,silk,chitosan,alginate,PEG,PCL,PLA,cartilage tissue engineering,cartilage tissue engineering materials”,中文检索词为“胶原,明胶,丝素蛋白,壳聚糖,海藻酸,聚乙二醇,聚已内酯,聚乳酸,软骨组织工程,软骨组织工程材料”。
1.1.5 检索文献类型 研究原著、综述、病例报告和荟萃分析。
1.1.6 手工检索情况 无。
1.1.7 检索策略 以 PubMed 数据库检索策略为例,见图1。

1.2 入组标准
1.2.1 纳入标准 ①有关上述生物材料介绍的相关文献;②上述材料在软骨组织工程中作用及应用的相关文献;③论点论据可靠、关联度高的文献。
1.2.2 排除标准 ①内容相关性差和重复的文献;②逻辑不严谨、缺乏相关实验证据的文献;③年限较久远的的相关文献。
1.3 文献质量评估与数据提取 共检索到中英文文献共367篇,选择与文章内容相关性大并具有价值的文章进行分析讨论,排除与研究目的相关性差及内容陈旧、重复的文献269篇,最终按入选标准严格筛选后最终纳入98篇符合标准的英文文献进行综述,见图2。

3.2 作者综述有别于他人他篇的特点 目前针对人工生物材料在关节软骨损伤修复组织工程中的综述多集中于单个材料在组织工程中的进展,且由于组织工程的迅速发展,近几年出现大量关于组织工程的新研究,对于这些研究目前尚缺少详细的总结。该文章根据材料种类划分总结归纳了多种人工生物材料在关节软骨组织工程的最新研究进展,包括材料的改性、复合研究、药物负载以及临床试验等,展现以凝胶/支架、生长因子和种子细胞为基础的软骨组织工程技术。
3.3 综述的局限性 综述中涉及多种材料加工方法,由于篇幅原因,文章仅根据材料种类进行划分介绍,并未详细介绍材料具体加工方式及优缺点。另外,文章内容多集中于软骨损伤修复研究,而其中关于骨软骨损伤相关研究进展较少。
3.4 综述的重要意义 文章从软骨组织工程修复材料为切入点,介绍软骨损伤治疗策略,综述了多种人工生物材料在软骨组织工程中的作用及应用研究,提出需要探索和解决的问题,为软骨组织工程材料的后续研究和应用提供参考,也为支架的研发和临床转化提供新的思路。
3.5 课题专家组对未来的建议 通过对以上人工生物材料的综述,希望在未来的研究中能够得到人工生物材料在关节软骨修复中应用的力学性能、孔隙大小、孔隙率等标准,并通过人工和天然材料的复合改进材料结构和性状的特性,研制和开发复合材料产品,为软骨损伤的临床治疗提供更多的选择。 中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
 #br#
#br#
文题释义:
软骨组织工程:是指将种子细胞植入模拟软骨细胞外基质结构的凝胶/支架中,并将支架材料移植在缺损软骨部位,在生长因子作用下使种子细胞增殖分化、合成并分泌细胞外基质,以达到软骨修复的目的。交联:是指通过物理和化学(如光、热、超声波、加入交联剂等)方法使支架中线性高分子聚合物通过共价键连接形成网状高分子聚合物,从而改善支架的机械性能。
软骨组织损伤修复是临床中一大难点,组织工程技术利用支架/凝胶模拟天然软骨组织细胞外基质作为种子细胞的培养环境,并提供物理力学刺激和生化因子刺激,可诱导促进软骨组织修复。文章从软骨组织工程修复材料为切入点,综述胶原、明胶、丝素蛋白和聚乙二醇等多种天然与合成材料在软骨组织工程中的作用及应用研究,涉及多种材料的改性、材料复合、药物负载及相关临床试验研究,为软骨组织工程材料的后续研究和应用提供参考,也为支架的研发和临床转化提供新的思路。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||