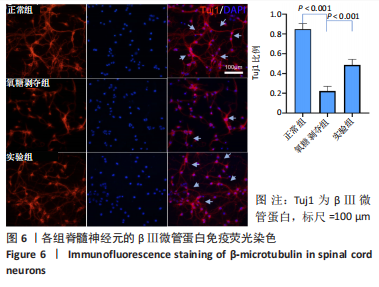
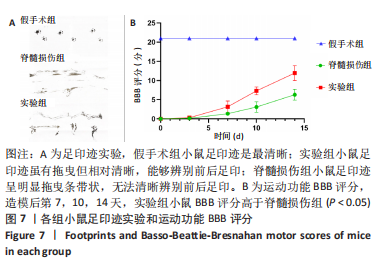
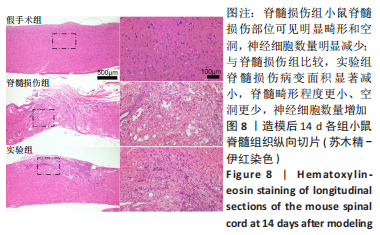
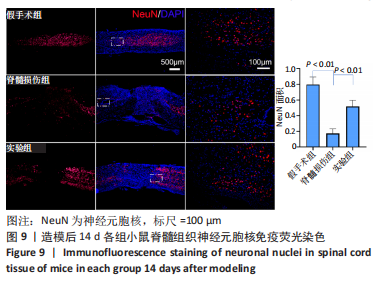
[1] COUILLARD-DESPRES S, BIELER L, VOGL M. Pathophysiology of traumatic spinal cord injury. Neurological aspects of spinal cord injury. 2017:503-528.
[2] TATOR CH, KOYANAGI I. Vascular mechanisms in the pathophysiology of human spinal cord injury. J Neurosurg. 1997;86(3):483-492.
[3] KOYONAGI I, TATOR C, LEA P. Silicone rubber microangiography of acute spinal cord injury. Neurosurgery. 1993;32:260-268.
[4] AGRAWAL SK, FEHLINGS MG. Mechanisms of secondary injury to spinal cord axons in vitro: role of Na+, Na (+)-K (+)-ATPase, the Na (+)-H+ exchanger, and the Na (+)-Ca2+ exchanger.J Neurosci. 1996;16(2):545.
[5] DYCK S, KATARIA H, AKBARI-KELACHAYEH K, et al. LAR and PTPσ receptors are negative regulators of oligodendrogenesis and oligodendrocyte integrity in spinal cord injury. Glia. 2019;67(1):125-145.
[6] CASHA S, YU W, FEHLINGS M. Oligodendroglial apoptosis occurs along degenerating axons and is associated with FAS and p75 expression following spinal cord injury in the rat. Neuroscience. 2001;103(1):203-218.
[7] ALMAD A, SAHINKAYA FR, MCTIGUE DM. Oligodendrocyte fate after spinal cord injury. Neurotherapeutics. 2011;8:262-273.
[8] BEATTIE MS, HERMANN GE, ROGERS RC, et al. Cell death in models of spinal cord injury. Prog Brain Res. 2002;137:37-47.
[9] FUJIKAWA D, SHINMEI S, CAI B. Kainic acid-induced seizures produce necrotic, not apoptotic, neurons with internucleosomal DNA cleavage: implications for programmed cell death mechanisms. Neuroscience. 2000;98(1):41-53.
[10] OYINBO CA. Secondary injury mechanisms in traumatic spinal cord injury: a nugget of this multiply cascade. Acta Neurobiol Exp (Wars). 2011;71(2):281-299.
[11] VANG S, LONGLEY K, STEER CJ, et al. The unexpected uses of urso-and tauroursodeoxycholic acid in the treatment of non-liver diseases. Glob Adv Health Med. 2014;3(3):58-69.
[12] CORTEZ L, SIM V. The therapeutic potential of chemical chaperones in protein folding diseases. Prion. 2014;8(2):197-202.
[13] CORTEZ LM, CAMPEAU J, NORMAN G, et al. Bile acids reduce prion conversion, reduce neuronal loss, and prolong male survival in models of prion disease. J Virol. 2015;89(15):7660-7672.
[14] LU X, YANG RR, ZHANG JL, et al. Tauroursodeoxycholic acid produces antidepressant‐like effects in a chronic unpredictable stress model of depression via attenuation of neuroinflammation, oxido‐nitrosative stress, and endoplasmic reticulum stress. Fundam Clin Pharmacol. 2018;32(4):363-377.
[15] ZHAN J, LI X, LUO D, et al. Polydatin promotes the neuronal differentiation of bone marrow mesenchymal stem cells in vitro and in vivo: Involvement of Nrf2 signalling pathway. J Cell Mol Med. 2020;24(9):5317-5329.
[16] BASSO DM, BEATTIE MS, BRESNAHAN JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 1995;12(1):1-21.
[17] RODRIGUES CM, STIEERS CL, KEENE CD, et al. Tauroursodeoxycholic acid partially prevents apoptosis induced by 3‐nitropropionic acid: evidence for a mitochondrial pathway independent of the permeability transition. J Neurochem. 2000;75(6):2368-2379.
[18] CASTRO-CALDAS M, CARVALHO AN, RODRIGUES E, et al. Tauroursodeoxycholic acid prevents MPTP-induced dopaminergic cell death in a mouse model of Parkinson’s disease. Mol Neurobiol. 2012;46:475-486.
[19] XIE Q, KHAOUSTOV VI, CHUNG CC, et al. Effect of tauroursodeoxycholic acid on endoplasmic reticulum stress–induced caspase‐12 activation. Hepatology. 2002;36(3):592-601.
[20] VIANA R, NUNES A, CASTRO R, et al. Tauroursodeoxycholic acid prevents E22Q Alzheimer’s Aβ toxicity in human cerebral endothelial cells. Cell Mol Life Sci. 2009;66:1094-1104.
[21] BREWER GJ, TORRICELLI JR. Isolation and culture of adult neurons and neurospheres. Nat Protoc. 2007;2(6):1490-1498.
[22] CANNON JR, GREENAMYRE JT. NeuN is not a reliable marker of dopamine neurons in rat substantia nigra. Neurosci Lett. 2009;464(1): 14-17.
[23] LEE MK, TUTTLE JB, REBHUN LI, et al. The expression and posttranslational modification of a neuron-specific β-tubulin isotype during chick embryogenesis. Cell Motil Cytoskeleton. 1990;17(2):118-132.
[24] HACHEM LD, AHUJA CS, FEHLINGS MG. Assessment and management of acute spinal cord injury: From point of injury to rehabilitation. J Spinal Cord Med. 2017;40(6):665-675.
[25] STEIN DM, KNIGHT WA. Emergency neurological life support: traumatic spine injury. Neurocrit Care. 2017;27:170-180.
[26] KANG Y, REN X, YUAN X, et al. The effects of combined micron-scale surface and different nanoscale features on cell response. Adv Mater Sci Eng. 2018;2018:1-9.
[27] CARLSON GD, GORDEN C. Current developments in spinal cord injury research. Spine J. 2002;2(2):116-128.
[28] ALIZADEH A, DYCK SM, KARIMI-ABDOLREZAEE S. Traumatic spinal cord injury: an overview of pathophysiology, models and acute injury mechanisms. Front Neurol. 2019;10:282.
[29] FAKHOURY M. Spinal cord injury: overview of experimental approaches used to restore locomotor activity. Rev Neurosci. 2015;26(4):397-405.
[30] AHUJA CS, NORI S, TETREAULT L, et al. Traumatic spinal cord injury—repair and regeneration. Neurosurgery. 2017;80(3S):S9-S22.
[31] KEENE CD, RODRIGUES CM, EICH T, et al. Tauroursodeoxycholic acid, a bile acid, is neuroprotective in a transgenic animal model of Huntington’s disease. Proc Natl Acad Sci U S A. 2002;99(16):10671-10676.
[32] RODRIGUES CM, SOLÁ S, NAN Z, et al. Tauroursodeoxycholic acid reduces apoptosis and protects against neurological injury after acute hemorrhagic stroke in rats. Proc Natl Acad Sci U S A. 2003;100(10): 6087-6092.
[33] NUNES AF, AMARAL JD, LO AC, et al. TUDCA, a bile acid, attenuates amyloid precursor protein processing and amyloid-β deposition in APP/PS1 mice. Mol Neurobiol. 2012;45(3):440-454.
[34] GRONBECK KR, RODRIGUES CM, MAHMOUDI J, et al. Application of tauroursodeoxycholic acid for treatment of neurological and non-neurological diseases: is there a potential for treating traumatic brain injury? Neurocrit Care. 2016;25(1):153-166.
[35] CASTRO-CALDAS M, CARVALHO AN, RODRIGUES E, et al. Tauroursodeoxycholic acid prevents MPTP-induced dopaminergic cell death in a mouse model of Parkinson’s disease. Mol Neurobiol. 2012;46(2):475-486.
[36] JUG M, KEJŽAR N, VESEL M, et al. Neurological recovery after traumatic cervical spinal cord injury is superior if surgical decompression and instrumented fusion are performed within 8 hours versus 8 to 24 hours after injury: a single center experience. J Neurotrauma. 2015;32(18): 1385-1392.
[37] FALASCA L, TISONE G, PALMIERI G, et al. PROTECTIVE ROLE OF TAUROURSODEOXYCHOLATE DURING HARVESTING AND COLD STORAGE OF HUMAN LIVER: A Pilot Study in Transplant Recipients: 1. Transplantation. 2001;71(9):1268-1276.
[38] KIM SY, KWON YW, JUNG IL, et al. Tauroursodeoxycholate (TUDCA) inhibits neointimal hyperplasia by suppression of ERK via PKCα-mediated MKP-1 induction. Cardiovasc Res. 2011;92(2):307-316.
[39] GUZY RD, HOYOS B, ROBIN E, et al. Mitochondrial complex III is required for hypoxia-induced ROS production and cellular oxygen sensing. Cell Metab. 2005;1(6):401-408.
[40] SARNAT HB, NOCHLIN D, BORN DE. Neuronal nuclear antigen (NeuN): a marker of neuronal maturation in the early human fetal nervous system. Brain Dev. 1998;20(2):88-94.
[41] HOU Y, LUAN J, HUANG T, et al. Tauroursodeoxycholic acid alleviates secondary injury in spinal cord injury mice by reducing oxidative stress, apoptosis, and inflammatory response. J Neuroinflammation. 2021;18: 1-13.
[42] ZOROV DB, JUHASZOVA M, SOLLOTT SJ. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev. 2014;94(3): 909-950.
[43] TRAN AP, WARREN PM, SILVER J. The biology of regeneration failure and success after spinal cord injury. Physiol Rev. 2018;98(2):881-917.
[44] YANGUAS-CASáS N, BARREDA-MANSO MA, NIETO-SAMPEDRO M,
et al. Tauroursodeoxycholic acid reduces glial cell activation in an animal model of acute neuroinflammation. J Neuroinflammation. 2014;11(1):50.
[45] ACKERMAN HD, GERHARD GS. Bile acids in neurodegenerative disorders. Front Aging Neurosci. 2016;8:263.
[46] XAVIER JM, MORGADO AL, RODRIGUES CM, et al. Tauroursodeoxycholic acid increases neural stem cell pool and neuronal conversion by regulating mitochondria-cell cycle retrograde signaling. Cell Cycle. 2014;13(22):3576-3589.
[47] SOARES R, RIBEIRO FF, XAPELLI S, et al. Tauroursodeoxycholic acid enhances mitochondrial biogenesis, neural stem cell pool, and early neurogenesis in adult rats. Mol Neurobiol. 2018;55(5):3725-3738. |