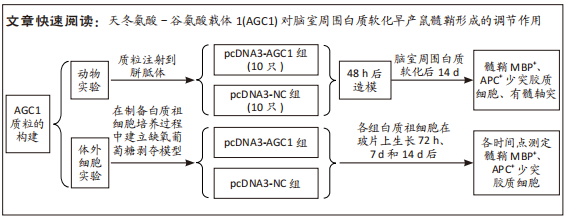
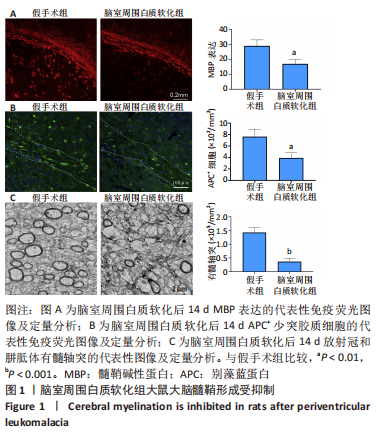
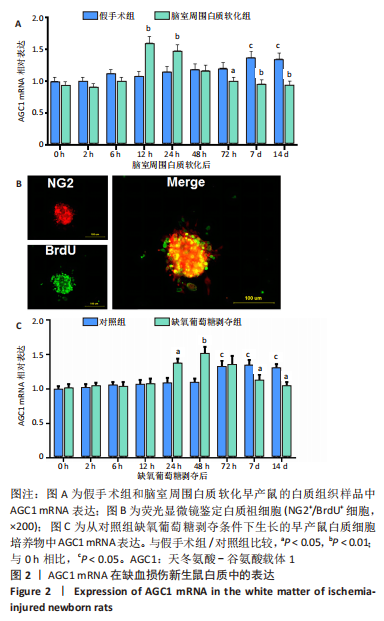
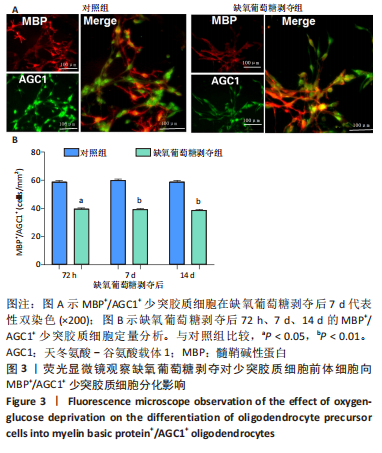
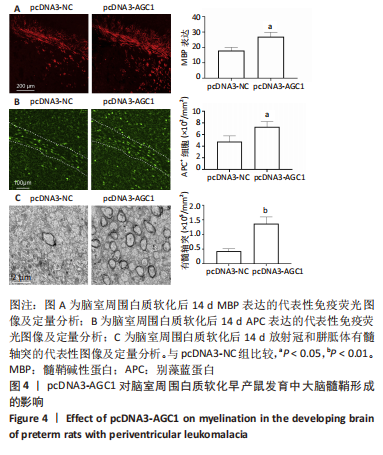
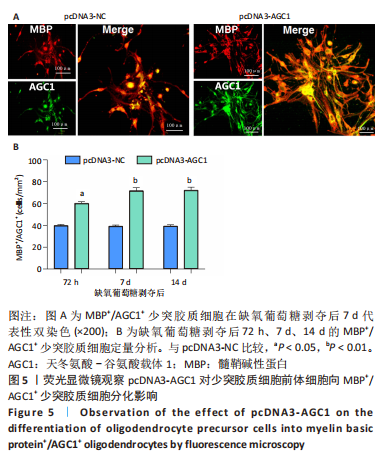
[1] 张玉,王向鹏,刘佳,等. 脑室周围白质软化大鼠脑组织Toll样受体4表达变化与细胞凋亡的关系[J]. 中华实用儿科临床杂志, 2019,34(16):1254-1258.
[2] HAO L, SHEN L, YIN Q. Clinical Diagnosis of White Matter Softening in Premature Infants Based on Electroencephalogram (EEG). J Healthc Eng. 2021;2021(98):1-10.
[3] HUANG XM. WEN SY, NG E, et al. Periventricular white matter hyperintensity burden and cognitive impairment in early Parkinson’s disease. Eur J Neurol. 2020;27(6):876-886.
[4] 叶孝严,王大雨,徐艳,等. 普仑司特对脑室周围白质软化新生大鼠的作用[J]. 中国当代儿科杂志,2020,22(6):129-134.
[5] REVIN VV, PINYAEV SI, PARCHAYKINA MV, et al. The Effect of Resveratrol on the Composition and State of Lipids and the Activity of Phospholipase A2 During the Excitation and Regeneration of Somatic Nerves. Front Physiol. 2019;10:384.
[6] BORHANI-HAGHIGHI M, MOHAMADI Y, KASHANI IR. In utero transplantation of neural stem cells ameliorates maternal inflammation-induced prenatal white matter injury. J Cell Biochem. 2019;120(8): 12785-12795.
[7] FU XM, WANG Y, FU WL, et al. The Combination of Adipose-derived Schwann-like Cells and Acellular Nerve Allografts Promotes Sciatic Nerve Regeneration and Repair through the JAK2/STAT3 Signaling Pathway in Rats. Neuroscience. 2019;422(6):134-145.
[8] YU J, YE K, LI J, et al. Schwann-like cell conditioned medium promotes angiogenesis and nerve regeneration. Cell Tissue Bank. 2021;23(1): 1-18.
[9] GROTH SL, DONAHUE SP, REDDY A, et al. Periventricular Leukomalacia in Patients With Pseudo-glaucomatous Cupping. Am J Ophthalmol. 2020;211:31-41.
[10] FORBES TA, GOLDSTEIN EZ, DUPREE JL, et al. Environmental enrichment ameliorates perinatal brain injury and promotes functional white matter recovery. Nature Commun. 2020;11(1):67-74.
[11] PETRALLA S, PEÑA-ALTAMIRA LE, POETA E, et al. Deficiency of mitochondrial aspartate-glutamate carrier 1 leads to oligodendrocyte precursor cell proliferation defects both in vitro and in vivo. Int J Mol Sci. 2019;20(18):4486.
[12] 何亚芳, 张建华, 陈惠金, 等. 美金胺对缺血诱导未成熟脑白质谷氨酸异常信号传输的体内外阻断研究[J]. 中华实用儿科临床杂志, 2020,35(14):1093-1097.
[13] PROFILO E, PEÑA-ALTAMIRA LE, CORRICELLI M, et al. Down-regulation of the mitochondrial aspartate-glutamate carrier isoform 1 AGC1 inhibits proliferation and N-acetylaspartate synthesis in Neuro2A cells. Biochim Biophys Acta Mol Basis Dis. 2017;1863(6):1422-1435.
[14] SHELTON GD, MINOR KM, LI K, et al. A Mutation in the Mitochondrial Aspartate/Glutamate Carrier Leads to a More Oxidizing Intramitochondrial Environment and an Inflammatory Myopathy in Dutch Shepherd Dogs. J Neur Dis. 2019;6(4):485-501.
[15] CONTRERAS L, RAMIREZ L, DU J, et al. Deficient glucose and glutamine metabolism in Aralar/AGC1/Slc25a12 knockout mice contributes to altered visual function. Mol Vis. 2016;22(3):1198.
[16] PROFILO E, PEÑA-ALTAMIRA LE, CORRICELLI M, et al. Down-regulation of the mitochondrial aspartate-glutamate carrier isoform 1 AGC1 inhibits proliferation and N-acetylaspartate synthesis in Neuro2A cells. BBA Mol Basis Dis. 2017;1863(6):1422-1435.
[17] PFEIFFER B, SEN K, KAUR S, et al. Expanding Phenotypic Spectrum of Cerebral Aspartate-Glutamate Carrier Isoform 1 (AGC1) Deficiency. Neuropediatrics. 2019;51(2):160-163.
[18] QIAO L, FU J, XUE X, et al. Neuronalinjury and roles of apoptosis and autophagy in a neonatal rat model of hypoxia-ischemia-induced periventricular leukomalacia. Mol Med Rep. 2018;17(4):5940-5949.
[19] MLODY B, LORENZ C, INAK G, et al. Energy metabolism in neuronal/glial induction and in iPSC models of brain disorders. Semin Cell Dev Biol. 2016;52(9):102-109.
[20] CAICEDO A, ZAMBRANO K, SANON S, et al. Extracellular mitochondria in the cerebrospinal fluid (CSF): Potential types and key roles in central nervous system (CNS) physiology and pathogenesis. Mitochondrion. 2021;58(12):1123-1132.
[21] VISCOMI C, BOTTANI E, ZEVIANI M. Emerging concepts in the therapy of mitochondrial disease. Biochim Biophys Acta. 2015;1847(6-7): 544-557.
[22] RANGARAJU V, LEWIS TL, HIRABAYASHI Y, et al. Pleiotropic Mitochondria: The Influence of Mitochondria on Neuronal Development and Disease. J Neurosci. 2019;39(42):8200-8208.
[23] FALK MJ, LI D, GAI X, et al. AGC1 deficiency causes infantile epilepsy, abnormal myelination, and reduced N-acetylaspartate. JIMD Rep. 2014;14(5):77-85.
[24] CASIMIR M, RUBI B, FRIGERIO F, et al. Silencing of the mitochondrial NADH shuttle component aspartate-glutamate carrier AGC1/Aralar1 in INS-1E cells and rat islets. Biochem J. 2009;424(3):459-466
[25] PAN S, CHAN JR. Regulation and dysregulation of axon infrastructure by myelinating glia. J Cell Biol. 2017;216(12):3903-3916.
[26] ETXEBERRIA A, HOKANSON KC, DAO DQ, et al. Dynamic modulation of myelination in response to visual stimuli alters optic nerve conduction velocity. J Neurosci. 2016;36(26):6937-6948.
[27] MUSAH AS, BROWN TL, JEFFRIES MA, et al. Mechanistic Target of Rapamycin Regulates the Oligodendrocyte Cytoskeleton during Myelination. J Neurosci. 2020;40(15):JN-RM-1434-18.
[28] TOMASSY GS, BERGER DR, CHEN HH, et al. Distinct profiles of myelin distribution along single axons of pyramidal neurons in the neocortex. Science. 2014;344(6181):319-324.
[29] ZUCCOLI GS, REIS-DE-OLIVEIRA G, GARBES B, et al. Linking proteomic alterations in schizophrenia hippocampus to NMDAr hypofunction in human neurons and oligodendrocytes. Eur Arch Psychiatry Clin Neurosci. 2021;24(3):409-416.
[30] REDLICH MJ, PRALL B, CANTO-SAID E, et al. High-pulse-energy multiphoton imaging of neurons and oligodendrocytes in deep murine brain with a fiber laser. Sci Rep. 2021;11(1):306-315.
[31] RAMOS M, PARDO B, LLORENTE-FOLCH I, et al. Deficiency of the mitochondrial transporter of aspartate/glutamate aralar/AGC1 causes hypomyelination and neuronal defects unrelated to myelin deficits in mouse brain. J Neurosci Res. 2011;89(12):2008-2017.
[32] SAKURAI T, RAMOZ N, BARRETO M, et al. Slc25a12 disruption alters myelination and neurofilaments: a model for a hypomyelination syndrome and childhood neurodevelopmental disorders. Biol Psychiatry. 2010;67(9):887-894.
[33] LLORENTE-FOLCH I, RUEDA CB, PÉREZ-LIÉBANA I, et al. L-Lactate-Mediated Neuroprotection against Glutamate-Induced Excitotoxicity Requires ARALAR/AGC1. J Neurosci. 2016;36(16):4443-4456.
[34] PÉREZ-LIÉBANA I, CASAREJOS MJ, ALCAIDE A, et al. βOHB Protective Pathways in Aralar-KO Neurons and Brain: An Alternative to Ketogenic Diet. J Neurosci. 2020;40(48):9293-9305.
[35] NAPOLIONI V, PERSICO AM, PORCELLI V, et al. The mitochondrial aspartate/glutamate carrier AGC1 and calcium homeostasis: physiological links and abnormalities in autism. Mol Neurobiol. 2011; 44(1):83-92.
[36] PUERTAS-FRÍAS G, DEL ARCO A, PARDO B, et al. Mitochondrial movement in Aralar/Slc25a12/AGC1 deficient cortical neurons. Neurochem Int. 2019;131:104541.
[37] FALK MJ, LI D, GAI X, et al. AGC1 Deficiency Causes Infantile Epilepsy, Abnormal Myelination, and Reduced N-Acetylaspartate. JIMD Rep. 2014;14:77-85.
[38] PARDO B, CONTRERAS L, SATRÚSTEGUI J. De novo Synthesis of Glial Glutamate and Glutamine in Young Mice Requires Aspartate Provided by the Neuronal Mitochondrial Aspartate-Glutamate Carrier Aralar/AGC1. Front Endocrinol (Lausanne). 2013;4:149.
[39] PROFILO E, PEÑA-ALTAMIRA LE, CORRICELLI M, et al. Down-regulation of the mitochondrial aspartate-glutamate carrier isoform 1 AGC1 inhibits proliferation and N-acetylaspartate synthesis in Neuro2A cells. Biochim Biophys Acta Mol Basis Dis. 2017;1863(6):1422-1435.
[40] JUARISTI I, GARCÍA-MARTÍN ML, RODRIGUES TB, et al. ARALAR/AGC1 deficiency, a neurodevelopmental disorder with severe impairment of neuronal mitochondrial respiration, does not produce a primary increase in brain lactate. J Neurochem. 2017;142(1):132-139. |