中国组织工程研究 ›› 2023, Vol. 27 ›› Issue (16): 2587-2594.doi: 10.12307/2023.169
• 生物材料综述 biomaterial review • 上一篇 下一篇
局部载药系统促进骨折愈合的研究与应用
贾源源1,2,段棉棉1,唐正龙1,2
- 1贵州医科大学口腔医学院,贵州省贵阳市 550000;2贵州医科大学基础医学院病理生理教研室,贵州省贵阳市 550025
-
收稿日期:2022-04-15接受日期:2022-06-13出版日期:2023-06-08发布日期:2022-11-11 -
通讯作者:唐正龙,博士,教授,贵州医科大学口腔医学院,贵州省贵阳市 550000;贵州医科大学基础医学院病理生理教研室,贵州省贵阳市 550025 -
作者简介:贾源源,女,1988年生,贵州省贵阳市人,汉族,2022年贵州医科大学毕业,博士,主治医师,主要从事骨组织工程相关研究。 -
基金资助:贵州省教育厅特色领域项目(黔教合KY字[2021]067),项目负责人:唐正龙;贵州省研究生科研基金(黔教合YJSCXJH[2020]146),项目负责人:贾源源
Research and application of local drug delivery system in promoting fracture healing
Jia Yuanyuan1, 2, Duan Mianmian1, Tang Zhenglong1, 2
- 1School of Stomatology, Guizhou Medical University, Guiyang 550000, Guizhou Province, China; 2Department of Pathophysiology, School of Basic Medicine, Guizhou Medical University, Guiyang 550025, Guizhou Province, China
-
Received:2022-04-15Accepted:2022-06-13Online:2023-06-08Published:2022-11-11 -
Contact:Tang Zhenglong, MD, Professor, School of Stomatology, Guizhou Medical University, Guiyang 550000, Guizhou Province, China; Department of Pathophysiology, School of Basic Medicine, Guizhou Medical University, Guiyang 550025, Guizhou Province, China -
About author:Jia Yuanyuan, MD, Attending physician, School of Stomatology, Guizhou Medical University, Guiyang 550000, Guizhou Province, China; Department of Pathophysiology, School of Basic Medicine, Guizhou Medical University, Guiyang 550025, Guizhou Province, China -
Supported by:the Special Field of the Department of Education Project of Guizhou Province, Grant No. KY[2021]067 (to TZL); Graduate Research Fund of the Department of Education Project of Guizhou Province, Grant No. YJSCXJH[2020]146 (To JYY)
摘要:
文题释义:
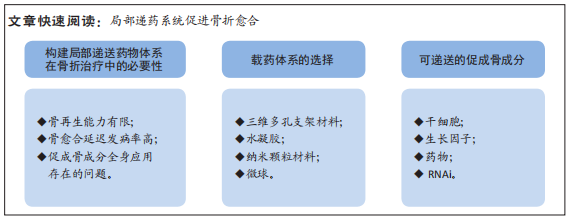
局部载药体系:给药途径有2种,分别为全身系统性给药和局部给药。传统的全身给药途径是药物通过循环系统递送,会导致如全身毒性、肝肾并发症、药物相互作用、药物达到靶器官的活性及剂量低以及患者依从性低等问题。而局部载药体系将药物递送到作用的靶器官,具有靶器官浓度高、不良反应小以及全身吸收少等优势。文章主要针对骨折愈合的药物递送体系,故而研究重点为术中植入材料负载活性因子的应用。背景:由于骨骼独特的解剖生理特征,成骨药物严格的剂量依赖性,使骨折区域给药系统仍然受到限制。近年来,随着组织工程技术的发展、载药体系的精细化与可控化,使局部用药的研究成为热点。促骨折愈合局部载药系统可以负载多种促成骨因子,也可采用多种不同的生物化合物协同作用共同促进骨折愈合。
目的:阐述局部递药系统促进骨折愈合的研究进展。
方法:以“drug delivery, fracture”为英文检索词,“载药系统,骨折”为中文检索词,检索PubMed和中国知网数据库中1990年1月至2022年2月的相关文献,最终纳入130篇文献进行分析。
结果与结论:用于局部递送药物的载体可分为三维多孔支架材料、水凝胶、纳米颗粒材料和微球,它们分别具有以下特性:良好的生物相容性和生物降解性、可控的粒径、高比表面积、易于制备等。载药体系可承载的促成骨成分有干细胞、生长因子、药物、小干扰RNA和微小RNA。骨折区局部输送药物系统具有剂量控制简单、无创给药、不良反应少的优点,使该项治疗方法促骨折愈合具有理想的治疗前景。
https://orcid.org/0000-0002-8706-1281(贾源源);https://orcid.org/0000-0002-3447-7902(唐正龙)
中图分类号:
引用本文
贾源源, 段棉棉, 唐正龙. 局部载药系统促进骨折愈合的研究与应用[J]. 中国组织工程研究, 2023, 27(16): 2587-2594.
Jia Yuanyuan, Duan Mianmian, Tang Zhenglong. Research and application of local drug delivery system in promoting fracture healing[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(16): 2587-2594.

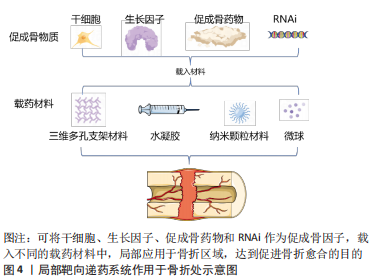
2.1.1 三维多孔支架材料 三维多孔支架材料是一种经典的骨组织工程材料,其材料内部的多孔状结构可保证高密度的细胞附着和生长因子的负载。组织工程的理想生物材料应与生物分子和细胞相结合,这些材料应促进细胞的迁移、增殖和分化,为组织再生提供机械支持。同时骨组织工程支架应具有骨诱导(促进多潜能细胞向成骨细胞分化)和骨传导(通过增强细胞活性支持骨生长)特性[7-8]。在骨组织工程中,支架的孔隙率和孔径等特性对成骨信号的表达起着至关重要的作用。孔隙率和孔径主要通过影响细胞在支架上的附着来影响细胞密度、分布和迁移。此外,相互连接的孔隙结构通过增加细胞间信号传递促进骨组织发育,并间接促进骨形态的发育。最近的研究表明,平均直径(或宽度)为100 μm或更大且开口孔隙度> 50%的互连孔隙通常被认为是允许细胞过滤和迁移到支架和组织内的最低要求[9]。
人们研究了多种方法来增加骨支架对宿主骨组织的适应性,其中有通过嫁接、图案化和涂层技术对骨诱导和骨传导因子进行表面修饰,从而改变支架的表面组成[10-11]。此外,有研究将支架负载生物信号因子、干细胞等促进骨整合,以获得更佳的成骨效果[12-13]。
2.1.2 水凝胶 水凝胶是通过物理或化学交联的方式形成的一种具有3D亲水性网络结构的聚合物,能够吸收大量水或生物流体,也具有较佳的机械强度和物理完整性[14]。水凝胶网络结构可以分为大孔径(尺寸0.1-1 μm)、微孔径(尺寸100-1 000 ?,1 ? = 0.1 nm)和无孔结构(尺寸10-100 ?)[15-17]。从水凝胶中释放的药物遵循多种释药机制,如分子扩散、化学和溶胀性释药[18]。水凝胶材料可由不同种类的生物材料构成,如天然材料(海藻酸、胶原蛋白、果胶、壳聚糖等)、合成材料(聚乙二醇、聚乳酸、聚乳酸-共乙醇酸、聚己内酯等)、组合材料(明胶-海藻酸钠、壳聚糖-海藻酸钠)等,以上水凝胶材料均可作为细胞、基因及药物的载体[19]。根据对环境的响应类型,水凝胶分为温度敏感型、pH敏感型、葡萄糖敏感型、电信号敏感型、光敏型、压敏型、离子敏感型、抗原敏感型、凝血酶诱导感染敏感型、纳米水凝胶、酶敏感和光聚合敏感型水凝胶[20]。
2.1.3 纳米颗粒材料 纳米颗粒材料是使用靶向纳米颗粒将药物输送到病变区域,其核心思想是将数百个药物分子包装成一个单一的纳米颗粒,并将合适的靶向基团连接到纳米颗粒的表面,以便将其引导至特定的损伤部位[21]。目前部分研究认为纳米材料在骨组织修复中是一种理想的再生材料[22],由于骨组织中无机矿物和有机基质是在纳米尺度上组装的,因此纳米颗粒的大小与骨组织的结构相似[23-24]。此外,纳米颗粒材料的高比表面积和体积,有利于邻近蛋白质和细胞的吸附以及发挥生物活性[25]。纳米材料的这些特性可单独或结合用于促进骨再生的治疗,尤其是在药物递送和骨组织再生领域,纳米颗粒材料可以通过包裹或表面附着来稳定药物的生物活性,从而进行分子内化,在计划靶点控制生物因子的释放[6,26]。纳米材料也可以作为化学和生物活性物质的刺激敏感传递载体,通过外部刺激,使这些物质触发传递[6]。目前人工制备的纳米颗粒缺乏批次内尺寸均匀性、批间重复性、体内稳定性和长期储存稳定性,阻碍了其临床转化和工业化生产[27-29]。因此进入临床使用的纳米颗粒种类数量相当少,开发医疗安全有效的纳米颗粒仍存在极大的挑战。
2.1.4 微球 微球(球形的中空颗粒)是一种典型的可自由流动的粉末,由天然材料或可生物降解合成聚合物组成,理想情况下其粒径小于200 μm[30]。药物分散或溶解在固体可生物降解基质中,具有药物控制和缓释的潜力[31-32]。现已发现有多种可生物降解以及不可生物降解的天然产物或合成聚合物被广泛用于微球的制备[33-34]。作为载体材料的天然聚合物包括白蛋白、明胶、胶原蛋白(蛋白质)、淀粉、琼脂糖、卡拉胶、壳聚糖(碳水化合物)、聚(丙烯酸)葡聚糖、聚(丙烯酸)淀粉(化学改性碳水化合物),作为载体材料的合成聚合物包括聚甲基丙烯酸甲酯、丙烯醛、甲基丙烯酸缩水甘油酯、环氧聚合物(不可生物降解)、聚酸酐、聚氰基丙烯酸烷基酯、丙交酯和乙交酯及其共聚物
等[35-36]。
2.2 可递送的促成骨的成分
2.2.1 干细胞 支架、细胞和外部刺激是组织工程的3个主要组成部分,其中支架充当细胞生长的模板。3D培养物的暴露微环境模拟体内条件,促进细胞与其微环境之间更有效的相互作
用[37]。有研究采用明胶支架作为载体负载人脂肪干细胞,在小鼠颅骨缺损模型中获得了理想的成骨效果[38]。同时,有研究指出,通过改变透明质酸的分子质量可以调节水凝胶支架的机械强度,低强度水凝胶中的骨髓间充质干细胞可以通过激活Wnt/β-连环蛋白途径维持干细胞特性,而具有较高机械强度的高分子量水凝胶有可能通过打开瞬时受体电位香草醛4(transient receptor potential vanilloid 4,TRPV4)/Ca2+分子通道来促进骨髓间充质干细胞的软骨分化方向,也增加了骨髓间充质干细胞中Ⅱ型胶原和SOX9的表达[39]。
尽管已有多篇文献提出骨髓间充质干细胞可以促进骨再生,但可以直接进行组织修复的成熟骨髓间充质干细胞数量有限[40-41]。同时,有研究提出,骨髓间充质干细胞的旁分泌作用增强了巨噬细胞和内皮细胞在损伤部位的募集,促进了血管生成和骨形成[42-43],这也是干细胞促进骨折愈合的方式之一。然而,基于细胞的治疗在实际临床工作中面临细胞数量限制以及患者来源细胞中存在批间变异性的限制[44-45],因此干细胞治疗在临床工作中仍处于研究阶段。
2.2.2 生长因子局部应用
(1)骨形态发生蛋白:骨形态发生蛋白2和骨形态发生蛋白7是研究最为深入的骨再生生长因子,因为它们能够促进间充质干细胞分化为软骨细胞和成骨细胞[46]。通过OP-1?递送的重组人骨形态发生蛋白7植入物被FDA批准用于修复不愈合的长骨缺损[47-48]。重组人骨形态发生蛋白2通过INFUSE?递送胶原海绵骨移植物也被临床批准用于急性骨折和脊柱融合治疗[49]。在临床试验中,胫骨骨干骨折采用自体骨移植或同种异体骨移植与重组人骨形态发生蛋白2-INFUSE?治疗进行随机对照试验[50],结果显示,自体骨移植组和重组人骨形态发生蛋白2组的愈合率分别为67%和87%,表明重组人骨形态发生蛋白2用于骨折愈合比自体骨移植的金标准更促进成骨。
将重组人骨形态发生蛋白7包裹在聚乳酸-羟基乙酸纳米球中,而后加载到左旋聚乳酸支架中,可获得重组人骨形态发生蛋白7的持续释放。将上述支架植入皮下,在3周后显示出具有矿化基质的新生骨形成[51]。控制释放速率取决于聚合物结构的物理化学性质、生物活性因子以及交联剂的类型和密度,例如:明胶水凝胶已被用于通过皮下植入传递重组人骨形态发生蛋白2,并显示出增强骨祖细胞募集和新生骨形成的作用[52]。通过向聚(ε-己内酯-共乳酸)和聚乙二醇单元制成的热敏嵌段共聚物中添加pH敏感的磺胺二甲基嘧啶低聚物(sulfamethazine oligomer,SMO)端基,设计出输送骨形态发生蛋白2的pH敏感和热敏水凝胶[53]。该系统在较窄的pH值和温度范围内显示出功能性骨形态发生蛋白2活性,皮下植入7周内碱性磷酸酶活性和矿化度升高。
尽管有大量积极的研究结果,但生长因子的使用受到其多效性的限制[54]。骨形态发生蛋白的骨诱导作用已在各种动物模型中得到证实[55],但由于其安全性和成本效益方面的某些缺陷,其在临床中的应用仍存在争议[56-57]。骨形态发生蛋白的半衰期短,再加上不理想的给药方式,使骨形态发生蛋白超生理剂量使用,导致成本增高。同时有研究报告了骨形态发生蛋白的一系列不良反应,包括异位骨化、软组织肿胀、免疫反应和癌症发生率升高,可能与使用高剂量骨形态发生蛋白有关[58]。
(2)成纤维生长因子:成纤维生长因子信号在骨骼发育和骨折愈合中起着至关重要的作用[59-60],局部应用成纤维生长因子可增强骨折愈合和骨修复[61-63]。有临床试验探讨采用可生物降解明胶水凝胶为载体的成纤维生长因子加速骨修复的有效性和安全性,一项用于截骨术部位[64],另一项用于新鲜胫骨干骨折[65]。这两项研究均证明了局部应用成纤维生长因子加速骨修复的临床疗效,且无不良反应。在截骨术部位,当剂量为200,400和800 mg时,明胶水凝胶中的成纤维生长因子呈剂量依赖性地加速骨愈合[64]。在胫骨骨折模型中,局部应用低剂量或高剂量的碱性成纤维细胞生长因子(0.8,2.4 mg/d)与安慰剂组相比,水凝胶加速骨折愈合[65]。这些临床试验以及临床前数据清楚地表明,成纤维生长因子具有作为骨折局部愈合剂的潜力。
2.2.3 药物局部递送
(1)锶和雷奈酸锶:目前有相关研究表明雷奈酸锶可以促进骨折愈合。NEGRI等[66]报告了口服雷奈酸锶(2 g/d)后,长骨不愈合的骨折成功愈合案例,完全愈合需要3-6个月。因此,开发一种在骨折部位局部释放锶类药物的递送系统被越来越多的学者关注。
一些研究使用含锶钛或含锶的羟基磷灰石局部输送锶离子,以增强种植体的骨整合[67-71]。另一种输送锶的方法是骨水泥。有研究显示,锶混合于骨水泥中可加速细胞成骨分化能力[72]。在卵巢切除术大鼠身上进行的体内实验表明,与单独使用CaP骨水泥相比,使用锶-CaP骨水泥时,骨折愈合和骨形成更快[73]。同时,含有锶混合羟基磷灰石的壳聚糖/右旋糖酐水凝胶材料在大鼠颅骨中具有极强的成骨潜能[74]。注射性水凝胶可以很容易地用于锶和骨髓间充质干细胞的输送,用于骨质疏松性骨折的修复。尽管已证实锶离子对骨愈合和骨质疏松有积极作用,但尚未见对雷奈酸锶局部递送的体内研究。
(2)双膦酸盐类:双膦酸盐如唑来膦酸盐、帕米膦酸盐、利塞膦酸盐和阿仑膦酸盐,通过抑制活跃重塑部位的破骨细胞活性来防止骨量损失,它们具有钙螯合能力,因此可以有效地作用于骨组织靶点。然而,口服二膦酸盐具有某些缺点,包括生物利用度低和不良反应,从胃肠道症状到非典型股骨骨折和颌骨骨坏死[75-76]。双膦酸盐的局部给药可用于提高骨折稳定性,具有更快的疗效和增加药物的可用性。
因为双膦酸盐对骨矿物质的高亲和力,学者们关注于该药物在骨缺损区的局部应用,研究最多的给药策略是采用植入物涂层和可注射骨水泥[77-81]。此外可生物降解的聚合物涂层已被研究用于双膦酸盐的输送。GREINER等[82]研究了唑来膦酸盐与植入物的聚(D,L-丙交酯)涂层结合,用于大鼠闭合性胫骨骨折的固定。与对照组相比,唑来膦酸盐缓释植入物在
42 d后显示骨折愈合得到改善,骨痂面积更大,机械强度更大,骨折桥接更快。有体内研究表明,释放双膦酸盐的植入物可增大其拔出力[83]。
(3)甲状旁腺激素:根据浓度和给药方式不同,甲状旁腺激素1-34可能具有合成代谢或分解代谢作用。一般来说,高剂量和持续剂量的甲状旁腺激素会导致骨吸收,但间歇性高剂量或低剂量注射会促进骨形成[84-85]。口服甲状旁腺素以及许多其他治疗性肽和蛋白质存在一些挑战[86],例如它们在胃肠道中的降解。因此,甲状旁腺激素通常通过注射给药。
ZOU等[87]学者利用纳米羟基磷灰石混合入壳聚糖/海藻酸钠水凝胶作为支架,载入甲状旁腺激素1-34,促进了大鼠颅骨缺损区骨再生。然而甲状旁腺激素在体外的释放速度较快,第8天时甲状旁腺激素释放量就达到总体系甲状旁腺激素含量的80%,并未达到一个长时间药物释放。WOJDA等[88-89]学者采用聚富马酸丙二醇酯作为支架,甲状旁腺激素混合于硫醇烯水凝胶包裹支架,有利于大鼠股骨缺损部位成骨。但甲状旁腺激素的释放量在前3 d就达到了80%,显示为突释效果,而后的甲状旁腺激素在14 d内释放,该种方法进行甲状旁腺激素局部载药释药速度过快,需在延长甲状旁腺激素缓释体系的释药时间上做更多的研究。有研究也直接采用纳米棒羟基磷灰石浸泡于甲状旁腺激素溶液中孵育过夜后,作用于鼠骨缺损区域[90],该研究仅检测了6 h内的释药比例,在生理酸碱度(pH 7.4)条件下,6 h内甲状旁腺激素释放率为(26.19±6.83)%,可见目前甲状旁腺激素缓释体系研究中普遍存在的问题是药物释放速度较快,与骨折愈合时间并不匹配。同时释药体系构建过程中,甲状旁腺激素可能有一定程度的降解[91],这些都是释药体系研发中需要解决的问题。
(4)辛伐他汀:他汀类药物是一种小分子药物,与双膦酸盐类似,可抑制HMG-CoA还原酶途径[92-93]。与双膦酸盐相比,他汀类药物对骨表面没有高亲和力,因此对骨没有特异性。他汀类药物通常用于降低血液胆固醇,但最近的研究也报告了他汀类药物使用者的骨吸收率降低、骨密度增加。
通过局部载药体系递送辛伐他汀的相关研究表明,局部释放辛伐他汀对成骨也有积极影响。聚乳酸-羟基乙酸是辛伐他汀常用的合成聚合物载体。辛伐他汀-聚乳酸-羟基乙酸复合支架可缓慢释放辛伐他汀,促进细胞成骨分化和大鼠颅骨缺损愈合[94-95]。然而,聚乳酸-羟基乙酸支架的主要缺点是酸性降解产物对正常成骨微环境有害[96]。在聚乳酸-羟基乙酸材料中加入无机材料,如羟基磷灰石和磷酸钙,可以中和酸性降解产物,改善材料的机械性能,同时与成骨相关物质如钙离子、磷离子等相结合。氧化纤维素纳米纤维是一种通过其羟基与生物活性分子结合的生物材料。辛伐他汀已被证明可附着在氧化纤维素纳米纤维-明胶水凝胶涂层的双向磷酸钙陶瓷支架上,此复合支架可促进MC3T3E1细胞的成骨分化[97]。然而,对于使用连接基团合成的辛伐他汀涂层,应注意这些化学连接的形成是否会影响材料本身的性能。
此外,含药纳米粒是理想的辛伐他汀给药平台选择之一,因为它们具有高比表面积、缓释性能、高药物包封率、增强的药物渗透性和高稳定性[98-99]。纳米粒由壳聚糖、聚乳酸-羟基乙酸等通过静电纺丝或离子凝胶过程制成,在此过程中辛伐他汀可以被包裹其中[98,100]。然后,随着周围纳米颗粒材料膨胀,辛伐他汀慢慢释放到骨缺损部位。这些含药物的纳米颗粒已被证明具有促进细胞外基质体外矿化和成骨分化的能力。邹伟龙等[101]学者采用聚左乳酸微球与磷酸钙共混的可注射体系为载体负载辛伐他汀,结果显示高剂量(1 mg)辛伐他汀组的成骨效果最佳。
(5)前列腺素E2受体激动剂:前列腺素是多不饱和脂肪酸的代谢产物,在体内有广泛的作用。特别是在骨愈合和形成中发现前列腺素E2发挥重要作用。动物模型中,前列腺素E2治疗可增加骨量并促进骨痂形成[102-103]。然而,全身系统性前列腺素E2的临床应用受到严重不良反应的阻碍。为了避免全身应用的不利影响,已经开发出对某些前列腺素E2受体具有选择性的小分子激动剂,尤其是前列腺素E2受体EP2和EP4的激动剂。
有研究表明,EP2受体激动剂CP-533536在聚乳酸-羟基乙酸基质中体内局部释放可增强骨愈合[104-105]。LI等[104]将EP2受体激动剂CP-533536加入溶于N-甲基-2-吡咯烷酮的聚乳酸-羟基乙酸中,注射到大鼠闭合性横向股骨骨折区。3周后,与对照组相比,在聚乳酸-羟基乙酸中使用0.5,5 mg
CP-533536治疗的骨折区显示出骨痂增大,其力学性能得到改善。另一项研究研究表明,在聚乳酸-羟基乙酸基质中局部递送的
CP-533536(5,10,50 mg)可加速犬尺骨和胫骨临界尺寸截骨缺损的骨愈合[105]。
有研究将EP4受体激动剂(ONO-4819)与低剂量骨形态发生蛋白2结合后,在局部释放,可刺激骨形成[106-107]。研究使用含有β-磷酸三钙粉末的聚合物凝胶(二恶烷酮/聚乙二醇嵌段共聚物)作为传递系统,首次在兔脊柱融合模型中显示了ONO-4819与低剂量骨形态发生蛋白2的协同效应[106],结果显示,联合投药
(45 μg EP4和7.5 μg 骨形态发生蛋白2)的融合率最高。与之对应的是,兔脊柱融合单独采用骨形态发生蛋白2所需剂量较高,含量在20-100 μg之间[108]。小鼠颅骨缺损模型中也观察到了类似的结果,丙烯酸酯基修饰的含胆固醇支链淀粉/聚乙二醇纳米凝胶为载体,载入0.5 μg 骨形态发生蛋白2与100 μg ONO-AE1-437(EP4受体激动剂)[107]。低剂量骨形态发生蛋白2或ONO-AE1-437单独使用,该材料均不能在4周内诱导颅骨缺损的骨形成,但这些因子联合使用的骨修复效果几乎与阳性对照组(即高剂量2 μg 骨形态发生蛋白2)相同的结果。
2.2.4 RNA干扰 RNA干扰是一种通过短RNA序列[包括小干扰RNA(siRNA)或微小RNA(miRNA)]分子转录后抑制基因表达的机制[109-110]。siRNA和miRNA途径相似,但siRNA被设计为与靶序列100%互补。因此,siRNA导致mRNA降解,而miRNA诱导翻译抑制。
对于骨折愈合,可以引入RNA干扰来沉默负调节骨折愈合的基因。有研究报道了利用RNA干扰将不同类型的干细胞分化为肌肉骨骼、心血管和神经元谱系细胞。例如,针对DKK-1或胍核苷酸结合蛋白α刺激活性多肽1(guanidine nucleotide binding
protein alpha stimulating activity polypeptide 1,GNAS1)的siRNA递送导致间充质干细胞的成骨分化增强[111-112]。研究结果表明,将siRNA应用于干细胞可以促进间充质干细胞分化、存活和组织再生[113]。
纳米技术可增强siRNA功效,特别是纳米颗粒递送系统方面也具有巨大潜力。阳离子脂质体、脂质体和脂质纳米粒是RNA干扰传递最常用的非病毒载体[114]。然而,由于膜完整性受损,脂质纳米粒在高剂量下具有毒性,会导致细胞溶解和坏死死
亡[115]。也有报道称脂质纳米粒可诱导免疫反应[115]。因此,仔细选择脂质和控制剂量对于脂质纳米粒的翻译非常重要。
聚(乙烯)胺、树状大分子、聚-L-赖氨酸、聚-L-精氨酸、壳聚糖及其衍生物等聚合物通常用于siRNA纳米载体的开发[111]。由于良好的细胞膜相互作用、高细胞摄取和高效的内体逃逸,聚(乙烯)胺一直是siRNA递送的载体[116]。阳离子线性聚(乙烯)胺通过可生物降解的酯连接剂结合到光交联右旋糖酐(DEX)水凝胶中,以促进siRNA的持续释放。阳离子线性聚(乙烯)胺和水凝胶之间共价酯键的降解导致线性聚(乙烯)胺/siRNA复合物随时间可调控释放[117]。
聚乳酸-羟基乙酸是乳酸和乙醇酸的共聚物,近年来已被用作siRNA递送的纳米载体。基于聚乳酸-羟基乙酸的siRNA载体具有高稳定性、生物降解性、低毒性和缓释特性[118]。然而,由于聚乳酸-羟基乙酸和siRNA之间的静电相互作用可以忽略不计,聚乳酸-羟基乙酸制剂在siRNA传递中的潜力有限[118]。
为了克服这些挑战,有研究采用各种阳离子纳米颗粒(如DOTAP、聚(乙烯)胺或多胺)修饰聚乳酸-羟基乙酸表面[118-119]。使用乳化扩散生产方法制备了聚(乙烯)胺功能化的聚乳酸-羟基乙酸纳米颗粒,结果显示优化了siRNA的递送能力[120]。
在过去十年中,已经开发出用于siRNA递送的聚合物胶束纳米颗粒[121-123]。自组装到纳米颗粒的聚合物具有易于加载siRNA的优势,这允许构建尺寸可控且单分散的siRNA加载纳米颗粒,并且在体内应用中显示出独特的优势[124]。复合siRNA和阳离子载体与带负电荷的细胞膜静电相互作用,以进行体内摄取[125-126]。这些聚合物还表现出pH值应激性,当pH值在内溶酶体中下降时,通过可原型化的嵌段和疏水性膜相互作用单体促进siRNA内体逃逸[127-129]。此外,纳米颗粒能够同时装载siRNA和疏水性药物,以实现潜在的协同效应[124,130]。
尽管基因或siRNA的呈递可能为未来治疗骨折提供强有力的方法,然而该技术仍然存在一些问题。如何将基因或siRNA可靠有效地传递到局部骨组织是一个挑战。病毒载体可以为基因转移提供足够的效率,但安全问题阻碍了它们在临床上的应用,而非病毒载体的低效率也使其无法得到广泛应用。这些将是未来科研工作中需要攻克的困难。

| [1] Turner CH. Biomechanics of bone: determinants of skeletal fragility and bone quality. Osteoporos Int. 2002;13(2):97-104. [2] WALTER N, KERSCHBAUM M, PFEIFER C, et al. Long-term patient-related quality of life after successfully treated aseptic non-unions of the long bones. Injury. 2021;52(7):1880-1885. [3] TÄGIL M, MCDONALD MM, MORSE A, et al. Intermittent PTH(1-34) does not increase union rates in open rat femoral fractures and exhibits attenuated anabolic effects compared to closed fractures. Bone. 2010;46(3):852-859. [4] LOW S, GALLIFORD C, YANG J, et al. Biodistribution of fracture-targeted GSK3β inhibitor-loaded micelles for improved fracture healing. Biomacromolecules. 2015;16(10):3145-3153. [5] ZHOU J, HOREV B, HWANG G, et al. Characterization and optimization of pH-responsive polymer nanoparticles for drug delivery to oral biofilms. J Mater Chem B. 2016;4(18):3075-3085. [6] WANG H, LEEUWENBURGH S, LI Y, et al. The use of micro- and nanospheres as functional components for bone tissue regeneration. Tissue Eng Part B Rev. 2012; 18(1):24-39. [7] WUBNEH A, TSEKOURA EK, AYRANCI C, et al. Current state of fabrication technologies and materials for bone tissue engineering. Acta Biomater. 2018;80: 1-30. [8] LOPES D, MARTINS-CRUZ C, OLIVEIRA MB, et al. Bone physiology as inspiration for tissue regenerative therapies. Biomaterials. 2018;185:240-275. [9] ZURITA-MÉNDEZ NN, CARBAJAL-DE LA TORRE G, FLORES-MERINO MV, et al. Development of Bioactive Glass-Collagen-Hyaluronic Acid-Polycaprolactone Scaffolds for Tissue Engineering Applications. Front Bioeng Biotechnol. 2022;10: 825903. [10] ZAMUNER A, BRUN P, SCORZETO M, et al. Smart biomaterials: Surfaces functionalized with proteolytically stable osteoblast-adhesive peptides. Bioact Mater. 2017;2(3):121-130. [11] ZHANG S, ZHAO G, WANG J, et al. Organic solvent-free preparation of chitosan nanofibers with high specific surface charge and their application in biomaterials. ACS Appl Mater Interfaces. 2021;13(10):12347-12358. [12] JO Y, RIZZI S, EHRBAR M, et al. Biomimetic PEG hydrogels crosslinked with minimal plasmin-sensitive tri-amino acid peptides. J Biomed Mater Res A. 2010;93(3): 870-877. [13] REN B, BETZ O, SEITZ D, et al. Osteogenic differentiation of human adipose-derived stem cells seeded on a biomimetic spongiosa-like scaffold: bone morphogenetic protein-2 delivery by overexpressing fascia. Int J Mol Sci. 2022; 23(5):2712. [14] DEMIR G, ERDEMLI Ö, KESKIN D, et al. Xanthan-gelatin and xanthan-gelatin-keratin wound dressings for local delivery of Vitamin C. Int J Pharm. 2022;614:121436. [15] SAZZAD F, KUZEMCZAK M, LOH E, et al. Targeted myocardial restoration with injectable hydrogels-in search of the holy grail in regenerating damaged heart tissue. Biomedicines. 2021;9(6):595. [16] JOHNSON T, CHRISTMAN K. Injectable hydrogel therapies and their delivery strategies for treating myocardial infarction. Expert Opin Drug Deliv. 2013;10(1): 59-72. [17] GARG T, GOYAL A. Biomaterial-based scaffolds--current status and future directions. Expert Opin Drug Deliv. 2014;11(5):767-789. [18] HUYNH D, IM G, CHAE S, et al. Controlled release of insulin from pH/temperature-sensitive injectable pentablock copolymer hydrogel. J Control Release. 2009; 137(1):20-24. [19] KIANFAR F, AYENSU I, BOATENG J. Development and physico-mechanical characterization of carrageenan and poloxamer-based lyophilized matrix as a potential buccal drug delivery system. Drug Dev Ind Pharm. 2014;40(3):361-369. [20] HIEW S, WANG J, KOH K, et al. Bioinspired short peptide hydrogel for versatile encapsulation and controlled release of growth factor therapeutics. Acta Biomater. 2021;136:111-123. [21] DEBBAGE P, JASCHKE W. Molecular imaging with nanoparticles: giant roles for dwarf actors. Histochem Cell Biol. 2008;130(5):845-875. [22] CHOI B, LEE S. Nano/micro-assisted regenerative medicine. Int J Mol Sci. 2018; 19(8):2187. [23] RAJESH R, RAVICHANDRAN Y. Development of a new carbon nanotube-alginate-hydroxyapatite tricomponent composite scaffold for application in bone tissue engineering. Int J Nanomedicine. 2015;10 Suppl 1(Suppl 1):7-15. [24] ZHAO X, LI L, CHEN M, et al. Nanotechnology assisted targeted drug delivery for bone disorders: potentials and clinical perspectives. Curr Top Med Chem. 2020; 20(30):2801-2819. [25] ZHANG K, MIN X, ZHANG T, et al. Biodeposited nano-CdS drives the in situ growth of highly dispersed sulfide nanoparticles during pyrolysis for enhanced oxygen evolution reaction. ACS Appl Mater Interfaces. 2020;12(49):54553-54562. [26] KANG M, JANG H, LEE S, et al. Functional graphene nanomaterials-based hybrid scaffolds for osteogenesis and chondrogenesis. Adv Exp Med Biol. 2022;1351: 65-87. [27] ĐORĐEVIĆ S, GONZALEZ M M, CONEJOS-SáNCHEZ I, et al. Current hurdles to the translation of nanomedicines from bench to the clinic. Drug Deliv Transl Res. 2022;12(3):500-525. [28] CAPUTO F, CLOGSTON J, CALZOLAI L, et al. Measuring particle size distribution of nanoparticle enabled medicinal products, the joint view of EUNCL and NCI-NCL. A step by step approach combining orthogonal measurements with increasing complexity. J Control Release. 2019;299:31-43. [29] SALAMANNA F, GAMBARDELLA A, CONTARTESE D, et al. Nano-based biomaterials as drug delivery systems against osteoporosis: a systematic review of preclinical and clinical evidence. Nanomaterials (Basel). 2021;11(2):530. [30] LEE J, OH Y, LEE S, et al. Facile control of porous structures of polymer microspheres using an osmotic agent for pulmonary delivery. J Control Release. 2010;146(1):61-67. [31] ZHANG J, TANG Q, XU X, et al. Development and evaluation of a novel phytosome-loaded chitosan microsphere system for curcumin delivery. Int J Pharm. 2013;448(1):168-174. [32] YUAN F, WANG H, GUAN J, et al. Fabrication of injectable chitosan-chondroitin sulfate hydrogel embedding kartogenin-loaded microspheres as an ultrasound-triggered drug delivery system for cartilage tissue engineering. Pharmaceutics. 2021;13(9):1487. [33] NINGSIH H, CHEN L, CHUNG R, et al. An investigation on spray-granulated, macroporous, bioactive glass microspheres for a controlled drug delivery system. Materials (Basel). 2021;14(11):3112. [34] SORIANO-SOUZA C, VALIENSE H, MAVROPOULOS E, et al. Doxycycline containing hydroxyapatite ceramic microspheres as a bone-targeting drug delivery system. J Biomed Mater Res B Appl Biomater. 2020;108(4):1351-1362. [35] GARCÍA-GARCÍA J, AZUARA G, FRAILE-MARTINEZ O, et al. Modification of the polymer of a bone cement with biodegradable microspheres of PLGA and loading with daptomycin and vancomycin improve the response to bone tissue infection. Polymers (Basel). 2022;14(5):888. [36] PARNAMI N, GARG T, RATH G, et al. Development and characterization of nanocarriers for topical treatment of psoriasis by using combination therapy. Artif Cells Nanomed Biotechnol. 2014;42(6):406-412. [37] XING Q, YATES K, VOGT C, et al. Increasing mechanical strength of gelatin hydrogels by divalent metal ion removal. Sci Rep. 2014;4:4706. [38] WANG C, HONG P, WANG D, et al. Polymeric gelatin scaffolds affect mesenchymal stem cell differentiation and its diverse applications in tissue engineering. Int J Mol Sci. 2020;21(22):8632. [39] REN Y, ZHANG H, WANG Y, et al. Hyaluronic acid hydrogel with adjustable stiffness for mesenchymal stem cell 3D culture via related molecular mechanisms to maintain stemness and induce cartilage differentiation. ACS Appl Bio Mater. 2021; 4(3):2601-2613. [40] GóMEZ-BARRENA E, PADILLA-EGUILUZ N, ROSSET P, et al. Early efficacy evaluation of mesenchymal stromal cells (MSC) combined to biomaterials to treat long bone non-unions. Injury. 2020;51 Suppl 1:S63-S73. [41] GRANERO-MOLTÓ F, WEIS J, MIGA M, et al. Regenerative effects of transplanted mesenchymal stem cells in fracture healing. Stem Cells. 2009;27(8):1887-1898. [42] FURUTA T, MIYAKI S, ISHITOBI H, et al. Mesenchymal stem cell-derived exosomes promote fracture healing in a mouse model. Stem Cells Transl Med. 2016;5(12):1620-1630. [43] WANG J, ZHANG D, ZHU Y, et al. Astragalus and human mesenchymal stem cells promote wound healing by mediating immunomodulatory effects through paracrine signaling. Regen Med. 2022;17(4):219-232. [44] RENNERFELDT DA, VAN VLIET KJ. Concise review: when colonies are not clones: evidence and implications of intracolony heterogeneity in mesenchymal stem cells. Stem Cells. 2016;34(5):1135-1141. [45] NEWMAN MR, BENOIT DS. Local and targeted drug delivery for bone regeneration. Curr Opin Biotechnol. 2016;40:125-132. [46] MBALAVIELE G, SHEIKH S, STAINS J, et al. Beta-catenin and BMP-2 synergize to promote osteoblast differentiation and new bone formation. J Cell Biochem. 2005;94(2):403-418. [47] HAIDAR ZS, HAMDY RC, TABRIZIAN M. Delivery of recombinant bone morphogenetic proteins for bone regeneration and repair. Part A: Current challenges in BMP delivery. Biotechnol Lett. 2009;31(12):1817-1824. [48] MATHAVAN N, TÄGIL M, ISAKSSON H. Do osteoporotic fractures constitute a greater recalcitrant challenge for skeletal regeneration? Investigating the efficacy of BMP-7 and zoledronate treatment of diaphyseal fractures in an open fracture osteoporotic rat model. Osteoporos Int. 2017;28(2):697-707. [49] RATKO TA, BELINSON SE, SAMSON DJ, et al. Bone Morphogenetic Protein: The State of the Evidence of On-Label and Off-Label Use [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2010 Aug 6. [50] JONES A, BUCHOLZ R, BOSSE M, et al. Recombinant human BMP-2 and allograft compared with autogenous bone graft for reconstruction of diaphyseal tibial fractures with cortical defects. A randomized, controlled trial. J Bone Joint Surg Am. 2006;88(7):1431-1441. [51] WEI G, JIN Q, GIANNOBILE W, et al. The enhancement of osteogenesis by nano-fibrous scaffolds incorporating rhBMP-7 nanospheres. Biomaterials. 2007;28(12): 2087-2096. [52] XIONG A, HE Y, GAO L, et al. The fabrication of a highly efficient hydrogel based on a functionalized double network loaded with magnesium ion and BMP2 for bone defect synergistic treatment. Mater Sci Eng C Mater Biol Appl. 2021;128:112347. [53] KIM H, SHIM W, KIM S, et al. Injectable in situ-forming pH/thermo-sensitive hydrogel for bone tissue engineering. Tissue Eng Part A. 2009;15(4):923-933. [54] KIRKER-HEAD CA, BOUDRIEAU RJ, KRAUS KH. Use of bone morphogenetic proteins for augmentation of bone regeneration. J Am Vet Med Assoc. 2007; 231(7):1039-1055. [55] MOHD ZAFFARIN A, NG S, NG M, et al. Nano-hydroxyapatite as a delivery system for promoting bone regeneration in vivo: a systematic review. Nanomaterials (Basel). 2021;11(10):2569. [56] GARRISON K, SHEMILT I, DONELL S, et al. Bone morphogenetic protein (BMP) for fracture healing in adults. Cochrane Database Syst Rev. 2010;2010(6):CD006950. [57] DECAMBRON A, DEVRIENDT N, LAROCHETTE N, et al. Effect of the bone morphogenetic protein-2 doses on the osteogenic potential of human multipotent stromal cells- containing tissue engineered constructs. Tissue Eng Part A. 2019;25(7-8):642-651. [58] ZARA J, SIU R, ZHANG X, et al. High doses of bone morphogenetic protein 2 induce structurally abnormal bone and inflammation in vivo. Tissue Eng Part A. 2011;17(9-10):1389-1399. [59] DU X, XIE Y, XIAN C, et al. Role of FGFs/FGFRs in skeletal development and bone regeneration. J Cell Physiol. 2012;227(12):3731-3743. [60] XIE Y, SU N, YANG J, et al. FGF/FGFR signaling in health and disease. Signal Transduct Target Ther. 2020;5(1):181. [61] NAKAMURA K, KUROKAWA T, AOYAMA I, et al. Stimulation of bone formation by intraosseous injection of basic fibroblast growth factor in ovariectomised rats. Int Orthop. 1998;22(1):49-54. [62] KATO T, KAWAGUCHI H, HANADA K, et al. Single local injection of recombinant fibroblast growth factor-2 stimulates healing of segmental bone defects in rabbits. J Orthop Res. 1998;16(6):654-659. [63] NAKAMURA T, HARA Y, TAGAWA M, et al. Recombinant human basic fibroblast growth factor accelerates fracture healing by enhancing callus remodeling in experimental dog tibial fracture. J Bone Miner Res. 1998;13(6):942-949. [64] KAWAGUCHI H, JINGUSHI S, IZUMI T, et al. Local application of recombinant human fibroblast growth factor-2 on bone repair: a dose-escalation prospective trial on patients with osteotomy. J Orthop Res. 2007;25(4):480-487. [65] KAWAGUCHI H, OKA H, JINGUSHI S, et al. A local application of recombinant human fibroblast growth factor 2 for tibial shaft fractures: A randomized, placebo-controlled trial. J Bone Miner Res. 2010;25(12):2735-2743. [66] NEGRI A, SPIVACOW F. Healing of subtrochanteric atypical fractures after strontium ranelate treatment. Clin Cases Miner Bone Metab. 2012;9(3):166-169. [67] LIU F, WANG X, LI S, et al. Strontium-loaded nanotubes of Ti-24Nb-4Zr-8Sn alloys for biomedical implantation. J Biomed Nanotechnol. 2021;17(9):1812-1823. [68] DANG Y, ZHANG L, SONG W, et al. In vivo osseointegration of Ti implants with a strontium-containing nanotubular coating. Int J Nanomedicine. 2016;11: 1003-1011. [69] TSAI S, HSU Y, PAN W, et al. The effect of strontium-substituted hydroxyapatite nanofibrous matrix on osteoblast proliferation and differentiation. Membranes (Basel). 2021;11(8):624. [70] MA P, CHEN T, WU X, et al. Effects of bioactive strontium-substituted hydroxyapatite on osseointegration of polyethylene terephthalate artificial ligaments. J Mater Chem B. 2021;9(33):6600-6613. [71] HARRISON C, HATTON P, GENTILE P, et al. Nanoscale strontium-substituted hydroxyapatite pastes and gels for bone tissue regeneration. Nanomaterials (Basel). 2021;11(6):1611. [72] WU T, YANG S, LU T, et al. Strontium ranelate simultaneously improves the radiopacity and osteogenesis of calcium phosphate cement. Biomed Mater. 2019;14(3):035005. [73] TAO Z, ZHOU W, JIANG Y, et al. Effects of strontium-modified calcium phosphate cement combined with bone morphogenetic protein-2 on osteoporotic bone defects healing in rats. J Biomater Appl. 2018;33(1):3-10. [74] DING X, LI X, LI C, et al. Chitosan/dextran hydrogel constructs containing strontium-doped hydroxyapatite with enhanced osteogenic potential in rat cranium. ACS Biomater Sci Eng. 2019;5(9):4574-4586. [75] SØLLING A, HARSLØF T, LANGDAHL B. Current status of bone-forming therapies for the management of osteoporosis. Drugs Aging. 2019;36(7): 625-638. [76] CHEN J, SAMBROOK P. Antiresorptive therapies for osteoporosis: a clinical overview. Nat Rev Endocrinol. 2011;8(2):81-91. [77] GAO Y, LUO E, HU J, et al. Effect of combined local treatment with zoledronic acid and basic fibroblast growth factor on implant fixation in ovariectomized rats. Bone. 2009;44(2):225-232. [78] VAN DE VEN C, BAKKER N, LINK D, et al. Sustained release of ancillary amounts of testosterone and alendronate from PLGA coated pericard membranes and implants to improve bone healing. PLoS One. 2021;16(5):e0251864. [79] ALBANO C, GOMES A, DA SILVA FELTRAN G, et al. Bisphosphonate-based surface biofunctionalization improves titanium biocompatibility. J Mater Sci Mater Med. 2020;31(11):109. [80] MASTROGIACOMO S, KOWNACKA A, DOU W, et al. Bisphosphonate functionalized gadolinium oxide nanoparticles allow long-term MRI/CT multimodal imaging of calcium phosphate bone cement. Adv Healthc Mater. 2018;7(19):e1800202. [81] VERRON E, PISSONNIER M, LESOEUR J, et al. Vertebroplasty using bisphosphonate-loaded calcium phosphate cement in a standardized vertebral body bone defect in an osteoporotic sheep model. Acta Biomater. 2014;10(11):4887-4895. [82] GREINER SH, WILDEMANN B, BACK DA, et al. Local application of zoledronic acid incorporated in a poly(D,L-lactide)-coated implant accelerates fracture healing in rats. Acta Orthop. 2008;79(5):717-725. [83] KHAMIS A, ELSHARKAWY S. The influence of local delivery of bisphosphonate on osseointegration of dental implants. Evid Based Dent. 2018;19(3):82-83. [84] DEDE A, MAKRAS P, ANASTASILAKIS A. Investigational anabolic agents for the treatment of osteoporosis: an update on recent developments. Expert Opin Investig Drugs. 2017;26(10):1137-1144. [85] YANG Y, AGHAZADEH-HABASHI A, PANAHIFAR A, et al. Bone-targeting parathyroid hormone conjugates outperform unmodified PTH in the anabolic treatment of osteoporosis in rats. Drug Deliv Transl Res. 2017;7(4):482-496. [86] HENRIKSEN K, ANDERSEN J, RIIS B, et al. Evaluation of the efficacy, safety and pharmacokinetic profile of oral recombinant human parathyroid hormone [rhPTH(1-31)NH(2)] in postmenopausal women with osteoporosis. Bone. 2013; 53(1):160-166. [87] ZOU Z, WANG L, ZHOU Z, et al. Simultaneous incorporation of PTH(1-34) and nano-hydroxyapatite into Chitosan/Alginate Hydrogels for efficient bone regeneration. Bioact Mater. 2020;6(6):1839-1851. [88] WOJDA SJ, MAROZAS IA, ANSETH KS, et al. Thiol-ene hydrogels for local delivery of PTH for bone regeneration in critical size defects. J Orthop Res. 2020;38(3):536-544. [89] WOJDA S, MAROZAS I, ANSETH K, et al. Impact of release kinetics on efficacy of locally delivered parathyroid hormone for bone regeneration applications. Tissue Eng Part A. 2021;27(3-4):246-255. [90] DAVE J, DEWLE A, MHASKE S, et al. Hydroxyapatite nanorods loaded with parathyroid hormone (PTH) synergistically enhance the net formative effect of PTH anabolic therapy. Nanomedicine. 2019;15(1):218-230. [91] KOTHARI R, KUMAR V, JENA R, et al. Modes of degradation and impurity characterization in rhPTH (1-34) during stability studies. PDA J Pharm Sci Technol. 2011;65(4):348-362. [92] YAZAWA H, ZIMMERMANN B, ASAMI Y, et al. Simvastatin promotes cell metabolism, proliferation, and osteoblastic differentiation in human periodontal ligament cells. J Periodontol. 2005;76(2):295-302. [93] LIU S, BERTL K, SUN H, et al. Effect of simvastatin on the osteogenetic behavior of alveolar osteoblasts and periodontal ligament cells. Hum Cell. 2012;25(2):29-35. [94] MENDES JUNIOR D, DOMINGUES J, HAUSEN M, et al. Study of mesenchymal stem cells cultured on a poly(lactic-co-glycolic acid) scaffold containing simvastatin for bone healing. J Appl Biomater Funct Mater. 2017;15(2):e133-e141. [95] HAJIHASANI BIOUKI M, MOBEDI H, KARKHANEH A, et al. Development of a simvastatin loaded injectable porous scaffold in situ formed by phase inversion method for bone tissue regeneration. Int J Artif Organs. 2019;42(2):72-79. [96] ENCARNAÇÃO I, SORDI M, ARAGONES Á, et al. Release of simvastatin from scaffolds of poly(lactic-co-glycolic) acid and biphasic ceramic designed for bone tissue regeneration. J Biomed Mater Res B Appl Biomater. 2019;107(6):2152-2164. [97] FARUQ O, SAYED S, KIM B, et al. A biphasic calcium phosphate ceramic scaffold loaded with oxidized cellulose nanofiber-gelatin hydrogel with immobilized simvastatin drug for osteogenic differentiation. J Biomed Mater Res B Appl Biomater. 2020;108(4):1229-1238. [98] DELAN W, ZAKARIA M, ELSAADANY B, et al. Formulation of simvastatin chitosan nanoparticles for controlled delivery in bone regeneration: Optimization using Box-Behnken design, stability and in vivo study. Int J Pharm. 2020;577:119038. [99] ESKINAZI-BUDGE A, MANICKAVASAGAM D, CZECH T, et al. Preparation of emulsifying wax/glyceryl monooleate nanoparticles and evaluation as a delivery system for repurposing simvastatin in bone regeneration. Drug Dev Ind Pharm. 2018;44(10):1583-1590. [100] IYER S, DONNELLY P, SPANIEL G, et al. Locally applied simvastatin as an adjunct to promote spinal fusion in rats. Spine (Phila Pa 1976). 2019;44(15):1042-1048. [101] 邹伟龙,于龙,王亮,等.不同剂量辛伐他汀载药体系对兔骨质疏松模型骨修复的实验研究[J].中国骨质疏松杂志,2016,22(4):437-442. [102] TOMINARI T, AKITA M, MATSUMOTO C, et al. Endosomal TLR3 signaling in stromal osteoblasts induces prostaglandin E-mediated inflammatory periodontal bone resorption. J Biol Chem. 2022;298(3):101603. [103] CHENG H, HUANG H, GUO Z, et al. Role of prostaglandin E2 in tissue repair and regeneration. Theranostics. 2021;11(18):8836-8854. [104] LI M, KE H, QI H, et al. A novel, non-prostanoid EP2 receptor-selective prostaglandin E2 agonist stimulates local bone formation and enhances fracture healing. J Bone Miner Res. 2003;18(11):2033-2042. [105] PARALKAR V, BOROVECKI F, KE H, et al. An EP2 receptor-selective prostaglandin E2 agonist induces bone healing. Proc Natl Acad Sci U S A. 2003;100(11):6736-6740. [106] NAMIKAWA T, TERAI H, HOSHINO M, et al. Enhancing effects of a prostaglandin EP4 receptor agonist on recombinant human bone morphogenetic protein-2 mediated spine fusion in a rabbit model. Spine (Phila Pa 1976). 2007;32(21): 2294-2299. [107] KAMOLRATANAKUL P, HAYATA T, EZURA Y, et al. Nanogel-based scaffold delivery of prostaglandin E(2) receptor-specific agonist in combination with a low dose of growth factor heals critical-size bone defects in mice. Arthritis Rheum. 2011; 63(4):1021-1033. [108] LEE JH, YU CH, YANG JJ, et al. Comparative study of fusion rate induced by different dosages of Escherichia coli-derived recombinant human bone morphogenetic protein-2 using hydroxyapatite carrier. Spine J. 2012;12(3): 239-248. [109] ALSHAER W, ZUREIGAT H, AL KARAKI A, et al. siRNA: Mechanism of action, challenges, and therapeutic approaches. Eur J Pharmacol. 2021;905:174178. [110] LUNDSTROM K. Are viral vectors any good for RNAi antiviral therapy? Viruses. 2020;12(10):1189. [111] WHITEHEAD K, LANGER R, ANDERSON D. Knocking down barriers: advances in siRNA delivery. Nat Rev Drug Discov. 2009;8(2):129-138. [112] LIETMAN S, DING C, COOKE D, et al. Reduction in Gsalpha induces osteogenic differentiation in human mesenchymal stem cells. Clin Orthop Relat Res. 2005; (434):231-238. [113] SCHOOLMEESTERS A, EKLUND T, LEAKE D, et al. Functional profiling reveals critical role for miRNA in differentiation of human mesenchymal stem cells. PLoS One. 2009;4(5):e5605. [114] ZATSEPIN T, KOTELEVTSEV Y, KOTELIANSKY V. Lipid nanoparticles for targeted siRNA delivery - going from bench to bedside. Int J Nanomedicine. 2016;11:3077-3086. [115] XUE H, LIU S, WONG H. Nanotoxicity: a key obstacle to clinical translation of siRNA-based nanomedicine. Nanomedicine (Lond). 2014;9(2):295-312. [116] ZINTCHENKO A, PHILIPP A, DEHSHAHRI A, et al. Simple modifications of branched PEI lead to highly efficient siRNA carriers with low toxicity. Bioconjug Chem. 2008;19(7):1448-1455. [117] NGUYEN K, DANG P, ALSBERG E. Functionalized, biodegradable hydrogels for control over sustained and localized siRNA delivery to incorporated and surrounding cells. Acta Biomater. 2013;9(1):4487-4495. [118] SINGHA K, NAMGUNG R, KIM WJ. Polymers in small-interfering RNA delivery. Nucleic Acid Ther. 2011;21(3):133-147. [119] KANG HC, HUH KM, BAE YH. Polymeric nucleic acid carriers: current issues and novel design approaches. J Control Release. 2012;164(3):256-264. [120] ANDERSEN M, LICHAWSKA A, ARPANAEI A, et al. Surface functionalisation of PLGA nanoparticles for gene silencing. Biomaterials. 2010;31(21):5671-5677. [121] HEREDIA KL, NGUYEN TH, CHANG CW, et al. Reversible siRNA-polymer conjugates by RAFT polymerization. Chem Commun (Camb). 2008;(28):3245-3247. [122] ZHU J, GUO M, CUI Y, et al. Surface coating of pulmonary siRNA delivery vectors enabling mucus penetration, cell targeting, and intracellular radical scavenging for enhanced acute lung injury therapy. ACS Appl Mater Interfaces. 2022;14(4): 5090-5100. [123] GUO S, CÁZAREZ-MáRQUEZ F, JIAO H, et al. Specific silencing of microglial gene expression in the rat brain by nanoparticle-based small interfering RNA delivery. ACS Appl Mater Interfaces. 2022;14(4):5066-5079. [124] SIU YS, LI L, LEUNG MF, et al. Polyethylenimine-based amphiphilic core-shell nanoparticles: study of gene delivery and intracellular trafficking. Biointerphases. 2012;7(1-4):16. [125] KOZISEK T, HAMANN A, SAMUELSON L, et al. Comparison of promoter, DNA vector, and cationic carrier for efficient transfection of hMSCs from multiple donors and tissue sources. Mol Ther Nucleic Acids. 2021;26:81-93. [126] AYDIN O, KANARYA D, YILMAZ U, et al. Determination of optimum ratio of cationic polymers and small interfering RNA with agarose gel retardation assay. Methods Mol Biol. 2022;2434:117-128. [127] MA D. Enhancing endosomal escape for nanoparticle mediated siRNA delivery. Nanoscale. 2014;6(12):6415-6425. [128] RAMESAN S, REZK A, DEKIWADIA C, et al. Acoustically-mediated intracellular delivery. Nanoscale. 2018;10(27):13165-13178. [129] DEIRRAM N, ZHANG C, KERMANIYAN S, et al. pH-responsive polymer nanoparticles for drug delivery. Macromol Rapid Commun. 2019;40(10):e1800917. [130] JIN M, HOU Y, QUAN X, et al. Smart polymeric nanoparticles with pH-responsive and PEG-Detachable properties (II): Co-delivery of paclitaxel and VEGF siRNA for synergistic breast cancer therapy in mice. Int J Nanomedicine. 2021;16:5479-5494. |
| [1] | 吴韬光, 聂少波, 陈 华, 朱正国, 祁 麟, 唐佩福. 新型多维交叉锁定钢板固定股骨转子下骨不连的生物力学特征[J]. 中国组织工程研究, 2023, 27(9): 1330-1334. |
| [2] | 彭志鑫, 闫文刚, 王 坤, 张振江. 3D打印前臂外固定支具的有限元分析与结构优化设计[J]. 中国组织工程研究, 2023, 27(9): 1340-1345. |
| [3] | 郑宏瑞, 张文杰, 王云华, 何 斌, 沈亚骏, 范 磊. 股骨颈动力交叉钉系统联合富血小板血浆治疗股骨颈骨折[J]. 中国组织工程研究, 2023, 27(9): 1390-1395. |
| [4] | 郑 博, 张秀莉, 周 浩, 何泽壁, 周 进, 周维云, 李 鹏. 关节镜辅助下锁定空心螺钉固定与切开复位钢板内固定治疗Schatzker Ⅱ-Ⅲ型胫骨平台骨折的早期CT评价[J]. 中国组织工程研究, 2023, 27(9): 1410-1416. |
| [5] | 孙佳佳, 朱海迪, 卢 赟, 张 凯. 髋部骨折合并2型糖尿病和非2型糖尿病患者骨代谢标志物的比较[J]. 中国组织工程研究, 2023, 27(8): 1156-1160. |
| [6] | 赵 伟, 冯 卫, 杨铁翼, 任 伟, 王玉鑫, 吕慧成, 常志强, 冯晓东, 王紫横, 郭世炳. 3D打印模具制备抗生素骨水泥髓内钉治疗下肢长骨感染[J]. 中国组织工程研究, 2023, 27(7): 1023-1030. |
| [7] | 张 伟, 黄致超, 赵锐锋, 梁 欢, 马玉峰, 申艳光, 钟红刚, 陈兆军, 张继川, 陈卫衡. 杜仲胶夹板固定兔骨折模型的疗效评价[J]. 中国组织工程研究, 2023, 27(7): 1055-1061. |
| [8] | 徐 聪, 赵 赫, 孙 岩. 生物材料导管修复面神经损伤与再生[J]. 中国组织工程研究, 2023, 27(7): 1089-1095. |
| [9] | 陈世崧, 刘晓红, 徐志云. 人工生物瓣膜的研究现状及展望[J]. 中国组织工程研究, 2023, 27(7): 1096-1102. |
| [10] | 唐昊天, 廖荣东, 田 京. 压电材料修复骨缺损的应用及设计思路[J]. 中国组织工程研究, 2023, 27(7): 1117-1125. |
| [11] | 许 言, 李 平, 赖春花, 朱培君, 杨 烁, 徐淑兰. 血管化骨再生中压电生物材料的应用[J]. 中国组织工程研究, 2023, 27(7): 1126-1132. |
| [12] | 黄贵江, 季雨伟, 赵 鑫, 杨 艺, 赵玉兰, 王佩锦, 唐 薇, 角建林. 胎盘间充质干细胞不同给药途径治疗骨质疏松性骨折树鼩的疗效和机制[J]. 中国组织工程研究, 2023, 27(6): 909-914. |
| [13] | 秦宇星, 任前贵, 李子龙, 全嘉星, 沈佩锋, 孙 韬, 王浩宇. 骨微血管内皮细胞在股骨头坏死中的作用机制及前景[J]. 中国组织工程研究, 2023, 27(6): 955-961. |
| [14] | 许其静, 杨伊春, 雷 微, 杨 莹, 余 江, 夏婷婷, 张 萌, 章 涛, 张 潜. 糖尿病皮肤慢性创面无细胞治疗的进展与问题[J]. 中国组织工程研究, 2023, 27(6): 962-969. |
| [15] | 张 敏, 张晓明, 刘童斌. 柚皮苷在骨组织再生领域的应用潜力[J]. 中国组织工程研究, 2023, 27(5): 787-792. |
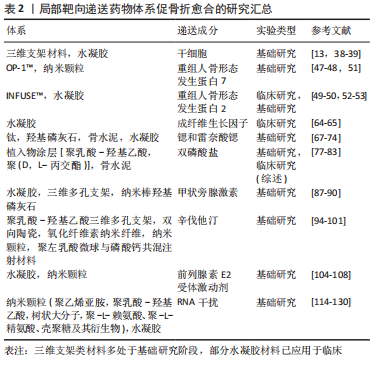
在骨折区域应用局部药物释放体系应考虑多种因素,如生物材料、促骨形成药物/活性蛋白、细胞治疗、外泌体等,可相互组合共同促进成骨。通过检索“骨折”“载药系统”的相关文献,将该文章相关的关键词出现频次整理,制作可视化词云图,为局部药物释放体系用于骨折治疗的多种方法做出直观简洁的可视化分析,见图1。
 中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 第一作者在2022年3月进行检索。
1.1.2 检索文献时限 1990年1月至2022年3月。
1.1.3 检索数据库 PubMed和中国知网(CNKI)数据库。
1.1.4 检索词 英文检索词为“drug delivery, fracture”,中文检索词为“载药系统,骨折”。
1.1.5 检索文献类型 研究性论文、综述性论文、著作。
1.1.6 手工检索情况 无。
1.1.7 检索策略 包括英文及中文检索词、检索词的逻辑组配等。以PubMed数据库为例,检索策略见图2。

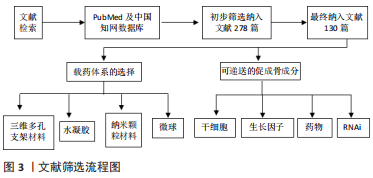
1.1.8 检索文献量 共检索到文献278篇,其中包括中文文献22篇,英文文献256篇。
1.2 纳入与排除标准
1.2.1 纳入标准 ①递送药物体系局部应用于骨折的实验性或基础研究、综述研究;②同一领域中论点、论据可靠的文献。
1.2.2 排除标准 ①重复性研究且与研究目的无相关性的文献;②逻辑不严谨可信度差的文献。
1.3 质量评估与数据提取 共检索到文献278篇,其中英文文献256篇,中文文献22篇,排除与研究目的相关性差及内容陈旧、重复的文献148篇,纳入130篇符合标准的文献进行综述,筛选流程见图3。
 3.2 作者综述区别与他人他篇的特点 骨骼类疾病及损伤影响了大量的人群。虽然目前已经批准了几种蛋白类药物治疗骨骼类疾病,但这些药物用于治疗局部骨折均可能会导致严重的不良反应。骨折愈合修复是一个复杂的生理过程,包括炎症免疫、血管生成、成骨分化和生物矿化早期调节。目前,尚未设计出针对骨折愈合不同阶段的特点,次序性给予相应刺激因子的临床治疗方案。未来研究可根据骨折愈合的各阶段重点不同,选择特定的治疗方法和药物输送系统。通过评估损伤程度、愈合潜力和每个病例的具体要求,可以将药物递送系统按各种方式组合,形成多途径治疗的协同疗法,以最大限度促进骨折愈合。文章根据生物材料和药物递送领域的最新进展,概述当前骨折区局部载药体系的治疗方法及未来的研究方向,将有效的治疗剂与理想的递送系统相结合,为将来药物递送体系促骨折愈合的临床应用提供一定的参考价值。
3.2 作者综述区别与他人他篇的特点 骨骼类疾病及损伤影响了大量的人群。虽然目前已经批准了几种蛋白类药物治疗骨骼类疾病,但这些药物用于治疗局部骨折均可能会导致严重的不良反应。骨折愈合修复是一个复杂的生理过程,包括炎症免疫、血管生成、成骨分化和生物矿化早期调节。目前,尚未设计出针对骨折愈合不同阶段的特点,次序性给予相应刺激因子的临床治疗方案。未来研究可根据骨折愈合的各阶段重点不同,选择特定的治疗方法和药物输送系统。通过评估损伤程度、愈合潜力和每个病例的具体要求,可以将药物递送系统按各种方式组合,形成多途径治疗的协同疗法,以最大限度促进骨折愈合。文章根据生物材料和药物递送领域的最新进展,概述当前骨折区局部载药体系的治疗方法及未来的研究方向,将有效的治疗剂与理想的递送系统相结合,为将来药物递送体系促骨折愈合的临床应用提供一定的参考价值。
3.3 综述的局限性 文章重点在于阐述促成骨因子和载药体系,然而并未对药物或分子释放的时效性进行讨论。
3.4 综述的重要意义与展望 到目前为止,还没有一种能够促进骨折修复并降低骨折相关并发症风险的非侵入性疗法获得批准,包括RNA干扰疗法和小分子化合物在内的新方法也正在开发中,以规避传统生长因子的挑战。研究表明,药物递送系统改变了治疗剂的生物分布,此外,这些局部递药系统增强了骨桥形成,并减少了不良反应。随着生物材料和药物输送技术的不断发展,这些技术在再生医学领域的应用将不断扩大,最终带来促进骨折愈合的新工具。
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
 #br#
#br#
文题释义:
局部载药体系:给药途径有2种,分别为全身系统性给药和局部给药。传统的全身给药途径是药物通过循环系统递送,会导致如全身毒性、肝肾并发症、药物相互作用、药物达到靶器官的活性及剂量低以及患者依从性低等问题。而局部载药体系将药物递送到作用的靶器官,具有靶器官浓度高、不良反应小以及全身吸收少等优势。文章主要针对骨折愈合的药物递送体系,故而研究重点为术中植入材料负载活性因子的应用。近年来,由于组织工程技术的发展以及载药体系的精细化与可控化,使骨折区局部用药研究成为热点。在骨折区域局部应用药物释放体系,解决药物释放障碍,如药物溶解性差、半衰期短和靶向效应。文章就目前采用的载药体系和促成骨因子的选择做出总结综述,旨在为后续研究者提供促进骨折修复的相应研究思路。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||