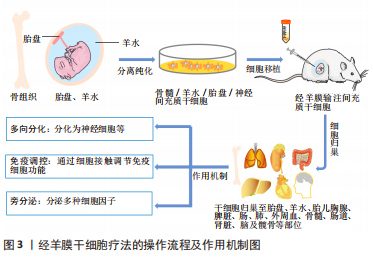
[1] 刘珍,周阳文,李小洪,等.出生缺陷防控健康教育专家共识[J].中国妇幼保健,2022,37(5):775-779.
[2] 卫生部发布《中国出生缺陷防治报告(2012)》[J].中国药房,2012, 23(39):3693.
[3] VANOVER M, WANG A, FARMER D. Potential clinical applications of placental stem cells for use in fetal therapy of birth defects. Placenta. 2017;59:107-112.
[4] SHAW SW, DAVID AL, DE COPPI P. Clinical applications of prenatal and postnatal therapy using stem cells retrieved from amniotic fluid. Curr Opin Obstet Gynecol. 2011;23(2):109-116.
[5] GOMBOS K, GALIK B, KALACS KI, et al. NGS-based application for routine non-invasive pre-implantation genetic assessment in IVF. Int J Mol Sci. 2021;22(5):2443.
[6] HUANG J, RONG L, ZENG L, et al. Embryo selection through non-invasive preimplantation genetic testing with cell-free DNA in spent culture media: a protocol for a multicentre, double-blind, randomised controlled trial. BMJ Open. 2022;12(7):e057254.
[7] KHALIL A, ARCHER R, HUTCHINSON V, et al. Noninvasive prenatal screening in twin pregnancies with cell-free DNA using the IONA test: a prospective multicenter study. Am J Obstet Gynecol. 2021;225(1):79 e1-79 e13.
[8] MELLER C, COVINI D, AIELLO H, et al. Update on prenatal diagnosis and fetal surgery for myelomeningocele. Arch Argent Pediatr. 2021;119(3): e215-e228.
[9] 揭秋玲,孙菲.宫内干细胞治疗多种胎儿遗传性疾病的可能性[J].中国组织工程研究,2021,25(31):5034-5039.
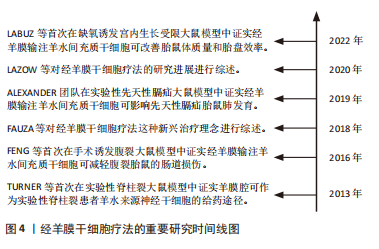
[10] FAUZA DO. Transamniotic stem cell therapy: a novel strategy for the prenatal management of congenital anomalies. Pediatr Res. 2018; 83(1-2):241-248.
[11] LAZOW SP, FAUZA DO. Transamniotic stem cell therapy. Adv Exp Med Biol. 2020;1237:61-74.
[12] TURNER CG, PENNINGTON EC, GRAY FL, et al. Intra-amniotic delivery of amniotic-derived neural stem cells in a syngeneic model of spina bifida. Fetal Diagn Ther. 2013;34(1):38-43.
[13] FENG C, GRAHAM CD, CONNORS JP, et al. Transamniotic stem cell therapy (TRASCET) mitigates bowel damage in a model of gastroschisis. J Pediatr Surg. 2016;51(1):56-61.
[14] CHALPHIN AV, TRACY SA, LAZOW SP, et al. Congenital diaphragmatic hernia as a potential target for transamniotic stem cell therapy. J Pediatr Surg. 2020;55(2):249-252.
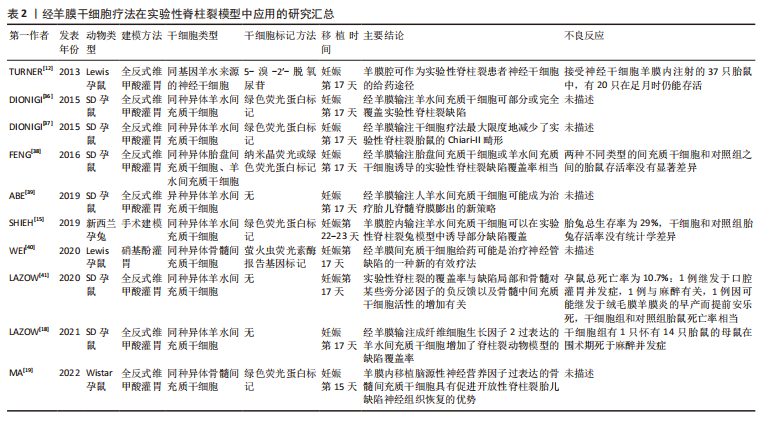
[15] SHIEH HF, TRACY SA, HONG CR, et al. Transamniotic stem cell therapy (TRASCET) in a rabbit model of spina bifida. J Pediatr Surg. 2019; 54(2):293-296.
[16] LABUZ DF, WHITLOCK AE, KYCIA I, et al. Intrauterine Growth Restriction (IUGR) as a potential target for transamniotic stem cell therapy. J Pediatr Surg. 2022;57(6):999-1003.
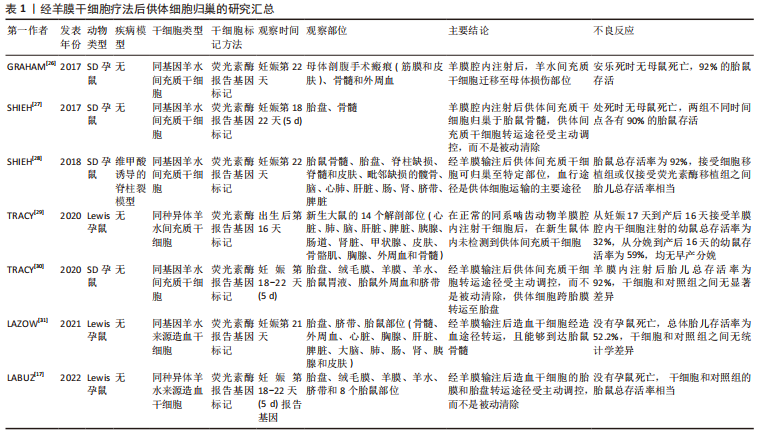
[17] LABUZ DF, WHITLOCK AE, KYCIA I, et al. Routing pathway of syngeneic donor hematopoietic stem cells after simple intra-amniotic delivery. J Pediatr Surg. 2022;57(6):986-990.
[18] LAZOW SP, LABUZ DF, KYCIA I, et al. Enhancement of transamniotic stem cell therapy for spina bifida by genetic engineering of donor mesenchymal stem cells with an Fgf2 transgene. J Pediatr Surg. 2021; 56(6):1226-1232.
[19] MA W, WEI X, GU H, et al. Intra-amniotic transplantation of brain-derived neurotrophic factor-modified mesenchymal stem cells treatment for rat fetuses with spina bifida aperta. Stem Cell Res Ther. 2022;13(1):413.
[20] EKBLAD-NORDBERG A, WALTHER-JALLOW L, WESTGREN M, et al. Prenatal stem cell therapy for inherited diseases: past, present, and future treatment strategies. Stem Cells Transl Med. 2020;9(2):148-157.
[21] CHANG YJ, SU HL, HSU LF, et al. Isolation of human neural stem cells from the amniotic fluid with diagnosed neural tube defects. Stem Cells Dev. 2015;24(15):1740-1750.
[22] PASSERA S, BOCCAZZI M, BOKOBZA C, et al. Therapeutic potential of stem cells for preterm infant brain damage: can we move from the heterogeneity of preclinical and clinical studies to established therapeutics? Biochem Pharmacol. 2021;186:114461.
[23] EBRAHIM N, MOSTAFA O, EL DOSOKY RE, et al. Human mesenchymal stem cell-derived extracellular vesicles/estrogen combined therapy safely ameliorates experimentally induced intrauterine adhesions in a female rat model. Stem Cell Res Ther. 2018;9(1):175.
[24] 李仲康,郑嘉华,田彦鹏,等.间充质干细胞治疗卵巢早衰的最新进展及机制[J].中国组织工程研究,2022,26(1):141-147.
[25] WESTGREN M, GOTHERSTROM C. Stem cell transplantation before birth - a realistic option for treatment of osteogenesis imperfecta? Prenat Diagn. 2015;35(9):827-832.
[26] GRAHAM CD, SHIEH HF, BRAZZO JA, 3RD, et al. Donor mesenchymal stem cells home to maternal wounds after transamniotic stem cell therapy (TRASCET) in a rodent model. J Pediatr Surg. 2017;52(6):1006-1009.
[27] SHIEH HF, AHMED A, TRACY SA, et al. Fetal bone marrow homing of donor mesenchymal stem cells after transamniotic stem cell therapy (TRASCET). J Pediatr Surg. 2017;12:S0022-3468(17)30655-30653.
[28] SHIEH HF, AHMED A, ROHRER L, et al. Donor mesenchymal stem cell linetics after transamniotic stem cell therapy (TRASCET) for experimental spina bifida. J Pediatr Surg. 2018;53(6):1134-1136.
[29] TRACY SA, CHALPHIN AV, LAZOW SP, et al. Postnatal fate of donor mesenchymal stem cells after transamniotic stem cell therapy in a healthy model. J Pediatr Surg. 2020;55(6):1113-1116.
[30] TRACY SA, CHALPHIN AV, KYCIA I, et al. Hematogenous donor cell routing pathway after transamniotic stem cell therapy. Stem Cells Dev. 2020;29(12):755-760.
[31] LAZOW SP, KYCIA I, LABUZ DF, et al. Fetal hematogenous routing of a donor hematopoietic stem cell line in a healthy syngeneic model of transamniotic stem cell therapy. J Pediatr Surg. 2021;56(6):1233-1236.
[32] MORENO R, MARTINEZ-GONZALEZ I, ROSAL M, et al. Fetal liver-derived mesenchymal stem cell engraftment after allogeneic in utero transplantation into rabbits. Stem Cells Dev. 2012;21(2):284-295.
[33] MARTINEZ-GONZALEZ I, MORENO R, PETRIZ J, et al. Engraftment potential of adipose tissue-derived human mesenchymal stem cells after transplantation in the fetal rabbit. Stem Cells Dev. 2012;21(18): 3270-3277.
[34] SOLTANI KHABOUSHAN A, SHAKIBAEI M, KAJBAFZADEH AM, et al. Prenatal neural tube anomalies: a decade of intrauterine stem cell transplantation using advanced tissue engineering methods. Stem Cell Rev Rep. 2022;18(2):752-767.
[35] MOLDENHAUER JS, FLAKE AW. Open fetal surgery for neural tube defects. Best Pract Res Clin Obstet Gynaecol. 2019;58:121-132.
[36] DIONIGI B, AHMED A, BRAZZO J 3RD, et al. Partial or complete coverage of experimental spina bifida by simple intra-amniotic injection of concentrated amniotic mesenchymal stem cells. J Pediatr Surg. 2015; 50(1):69-73.
[37] DIONIGI B, BRAZZO JA, 3RD, AHMED A, et al. Trans-amniotic stem cell therapy (TRASCET) minimizes Chiari-II malformation in experimental spina bifida. J Pediatr Surg. 2015;50(6):1037-1041.
[38] FENG C, C DG, CONNORS JP, et al. A comparison between placental and amniotic mesenchymal stem cells for transamniotic stem cell therapy (TRASCET) in experimental spina bifida. J Pediatr Surg. 2016; 51(6):1010-1013.
[39] ABE Y, OCHIAI D, MASUDA H, et al. In utero amniotic fluid stem cell therapy protects against myelomeningocele via spinal cord coverage and hepatocyte growth factor secretion. Stem Cells Transl Med. 2019; 8(11):1170-1179.
[40] WEI X, MA W, GU H, et al. Transamniotic mesenchymal stem cell therapy for neural tube defects preserves neural function through lesion-specific engraftment and regeneration. Cell Death Dis. 2020; 11(7):523.
[41] LAZOW SP, TRACY SA, CHALPHIN AV, et al. Initial mechanistic screening of transamniotic stem cell therapy in the rodent model of spina bifida: host bone marrow and paracrine activity. Fetal Diagn Ther. 2020; 47(12):902-911.
[42] CHUAIRE NOACK L. New clues to understand gastroschisis. Embryology, pathogenesis and epidemiology. Colomb Med (Cali). 2021;52(3): e4004227.
[43] JONES AM, ISENBURG J, SALEMI JL, et al. Increasing Prevalence of Gastroschisis--14 States, 1995-2012. MMWR Morb Mortal Wkly Rep. 2016;65(2):23-26.
[44] CHALPHIN AV, TRACY SA, LAZOW SP, et al. A comparison between placental and amniotic mesenchymal stem cells in transamniotic stem cell therapy for experimental gastroschisis. J Pediatr Surg. 2020; 55(1):49-53.
[45] FENG C, GRAHAM CD, SHIEH HF, et al. Transamniotic stem cell therapy (TRASCET) in a leporine model of gastroschisis. J Pediatr Surg. 2017;52(1):30-34.
[46] CHALPHIN AV, TRACY SA, KYCIA I, et al. Donor mesenchymal stem cell kinetics after transamniotic stem cell therapy (TRASCET) in a rodent model of gastroschisis. J Pediatr Surg. 2020;55(3):482-485.
[47] CHATTERJEE D, ING RJ, GIEN J. Update on congenital diaphragmatic hernia. Anesth Analg. 2020;131(3):808-821.
[48] KIRBY E, KEIJZER R. Congenital diaphragmatic hernia: current management strategies from antenatal diagnosis to long-term follow-up. Pediatr Surg Int. 2020;36(4):415-429.
[49] CHALPHIN AV, LAZOW SP, LABUZ DF, et al. Transamniotic stem cell therapy for experimental congenital diaphragmatic hernia: structural, transcriptional, and cell kinetics analyses in the nitrofen model. Fetal Diagn Ther. 2021;48(5):381-391.
[50] HINES EA, SUN X. Tissue crosstalk in lung development. J Cell Biochem. 2014;115(9):1469-1477.
[51] BATES DO, BEAZLEY-LONG N, BENEST AV, et al. Physiological role of vascular endothelial growth factors as homeostatic regulators. Compr Physiol. 2018;8(3):955-979.
[52] DI BERNARDO J, MAIDEN MM, HERSHENSON MB, et al. Amniotic fluid derived mesenchymal stromal cells augment fetal lung growth in a nitrofen explant model. J Pediatr Surg. 2014;49(6):859-864; discussion 864-855.
[53] GUILBERT TW, GEBB SA, SHANNON JM. Lung hypoplasia in the nitrofen model of congenital diaphragmatic hernia occurs early in development. Am J Physiol Lung Cell Mol Physiol. 2000;279(6):L1159-L1171.
[54] CONG P, TONG C, MAO S, et al. DDAH1 promotes lung endothelial barrier repair by decreasing leukocyte transendothelial migration and oxidative stress in explosion-induced lung injury. Oxid Med Cell Longev. 2022;2022:8407635.
[55] IHUNNAH CA, GHOSH S, HAHN S, et al. Nrf2 activation with CDDO-Methyl promotes beneficial and deleterious clinical effects in transgenic mice with sickle cell anemia. Front Pharmacol. 2022;13:880834.
[56] KAUR G, PORTER CBM, ASHENBERG O, et al. Mouse fetal growth restriction through parental and fetal immune gene variation and intercellular communications cascade. Nat Commun. 2022;13(1):4398.
[57] SILBER M, DEKEL N, HEUSLER I, et al. Inflammasome activation in preeclampsia and intrauterine growth restriction. Am J Reprod Immunol. 2022;88(4):e13598.
[58] LI P, HE L, LAN Y, et al. Intrauterine growth restriction induces adulthood chronic metabolic disorder in cardiac and skeletal muscles. Front Nutr. 2022;9:929943.
[59] TRAVIS OK, BAIK C, TARDO GA, et al. Adoptive transfer of placental ischemia-stimulated natural killer cells causes a preeclampsia-like phenotype in pregnant rats. Am J Reprod Immunol. 2021;85(6):e13386.
[60] KUNISAKI SM, ARMANT M, KAO GS, et al. Tissue engineering from human mesenchymal amniocytes: a prelude to clinical trials. J Pediatr Surg. 2007;42(6):974-979; discussion 979-980.
[61] STEIGMAN SA, ARMANT M, BAYER-ZWIRELLO L, et al. Preclinical regulatory validation of a 3-stage amniotic mesenchymal stem cell manufacturing protocol. J Pediatr Surg. 2008;43(6):1164-1169. |