中国组织工程研究 ›› 2023, Vol. 27 ›› Issue (21): 3430-3437.doi: 10.12307/2023.131
• 生物材料综述 biomaterial review • 上一篇 下一篇
医用含铜钛合金抗菌性能的研究与进展
申 勇1,刘时璋2
- 1西安医学院,陕西省西安市 710021;2陕西省人民医院骨科,陕西省西安市 710068
-
收稿日期:2022-04-01接受日期:2022-05-11出版日期:2023-07-28发布日期:2022-11-26 -
通讯作者:刘时璋,博士,硕士生导师,主任医师,教授,陕西省人民医院骨科,陕西省西安市 710068 -
作者简介:申勇,男,1996年生,陕西省渭南市人,汉族,西安医学院在读硕士,主要从事医用钛合金抗菌性能及生物相容性研究。 -
基金资助:陕西省重点研发计划项目(2018ZDXM-ZF-057),项目负责人:刘时璋
Research progress on antibacterial properties of medical copper-containing titanium alloys
Shen Yong1, Liu Shizhang2
- 1Xi’an Medical College, Xi’an 710021, Shaanxi Province, China; 2Department of Orthopedics, Shaanxi Provincial People’s Hospital, Xi’an 710068, Shaanxi Province, China
-
Received:2022-04-01Accepted:2022-05-11Online:2023-07-28Published:2022-11-26 -
Contact:Liu Shizhang, MD, Master’s supervisor, Chief physician, Professor, Department of Orthopedics, Shaanxi Provincial People’s Hospital, Xi’an 710068, Shaanxi Province, China -
About author:Shen Yong, Master candidate, Xi’an Medical College, Xi’an 710021, Shaanxi Province, China -
Supported by:the Key Research & Development Project of Shaanxi Province, No. 2018ZDXM-ZF-057 (to LSZ)
摘要:
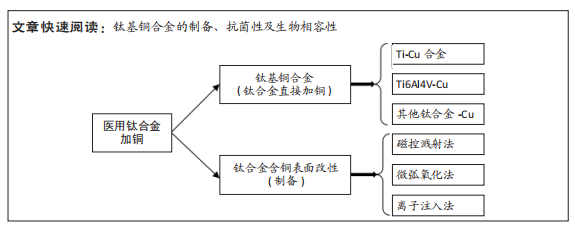
文题释义:
医用钛合金:用于制造植入人体内的医疗器件、假体、辅助治疗设备的钛合金,生物力学性能接近人骨,还具备耐疲劳、耐腐蚀及良好的生物相容性等优势。细菌生物膜:细菌黏附于表面分泌多糖基质、纤维蛋白、脂质蛋白等,将其自身包绕其中而形成的大量细菌聚集膜样物,是为适应自然环境有利生存的一种生命现象。
背景:作为骨科内植物的主要材料,钛及其合金在包括骨科在内的多个医学领域得到广泛的应用,内植物相关性感染也随之增多,采取不同的制备方法将优异且机制清楚的抗菌金属铜加入其中,在不影响生物力学性能的同时可使其具备一定的抗菌能力。
目的:综述医用含铜钛合金的抗菌性能研究进展及发展趋势。
方法:应用计算机检索PubMed、Web of Science数据库中2003年1月至2022年3月的相关文献,检索词为“Titanium alloy,Copper,Antibacterial,Surface modification,Coating”,根据纳入排除标准对选定的76篇文献进行分析讨论。
结果与结论:①加入铜的钛合金在体内外抗菌实验中均显示出良好的抗菌性能,并随着铜含量的提升其抗菌性能更加明显。②但目前仍存在较多问题有待解决:探索、优化加入铜的含量、方式及采取合适的制备工艺使合金的抗菌性能、生物相容性、机械性能达到平衡需要进一步的实验;表面改性对合金表面的微损伤是否会对合金造成不利影响;铜离子释放速率的控制以及如何避免早期突然大量释放铜离子的“突释现象”。
https://orcid.org/0000-0003-0526-9589(刘时璋)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
中图分类号:
引用本文
申 勇, 刘时璋. 医用含铜钛合金抗菌性能的研究与进展[J]. 中国组织工程研究, 2023, 27(21): 3430-3437.
Shen Yong, Liu Shizhang. Research progress on antibacterial properties of medical copper-containing titanium alloys[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3430-3437.
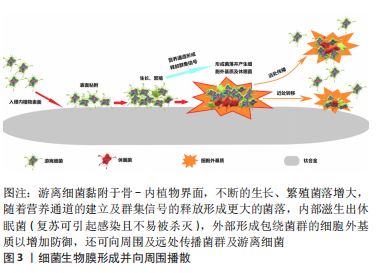
2.1.1 细菌生物膜的形成过程 植入物相关感染的发生与细菌生物膜的形成有直接关系[16]。游离的细菌微生物黏附于骨、钛合金及周围软组织未被人体免疫力或抗生素杀灭,进而生长、繁殖逐渐形成菌落。随着细菌内外毒素的释放并产生大量的细胞外基质,菌落的群集感应通道及营养通道进一步形成后使得更多细菌聚集,表面覆盖着细胞外基质以及内部存在的休眠菌的更大菌落相互连接成具有三维结构的细菌生物膜[17]。细菌生物膜对各种抗菌剂有强大的防御能力,而且还可作为细菌母体进向远处传播细菌种子引起更多部位的感染(图3)。常规治疗植入物相关感染多在细菌生物膜形成之后,所以通常难以获得期望的效果,因此抑制游离细菌的侵袭为内植物植入部位的周围创造一个无菌环境十分重要。
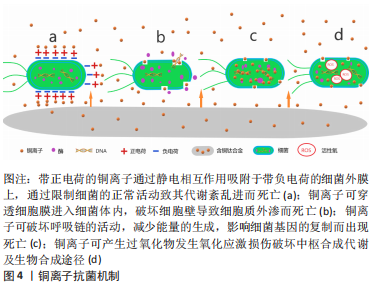
2.1.2 铜的抗菌机制 铜作为人体内必需的微量元素以其良好的生物相容性成为美国环境保护署注册的第一个抗菌金属材料,而且足够浓度的铜离子可以在2 h内杀灭99%以上的细菌[18]。抗菌机制:①带正电荷的铜离子通过静电相互作用吸附于带负电荷的细菌外膜上,通过限制细菌的正常活动致其代谢紊乱进而死亡(图4a);②铜离子可穿透细胞膜进入细菌体内,破坏细胞壁导致细胞质外渗而死亡(图4b);③铜离子可破坏呼吸链的活动,减少能量的生成,影响细菌基因的复制而出现死亡(图4c);④铜离子可产生过氧化物发生氧化应激损伤破坏中枢合成代谢及生物合成途径[19](图4d)。加入铜元素的钛合金材料不仅可以杀灭与材料直接接触的细菌,而且游离的铜离子对周围一定范围内的细菌仍有杀伤作用,抑制了游离细菌对钛-骨界面的“表面竞争”,有效地阻止了细菌生物膜的形成。
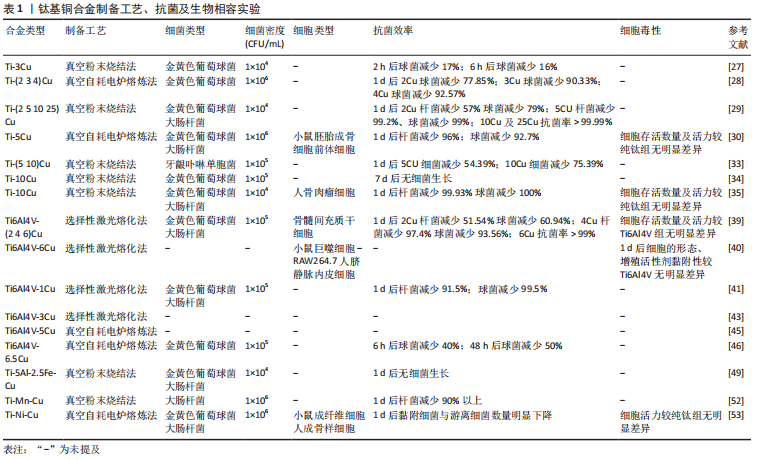
2.2 钛基铜合金
2.2.1 Ti-Cu合金 纯钛在医学中的首次报道在1940年,当时基于对动物骨骼与多种金属植入物的反应进行测试的结果,发现这种金属与骨骼可以很好地匹配[20]。在随后的十年中对纯钛进行了大量的生物相容性、耐腐蚀性、耐磨性等实验,而且随着钛的制造工艺越来越完备,这些也为钛的医学应用研究铺平了道路。纯钛制备的钢板、螺钉、克氏针等内植物以其良好的综合性能被广泛用于骨科手术中[21]。而为预防内植物相关感染加入具有良好抗菌性的金属铜的钛-铜二元合金备受关注。钛-铜合金中铜的含量及合金的加工过程会影响铜的存在形式以及铜离子的释放行为,进而影响合金的力学性能和抗菌性能。研究发现铜离子在生物环境中的释放,特别是在酸性和含氟条件下的释放,可产生更加优异的抗菌性能[22]。纯钛中钛是以α相存在的,而铜是钛β相的稳定剂,铜含量越高β相钛的占比量越大。β相钛较α相钛有更高的强度,强度和可塑性是互斥的,所以随着铜含量的增加,可塑性会有所降低[23]。实验表明铸态Ti-Cu合金的硬度和机械强度较高,抗菌性能也较为可观,但耐腐蚀性及可塑性较纯钛低;而900 ℃加热处理2 h后可以提高钛铜合金的耐腐蚀性,但对抗菌性能影响不大,900 ℃热处理2 h后再用400 ℃热处理12 h进一步提高了硬度、机械强度、耐腐蚀性,还显著提高了抗菌性能[24]。固溶态的铜可以强化合金的机械性能,但抗菌性能较差,这是由于抗菌能力因Ti2Cu相的多少决定,其不仅强化合金性能,而且还可以显著提升抗菌性能[24]。
钛-铜二元合金中3%质量占比的铜是是对金黄色葡萄球菌及大肠杆菌达到90%抗菌率的最小值,而且Ti2Cu相对细菌有较强的抗黏附性及耐腐蚀能力[25-26]。实验表明,为了获得99%的抗菌率,钛-铜合金中铜含量至少需要5%或铜离子平均释放浓度达到0.036 mg/L。LIU等[27]通过对Ti-(2.5 5 10 25)Cu研究表明,当铜含量达到合金占比5%以上时,释放出的铜离子才能获得对金黄色葡萄球菌和大肠杆菌强大而稳定的抗菌性能,并且发现Ti-5Cu中Ti2Cu相较Ti-2.5Cu明显增加,所以考虑铜离子释放与Ti2Cu富铜相有关。MA等[28]发现Ti-5Cu合金中Ti2Cu相的沉淀提高了合金的硬度及耐磨性,而且铜离子累计释放浓度远低于WHO推荐的人体每天铜摄入量,因此认为Ti-5Cu具有良好的生物相容性。LIU等[29]也认为Ti-5Cu的铜质量是力学性能、生物相容性、抗菌性能等综合性能的最佳选择,可通过杀灭细菌、抑制细菌黏附明显抑制高密度细菌产生的细菌生物膜厚度,而且相对纯钛具有良好的骨结合及较低的骨吸收作用,可降低植入失败的风险。在Ti-(1 3 5 10)Cu合金对比研究中发现,Ti-10Cu是唯一在仅2 h后就表现出抗菌效果的合金,而且随着铜的质量分数升高合金的硬度逐渐增加,这可能与合金在添加铜后出现的钛β相结以及Ti2Cu、Ti3Cu相有关,而且研究发现释放的铜离子不仅可以杀灭细菌还可抑制存活细菌的活性[30-31]。WANG等[32]将Ti-10Cu合金植入感染金黄色葡萄球菌引起的骨髓炎兔股骨髓腔中,通过在1,4,7,14 d对植入部位的伤口观察、白细胞及感染部位提取液进行细菌培养计数、股骨骨质的苏木精-伊红染色,发现相较于纯钛组,Ti-10Cu仅在第4天有轻微炎症反应,而且7 d以后的提取液菌落计数皆为0。Ti-10Cu合金对金黄色葡萄球菌及大肠杆菌24 h后的抑菌率高达100%,然而Ti-10Cu合金可塑性明显低于纯钛这一点较为不利,而且过量的铜摄入会导致胃部不适、恶心和腹泻,并可能导致组织损伤和疾病,但是Ti-10Cu合金的生物相容性实验早期培养阶段表现出无明显细胞毒性反而会促进碱性磷酸酶活性提升成骨能力[33]。
钛铜合金作为研究最为广泛的抗菌钛合金,无论是体内或体外实验都表明合金中铜含量的增加可以明显提升抗菌性能,但相较于纯钛,铜的加入使得合金的可塑性及耐腐蚀有所影响。虽然较低铜质量(3%-5%)的钛铜合金抗菌效率相对较低,但临床通常对于严重无污染开放性骨折采取多次清创、骨折患者合并的多种基础疾病进行及时的干预、调整后,潜在感染的风险或者存在细菌的数量已经显著降低。结合预防性抗生素的使用,所以具备一定抗菌能力且不会对合金本身生物力学性能造成严重影响的低铜质量钛铜合金才更适合临床。因此低铜质量合金通过制备工艺、加工技术等方面的改进使得抗菌性能与机械性能之间达到良好平衡的研究需要更进一步探索。
2.2.2 Ti6Al4V-Cu合金 Ti6Al4V作为α+β相钛合金的代表,最初被应用于航空航天工业材料,而良好的生物相容性、卓越的机械强度、优异的耐腐蚀性以及较低的弹性模量使其成为合适的生物医用材料[34-35]。加入铜元素可以增加Ti6Al4V合金β相的含量,进一步降低弹性模量使其更加贴近于骨皮质并强化其机械性能,而且可以赋予合金抗菌能力[36]。加入铜的合金形成的Ti2Cu相对合金在力学性能和抗菌性能中起着重要作用,而且实验表明细菌更喜欢黏附在钛α相区域,而不是β相区域,这可能与铜促进并稳定β相的形成有关。而且,通过加入不同含量的铜元素以及采取不同的加工工艺可以调整、强化甚至改变Ti6Al4V-Cu合金的单一或整体性能。GUO等[37]采用选择性激光熔融技术使金属粉末快速成型为合金成品,研究表明所制备的Ti6Al4V合金中添加铜可提高其金属离子释放率,而且铜含量达到6%也具有良好的生物相容性。同样采取激光熔融技术制备的Ti6Al4V-6Cu合金不仅可以增强血管生成作用,并通过抑制巨噬细胞降低了局部炎症反应,发现铜元素的加入使得合金出现微孔,并随着铜含量的增加孔隙率和β相逐渐增加,弹性模量的下降可能与其有关[38]。KRAKHMALEV等[39]采用激光粉末床融合原位合金化工艺制备的Ti6Al4V-Cu合金显著减少了大肠杆菌和金黄色葡萄球菌的纯培养物的生长,而且硬度较于普通Ti6Al4V合金有明显的提高。铜含量的增加自然可以提高合金的抗菌性能,但可塑性、耐腐蚀性等性能会有所牺牲。对Ti6Al4V-Cu合金的机械性能研究表明合金的可塑性随铜质量分数的增加而降低,并且当添加10%铜时合金甚至会出现脆性断裂,而且KIKUCHI等[40]指出,随着铜质量分数从1% 增加到10%,Ti-Cu合金的可塑性从7%降低到1%。而同样采取激光粉末床融合原位合金化技术制备Ti6Al4V-3Cu抗菌性能更加明显,并且指出通过原位合金化 Ti6Al4V和纯铜粉末通过拓扑优化其晶格结构使合金保持良好的机械性能[41]。为了避免合金可塑性的降低影响合金的整体性能,WANG等[42]采取“共析元素合金化-淬火-热变形”策略制备的Ti6Al4V-5Cu合金产生大量的纳米级α相晶粒,而且Ti2Cu相及β相快速共析沉淀沿晶界形成“双相蜂窝壳”结构稳定了α晶粒,极大提升了合金的强度,此外可塑性较Ti6Al4V提升约60%。ZHANG等[8,43]也对其制备的Ti6Al4V-5Cu骨板、螺钉等内植物进行拉伸、扭转、弯曲等力学性能检测,发现较Ti6Al4V皆有明显提升。有研究在不同的加热温度(700-910 ℃)下对Ti6Al4V-5Cu合金进行退火处理,发现加热740 ℃退火处理后的Ti6Al4V-5Cu合金有着综合性能的最佳表现;740 ℃的加热温度在对照组(Ti6Al4V)及实验组(Ti6Al4V-6.5Cu)进行退火处理后进行体内外抗菌试验发现,实验组可明显抑制细菌生物膜的形成,监测骨缺损感染大鼠的体温、伤口外观、血清学生化等指标无明显炎症表现,而且组织学及影像学表明Ti6Al4V-6.5Cu具有明显的促进成骨能力[44]。
然而,Ti6Al4V至今为止仍广泛应用于临床中主要依赖其良好的生物相容性及优异的机械性能,因此加入铜获得抗菌效率进而影响其机械性能是得不偿失的。而且Ti6Al4V的元素成分较纯钛复杂,铜的加入对合金的微观结构及金属相影响及重构会更加明显,反馈影响宏观生物力学性能变化亦较大。实验表明,当铜加至一定占比时才具备抗菌性能,而且根据临床数据统计,当前闭合性骨折或感染评分较低的骨折不在少数,所以Ti6Al4V合金仍大有用处,因此Ti6Al4V合金加铜使其获得抗菌性能是否适用于临床有待实验进一步证明。
2.2.3 其他含铜钛合金 为了进一步优化钛合金的综合性能,通过加入其他金属元素制备的钛合金既可以满足作为内植物的需求,又可以达到较纯钛更加优异的机械性能且并不含有毒元素。因此,许多研究人员正在尝试开发更安全、适用钛合金材料,并通过加入具有抗菌作用的铜元素使合金材料的机械性能及抗菌性能达到良好的平衡[45-46]。YAMANOGLU等[47]认为Ti-5Al-2.5Fe(Ferrum)与Ti6Al4V合金的机械性能无明显差异,而且加入铜元素的Ti-5Al-2.5Fe-Cu具备了抗菌性能,随着铜的加入产生Ti2Cu相,这种富铜相对的存在是与合金的抗菌能力呈正相关的。此外,当添加的铜质量达到3%或更多时,Ti-Al相会逐渐减少,而Ti-Cu相的增多使得合金硬度和弯曲强度得到显著改善。体外实验表明加入铜元素的合金可防止样品其表面形成大肠杆菌和金黄色葡萄球菌菌落,而且合金表现出良好的细胞生物相容性[47]。锰(Manganese,Mn)和铜作为廉价的金属元素具有稳定钛合金β相的作用,这样既可以降低制造成本也使得合金的制备更接近最终形状的结构假体。虽然锰以其细胞毒性闻名,但是通过对Ti-Mn合金的机械行为和细胞毒性研究发现,合金表面形成的氧化层可明显降低锰元素的细胞毒性[48-49]。通过粉末烧结工艺并进行冷压处理制备Ti-Mn-Cu合金,研究表明其具有更细片层和逐渐细化的片层结构,并可以防止形成和析出会使材料脆化的次级相。因此锰和铜的加入既可以增加合金的静态载荷抗性,也使得合金具备抗菌能力,但合金延展性的降低使得合金耐受应力而不发生永久变形的能力下降[50-51]。而钛镍(Nickel,Ni)记忆合金具备传统生物医学金属合金所不具备的独特特性,因为它们能够表现出高弹性变形或者称为“伪弹性”和“形状记忆效应”。LI等[52]对Ti-Ni-Cu合金研究发现,在添加铜合金元素后仍保持良好的形状记忆效应,在具备一定抗菌性能同时,耐腐蚀性及细胞相容性也相当不俗。
所有的内植物材料都不会尽善尽美,相应的金属元素加入使钛合金在机械性能、生物相容性、耐腐蚀性等这些植入体内必备的特性得到改善。作为使其具备抗菌性能的铜元素加入这些多元素组成的钛合金中,是否会对原本良好的综合性能产生不利影响仍有待研究证明。而且金属元素种类越丰富所需要考虑金属相互之间的影响及元素占比问题显得格外重要,这将直接影响合金最终是否能服务患者的最终结果。钛基铜合金制备工艺、细菌实验、生物相容性实验汇总,见表1。
2.3 钛合金的含铜表面处理
2.3.1 表面处理介绍 表面处理是在不破坏材料自身具备的整体性能条件下仅对材料表面进行处理,使其获得需要在表面或接触外界环境的界面发挥改性所赋予的性能的一项技术。表面处理一般分为两大类——表面改性技术和涂层技术[53-55]。表面改性意味着材料结构临近表面部分被改性,这种技术可以在原子、分子等结构尺度上进行,对材料本身的综合性能并不会造成大的影响;而涂层技术则是在基材表面进行功能涂层的附着,在表面上形成涂层可以通过各种技术来实现,这些技术又可以是物理的、化学的或两者的组合。而随着内植物材料应用的日趋增多,发生内植物相关性感染的病例也随之水涨船高,治疗感染所需高昂的花销及较差的预期使得钛合金植入体内具备一定的抗菌性能是有必要的[56]。众所周知钛合金以其优异的机械性能及生物相容性见长,并随着对合金的认识加深以及加工工艺进步更加个体化的运用使其综合性能更加适用于多个临床科室[57-58]。金属铜具备良好抗菌能力而作为基础成分加入钛合金后虽然可以使其具备一定的抗菌性能,但为达到预定的抗菌程度随着铜含量增加不可避免的会对钛合金性能造成影响,对钛合金进行含铜的表面处理可以很好地规避这一缺点。而且随着表面处理工艺的进步更加精确且易控制的微弧氧化法、磁控溅射法、离子注入法等方法的应用使表面含铜钛合金在保持本身性能的同时具备抗菌性能,并通过控制铜的含量及释放速率并不会影响合金的生物相容性[59-61]。
2.3.2 微弧氧化法 微弧氧化法主要依靠电解液与电参数进行匹配调节,利用弧光放电产生的瞬时高温、高压在合金表面形成以基材为主的氧化物涂层并辅以电解质液组分的改性涂层,以获得涂层所赋予的功能特点,见图5。
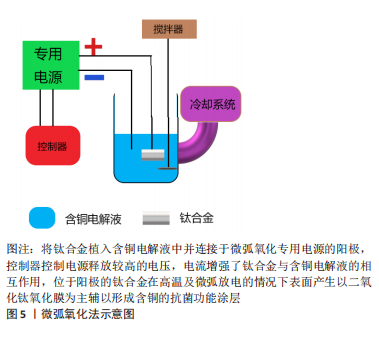
KOMAROVA等[62]在Ti-40Nb(Niobium)合金上通过微弧氧化法制备的含锌和铜的磷酸钙涂层具有良好的亲水性,实验表明涂层与液体的接触角不超过45°,可明显抑制细菌的黏附,而且电压的增加导致涂层粗糙度和孔隙率的增加,这是导致水接触角降低以及成骨细胞活性提升的直接原因,而且铜和锌的加入与水接触角的降低更加使合金具备了良好的抑菌性能。WANG等[63-65]采用微弧氧化法制备的钛基铜-二氧化钛涂层在体内、外及组织学实验中也验证了这一点,但合金孔隙率的形成及大小分布对新骨形成有一定影响,实验表明当电解质液中铜离子所含元素浓度达到3.0%时才显示出一定的细胞毒性,因此在选择铜的添加方式及浓度时细胞毒性是整个合金应用的关键之处。这种加入其他的抗菌元素以避免高浓度铜带来的细胞毒性还可达到预期的抗菌效果,这种思路不失为可行之举。VAN HENGEL等[66]将铜和银(Argentum)纳米粒子通过微弧氧化法加载于钛合金表面的二氧化钛微孔中,铜加银组既可以协同杀菌,在达到同样抗菌效果前提下使得相应离子浓度下降数倍,获得良好的生物相容性的同时在多重抗菌机制的作用下抗菌范围可能更广。在二氧化钛微孔中加载Cu-Mg(Magnesium)-F(Fluorine)纳米粒子获得抗菌性能的同时其他功能性粒子的加入同样可以达到效果,而且适当的镁离子能促进钙盐的沉积,钙盐则是骨组织生长必不可少的元素,氟粒子可以通过刺激成骨细胞增殖和增强碱性磷酸酶活性直接促进成骨[67]。通过改善工艺,加入超声波处理的微弧氧化可以提升涂层厚度分布的均匀性、结合强度和阳离子沉积量,提高材料表面的生物活性,进一步强化涂层的功能特点[68]。目前微弧氧化法在医用钛合金材料的研究应用较多,但多集中于防腐、耐磨、成骨等研究,加入金属抗菌元素使其具备抗菌性能的研究仍然较少。
2.3.3 磁控溅射法 磁控溅射法作为物理气相沉积的一种,采取真空状态利用荷能粒子轰击靶材料表面,使得轰击出的粒子沉积在基材表面,溅射所获得的涂层与基材结合好可以更好地控制镀层的厚度及均匀性,沉积速度快、基材温升低等特点对涂层损伤较小,其方法原理决定了既可应用于大多数材料也易于实现工业化,这些优势使磁控溅射法可以很好地胜任钛合金表面含铜涂层的涂覆并加以推广,见图6。
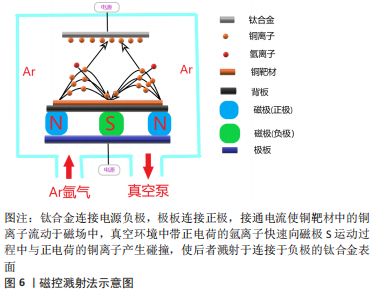
DING等[69]采取磁控溅射法在Ti6Al4V表面形成的铜-钽(Tantalum,Ta)的氧化复合涂层可显著提升材料表面的粗糙度及疏水性,在具备一定抗菌性的同时氧化涂层也可提升植入物的耐腐蚀性;JIN等[70]的实验也同样验证这一点,抗菌试验表明革兰阴性杆菌相较于革兰阳性球菌对铜离子更敏感,尽管铜元素的含量可能不同,但更薄更易穿透的细胞壁的确使得阴性杆菌更易被杀伤。磁控溅射在钛合金基材表面的铜-锆(Zirconium,Zr)氧化涂层具有更好的结晶性和疏水性,在模拟体液的湿滑动条件下耐磨性比干燥滑动有了显著提高。由于结晶度、晶粒特征和机械性能的改善,含Cu-Zr涂层钛合金的耐腐蚀性也显著增强,而且抗菌试验显示涂层对铜绿假单胞菌均表现出良好的抗菌活性[71]。ALHUSSEIN等[72]通过磁控溅射法为β型钛合金Ti-23Nb-0.7Ta-2Zr(TNTZ)制备了超弹性含铜钛基薄膜,实验表明与铜含量有关的非晶相的形成导致了机械性能的提高,而且发现铜质量分数达到8.3%时表现出最强的灭菌活性。然而磁控溅射法对基材利用率不高,等离子体不稳定等特点也限制其发展,而且对钛合金的含铜涂层制备无论是铜含量的优化、生物相容性的平衡以及加入铜元素对机械性能的影响有待进一步研究。
2.3.4 离子注入法 离子注入法是在真空系统中,对需要掺杂原子的离子经过加速注入材料表面,在所选择的特定区域形成特定的表层,见图7。作为最初应用半导体材料的技术,离子注入的纯净度较高,注入的深度、剂量可控性强,也可以处理各种不规则形状的基材所以应用也较广[73]。
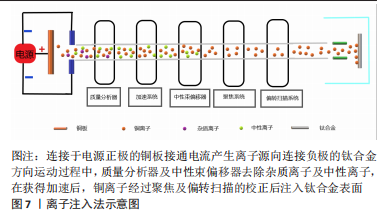
通过利用离子注入和沉积技术对钛合金表面进行铜-碳(Carbon,C)注入改性,结果表明表面的铜-碳离子可以形成电偶腐蚀对,其中铜作为阳极、碳作为阴极从而保护合金表面的防腐性能,而且电偶腐蚀对还可以控制铜离子的释放更有利保持良好的生物相容性[74]。将铜和氮(Nitrogen,N)注入钛合金表面,电镜观察在合金的表面形成了嵌入有铜纳米颗粒的氮化钛薄膜。而Cu-N的电偶腐蚀对在提升耐腐蚀性同时也可以有效地控制铜离子的释放,从而增强合金表面的抗菌,而且还具有刺激血管生成作用;在加入锌(Zinc,Zn)后同样可以提升耐腐蚀性,在铜、锌离子的扩散和相互作用下减少了相应离子积累降低的细胞毒性的同时具有良好的抑菌能力[75-76]。离子注入法对钛合金进行含铜表面改性使其具有抗菌性能的同时还可以改善合金的耐腐蚀性,使合金可以提升体内复杂环境的适应度,但相应抗菌研究内容的匮乏,也禁锢了发展。
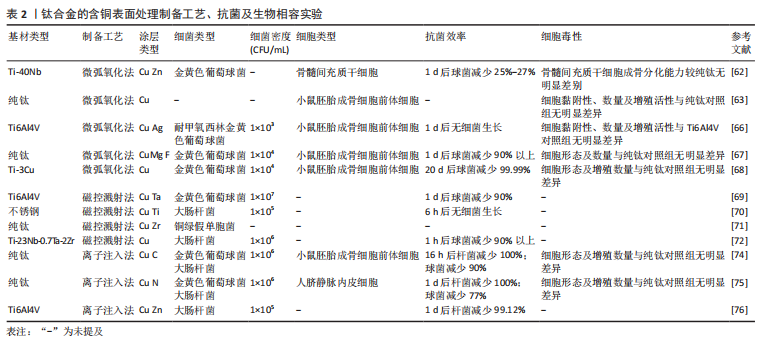
钛合金的含铜表面处理制备工艺、细菌实验、生物相容性实验汇总,见表2。
| [1] ZHANG Y, CHU K, HE S, et al. Fabrication of high strength, antibacterial and biocompatible Ti-5Mo-5Ag alloy for medical and surgical implant applications. Mater Sci Eng C Mater Biol Appl. 2020;106:110165. [2] AZMAT A, TUFAIL M, CHANDIO AD. Synthesis and Characterization of Ti-Sn Alloy for Orthopedic Application. Materials (Basel). 2021; 14(24):7660. [3] STEPANOVSKA J, MATEJKA R, ROSINA J, et al. Treatments for enhancing the biocompatibility of titanium implants. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2020;164(1):23-33. [4] BARBERI J, SPRIANO S. Titanium and Protein Adsorption: An Overview of Mechanisms and Effects of Surface Features. Materials (Basel). 2021;14(7):1590. [5] DIEFENBECK M, SCHRADER C, GRAS F, et al. Gentamicin coating of plasma chemical oxidized titanium alloy prevents implant-related osteomyelitis in rats. Biomaterials. 2016;101:156-164. [6] KOMISAR V, ROBINOVITCH SN. The Role of Fall Biomechanics in the Cause and Prevention of Bone Fractures in Older Adults. Curr Osteoporos Rep. 2021;19(4):381-390. [7] REIDER L, POLLAK A, WOLFF JL, et al. National trends in extremity fracture hospitalizations among older adults between 2003 and 2017. J Am Geriatr Soc. 2021;69(9):2556-2565. [8] PENG C, ZHANG S, SUN Z, et al. Effect of annealing temperature on mechanical and antibacterial properties of Cu-bearing titanium alloy and its preliminary study of antibacterial mechanism. Mater Sci Eng C Mater Biol Appl. 2018;93:495-504. [9] WALTER N, RUPP M, LANG S, et al. The epidemiology of fracture-related infections in Germany. Sci Rep. 2021;11(1):10443. [10] METSEMAKERS WJ, KORTRAM K, FERREIRA N, et al. Fracture-related outcome study for operatively treated tibia shaft fractures (F.R.O.S.T.): registry rationale and design. BMC Musculoskelet Disord. 2021;22(1):57. [11] CLYNES MA, HARVEY NC, CURTIS EM, et al. The epidemiology of osteoporosis. Br Med Bull. 2020;133(1):105-117. [12] WANG R, SHI M, XU F, et al. Graphdiyne-modified TiO2 nanofibers with osteoinductive and enhanced photocatalytic antibacterial activities to prevent implant infection. Nat Commun. 2020;11(1):4465. [13] DA SILVA RAG, AFONINA I, KLINE KA. Eradicating biofilm infections: an update on current and prospective approaches. Curr Opin Microbiol. 2021;63:117-125. [14] JIANG N, WANG BW, CHAI YM, et al. Chinese expert consensus on diagnosis and treatment of infection after fracture fixation. Injury. 2019;50(11):1952-1958. [15] DE MEO D, CANNARI FM, PETRIELLO L, et al. Gentamicin-Coated Tibia Nail in Fractures and Nonunion to Reduce Fracture-Related Infections: A Systematic Review. Molecules. 2020;25(22):5471. [16] SHIMABUKURO M. Antibacterial Property and Biocompatibility of Silver, Copper, and Zinc in Titanium Dioxide Layers Incorporated by One-Step Micro-Arc Oxidation: A Review. Antibiotics (Basel). 2020;9(10):716. [17] KOO H, ALLAN RN, HOWLIN RP, et al. Targeting microbial biofilms: current and prospective therapeutic strategies. Nat Rev Microbiol. 2017;15(12):740-755. [18] ZHANG L, GUO J, HUANG X, et al. The dual function of Cu-doped TiO(2) coatings on titanium for application in percutaneous implants. J Mater Chem B. 2016;4(21):3788-3800. [19] ZHANG D, LIU Y, LIU Z, et al. Advances in Antibacterial Functionalized Coatings on Mg and Its Alloys for Medical Use-A Review. Coatings. 2020;10(9):828. [20] ZAJC J, MOLIČNIK A, FOKTER SK. Dual Modular Titanium Alloy Femoral Stem Failure Mechanisms and Suggested Clinical Approaches. Materials (Basel). 2021;14(11):3078. [21] KOPEC M, BRODECKI A, SZCZĘSNY G, et al. Microstructural Analysis of Fractured Orthopedic Implants. Materials (Basel). 2021;14(9):2209. [22] LIU C, ZHANG E. Biocorrosion properties of antibacterial Ti-10Cu sintered alloy in several simulated biological solutions. J Mater Sci Mater Med. 2015;26(3):142. [23] ALSHAMMARI Y, YANG F, BOLZONI L. Low-cost powder metallurgy Ti-Cu alloys as a potential antibacterial material. J Mech Behav Biomed Mater. 2019;95:232-239. [24] ZHANG E, WANG X, CHEN M, et al. Effect of the existing form of Cu element on the mechanical properties, bio-corrosion and antibacterial properties of Ti-Cu alloys for biomedical application. Mater Sci Eng C Mater Biol Appl. 2016;69:1210-1221. [25] FOWLER L, JANSON O, ENGQVIST H, et al. Antibacterial investigation of titanium-copper alloys using luminescent Staphylococcus epidermidis in a direct contact test. Mater Sci Eng C Mater Biol Appl. 2019;97:707-714. [26] ZHANG E, REN J, LI S, et al. Optimization of mechanical properties, biocorrosion properties and antibacterial properties of as-cast Ti-Cu alloys. Biomed Mater. 2016;11(6):065001. [27] LIU J, LI F, LIU C, et al. Effect of Cu content on the antibacterial activity of titanium-copper sintered alloys. Mater Sci Eng C Mater Biol Appl. 2014;35:392-400. [28] MA Z, LI M, LIU R, et al. In vitro study on an antibacterial Ti-5Cu alloy for medical application. J Mater Sci Mater Med. 2016;27(5):91. [29] LIU R, TANG Y, ZENG L, et al. In vitro and in vivo studies of anti-bacterial copper-bearing titanium alloy for dental application. Dent Mater. 2018; 34(8):1112-1126. [30] FOWLER L, JANSE VAN VUUREN A, GOOSEN W, et al. Investigation of Copper Alloying in a TNTZ-Cu(x) Alloy. Materials (Basel). 2019;12(22):3691. [31] BAI B, ZHANG E, LIU J, et al. The anti-bacterial activity of titanium-copper sintered alloy against Porphyromonas gingivalis in vitro. Dent Mater J. 2016;35(4):659-667. [32] WANG X, DONG H, LIU J, et al. In vivo antibacterial property of Ti-Cu sintered alloy implant. Mater Sci Eng C Mater Biol Appl. 2019;100:38-47. [33] LIU J, ZHANG X, WANG H, et al. The antibacterial properties and biocompatibility of a Ti-Cu sintered alloy for biomedical application. Biomed Mater. 2014;9(2):025013. [34] KIRMANIDOU Y, SIDIRA M, BAKOPOULOU A, et al. Assessment of cytotoxicity and antibacterial effects of silver nanoparticle-doped titanium alloy surfaces. Dent Mater. 2019;35(9):e220-e233. [35] GUPTA NK, SOMANI N, PRAKASH C, et al. Revealing the WEDM Process Parameters for the Machining of Pure and Heat-Treated Titanium (Ti-6Al-4V) Alloy. Materials (Basel). 2021;14(9):2292. [36] DING Z, WANG Y, ZHOU Q, et al. The Preparation and Properties of Multilayer Cu-MTa(2)O(5) Composite Coatings on Ti6Al4V for Biomedical Applications. Nanomaterials (Basel). 2019;9(10):1498. [37] GUO S, LU Y, WU S, et al. Preliminary study on the corrosion resistance, antibacterial activity and cytotoxicity of selective-laser-melted Ti6Al4V-xCu alloys. Mater Sci Eng C Mater Biol Appl. 2017;72:631-640. [38] XU X, LU Y, LI S, et al. Copper-modified Ti6Al4V alloy fabricated by selective laser melting with pro-angiogenic and anti-inflammatory properties for potential guided bone regeneration applications. Mater Sci Eng C Mater Biol Appl. 2018;90:198-210. [39] KRAKHMALEV P, YADROITSEV I, YADROITSAVA I, et al. Functionalization of Biomedical Ti6Al4V via In Situ Alloying by Cu during Laser Powder Bed Fusion Manufacturing. Materials (Basel). 2017;10(10):1154. [40] KIKUCHI M, TAKADA Y, KIYOSUE S, et al. Mechanical properties and microstructures of cast Ti-Cu alloys. Dent Mater. 2003;19(3):174-181. [41] VILARDELL AM, TAKEZAWA A, DU PLESSIS A, et al. Mechanical behavior of in-situ alloyed Ti6Al4V(ELI)-3 at.% Cu lattice structures manufactured by laser powder bed fusion and designed for implant applications. J Mech Behav Biomed Mater. 2021;113:104130. [42] WANG H, SONG W, LIU M, et al. Manufacture-friendly nanostructured metals stabilized by dual-phase honeycomb shell. Nat Commun. 2022; 13(1):2034. [43] ZHANG S, YU Y, WANG H, et al. Study on mechanical behavior of Cu-bearing antibacterial titanium alloy implant. J Mech Behav Biomed Mater. 2022;125:104926. [44] YANG J, QIN H, CHAI Y, et al. Molecular mechanisms of osteogenesis and antibacterial activity of Cu-bearing Ti alloy in a bone defect model with infection in vivo. J Orthop Translat. 2021;27:77-89. [45] CHALLA VS, MALI S, MISRA RD. Reduced toxicity and superior cellular response of preosteoblasts to Ti-6Al-7Nb alloy and comparison with Ti-6Al-4V. J Biomed Mater Res A. 2013;101(7):2083-2089. [46] COSTA B C, TOKUHARA CK, ROCHA LA, et al. Vanadium ionic species from degradation of Ti-6Al-4V metallic implants: In vitro cytotoxicity and speciation evaluation. Mater Sci Eng C Mater Biol Appl. 2019;96:730-739. [47] YAMANOGLU R, EFENDI E, KOLAYLI F, et al. Production and mechanical properties of Ti-5Al-2.5Fe-xCu alloys for biomedical applications. Biomed Mater. 2018;13(2):025013. [48] BOLZONI L, ALQATTAN M, PETERS L, et al. Ternary Ti alloys functionalised with antibacterial activity. Sci Rep. 2020;10(1):22201. [49] SANTOS P F, NIINOMI M, LIU H, et al. Fabrication of low-cost beta-type Ti-Mn alloys for biomedical applications by metal injection molding process and their mechanical properties. J Mech Behav Biomed Mater. 2016;59:497-507. [50] ALQATTAN M, PETERS L, ALSHAMMARI Y, et al. Antibacterial Ti-Mn-Cu alloys for biomedical applications. Regen Biomater. 2021;8(1):rbaa050. [51] ALSHAMMARI Y, YANG F, BOLZONI L. Mechanical properties and microstructure of Ti-Mn alloys produced via powder metallurgy for biomedical applications. J Mech Behav Biomed Mater. 2019;91:391-397. [52] LI HF, QIU K J, ZHOU F Y, et al. Design and development of novel antibacterial Ti-Ni-Cu shape memory alloys for biomedical application. Sci Rep. 2016;6:37475. [53] ZHAO QM, LI B, YU FX, et al. Cu-Co Co-Doped Microporous Coating on Titanium with Osteogenic and Antibacterial Properties. J Biomed Nanotechnol. 2021;17(7):1435-1447. [54] CHOUIRFA H, BOULOUSSA H, MIGONNEY V, et al. Review of titanium surface modification techniques and coatings for antibacterial applications. Acta Biomater. 2019;83:37-54. [55] LIU H, XU D, MA Y, et al. Mechanisms of Hierarchical Topographies Tuning Bacteria and Cell Biological Responses to the Surfaces of Pure Titanium and Cu-Bearing Titanium Alloy. ACS Appl Mater Interfaces. 2022;14(17):19226-19240. [56] DENG W, SHAO H, LI H, et al. Is surface modification effective to prevent periprosthetic joint infection? A systematic review of preclinical and clinical studies. Orthop Traumatol Surg Res. 2019;105(5):967-974. [57] KAUR M, SINGH K. Review on titanium and titanium based alloys as biomaterials for orthopaedic applications. Mater Sci Eng C Mater Biol Appl. 2019;102:844-862. [58] FU X, WANG XD, ZHAO B, et al. Atomic-scale observation of non-classical nucleation-mediated phase transformation in a titanium alloy. Nat Mater. 2022;21(3):290-296. [59] KOMAROVA EG, SHARKEEV YP, SEDELNIKOVA MB, et al. Zn- or Cu-Containing CaP-Based Coatings Formed by Micro-arc Oxidation on Titanium and Ti-40Nb Alloy: Part I-Microstructure, Composition and Properties. Materials (Basel). 2020;13(18):4116. [60] WU Y, ZHOU H, ZENG Y, et al. Recent Advances in Copper-Doped Titanium Implants. Materials (Basel), 2022;15(7):2342. [61] ULLAH I, SIDDIQUI MA, LIU H, et al. Mechanical, Biological, and Antibacterial Characteristics of Plasma-Sprayed (Sr,Zn) Substituted Hydroxyapatite Coating. ACS Biomater Sci Eng. 2020;6(3):1355-1366. [62] KOMAROVA EG, SHARKEEV YP, SEDELNIKOVA MB, et al. Zn- or Cu-containing CaP-Based Coatings Formed by Micro-Arc Oxidation on Titanium and Ti-40Nb Alloy: Part II-Wettability and Biological Performance. Materials (Basel). 2020;13(19):4366. [63] WANG L J, NI X H, ZHANG F,et al. Osteoblast Response to Copper-Doped Microporous Coatings on Titanium for Improved Bone Integration. Nanoscale Res Lett. 2021;16(1):146. [64] ZHANG X, LI J, WANG X, et al. Effects of copper nanoparticles in porous TiO(2) coatings on bacterial resistance and cytocompatibility of osteoblasts and endothelial cells. Mater Sci Eng C Mater Biol Appl. 2018;82:110-120. [65] PRINZ C, ELHENSHERI M, RYCHLY J, et al. Antimicrobial and bone-forming activity of a copper coated implant in a rabbit model. J Biomater Appl. 2017;32(2):139-149. [66] VAN HENGEL IAJ, TIEROLF M, VALERIO VPM, et al. Self-defending additively manufactured bone implants bearing silver and copper nanoparticles. J Mater Chem B. 2020;8(8):1589-1602. [67] ZHAO Q, YI L, HU A, et al. Antibacterial and osteogenic activity of a multifunctional microporous coating codoped with Mg, Cu and F on titanium. J Mater Chem B. 2019;7(14):2284-2299. [68] HU J, LI H, WANG X, et al. Effect of ultrasonic micro-arc oxidation on the antibacterial properties and cell biocompatibility of Ti-Cu alloy for biomedical application. Mater Sci Eng C Mater Biol Appl. 2020;115:110921. [69] DING Z, WANG Y, ZHOU Q, et al. Microstructure, Wettability, Corrosion Resistance and Antibacterial Property of Cu-MTa(2)O(5) Multilayer Composite Coatings with Different Cu Incorporation Contents. Biomolecules. 2019;10(1):68. [70] JIN X, GAO L, LIU E, et al. Microstructure, corrosion and tribological and antibacterial properties of Ti-Cu coated stainless steel. J Mech Behav Biomed Mater. 2015;50:23-32. [71] KUMAR DD, KALIARAJ GS. Multifunctional zirconium nitride/copper multilayer coatings on medical grade 316L SS and titanium substrates for biomedical applications. J Mech Behav Biomed Mater. 2018;77:106-115. [72] ALHUSSEIN A, ACHACHE S, DETURCHE R, et al. Beneficial effect of Cu on Ti-Nb-Ta-Zr sputtered uniform/adhesive gum films accelerating bacterial inactivation under indoor visible light. Colloids Surf B Biointerfaces. 2017;152:152-158. [73] YU L, JIN G, OUYANG L, et al. Antibacterial activity, osteogenic and angiogenic behaviors of copper-bearing titanium synthesized by PIII&D. J Mater Chem B. 2016;4(7):1296-1309. [74] XIA C, MA X, ZHANG X, et al. Enhanced physicochemical and biological properties of C/Cu dual ions implanted medical titanium. Bioact Mater. 2020;5(2):377-386. [75] LI Q, LI L, ZHAO M, et al. Biological actions of Cu/Zn coimplanted TiN on Ti-6Al-4V alloy. Biointerphases. 2019;14(5):051008. [76] XIA C, CAI D, TAN J, et al. Synergistic Effects of N/Cu Dual Ions Implantation on Stimulating Antibacterial Ability and Angiogenic Activity of Titanium. ACS Biomater Sci Eng. 2018;4(9):3185-3193. |
| [1] | 王彦金, 周英杰, 柴旭斌, 禚汉杰. 3D打印多孔钛合金椎间融合器在颈椎前路椎间盘切除植骨融合中应用效果与安全性的Meta分析[J]. 中国组织工程研究, 2023, 27(9): 1434-1440. |
| [2] | 王书祥, 伍 权, 周小淞, 况现桃. 钛合金全冠选择性激光熔化成形工艺及变形实验[J]. 中国组织工程研究, 2023, 27(7): 1012-1016. |
| [3] | 张婷婷, 刘 娟, 张 旭. 相转移溶菌酶用于氧化锆全瓷种植材料表面改性后介导羟基磷灰石涂层的生物活性[J]. 中国组织工程研究, 2023, 27(7): 1043-1049. |
| [4] | 徐星星, 文超举, 孟茂花, 王勤英, 陈镜桥, 董 强. 口腔种植中的碳纳米材料[J]. 中国组织工程研究, 2023, 27(7): 1062-1070. |
| [5] | 史业弘, 王 成, 陈世玖. 小口径人工血管的早期血栓形成与预防[J]. 中国组织工程研究, 2023, 27(7): 1110-1116. |
| [6] | 李 睿, 刘 珍, 郭子歌, 卢瑞杰, 王 晨. 纯钛表面载阿司匹林微球与聚多巴胺复合涂层促进成骨分化[J]. 中国组织工程研究, 2023, 27(3): 374-379. |
| [7] | 牛 林, 梅钰坤, 邹 蕊, 张雨薇, 张翼飞, 郝雅琪, 董少杰. 无机非金属生物材料在骨肉瘤治疗与伴发骨缺损再生中的应用[J]. 中国组织工程研究, 2023, 27(21): 3368-3374. |
| [8] | 蒋成明, 蒋赛男, 唐 烨, 蒋 林. 3D打印微孔钛合金融合器用于颈椎前路减压植骨融合对颈椎解剖学及应激激素的影响[J]. 中国组织工程研究, 2023, 27(18): 2837-2841. |
| [9] | 王杰杰, 殷俊飞扬, 钟 静, 宫海环, 王艺霖, 赵艳艳, 李严兵, 黄文华. 3D打印高密度聚乙烯支架表面涂层的性能[J]. 中国组织工程研究, 2023, 27(16): 2501-2509. |
| [10] | 黄燕妮, 杨 华, 杨东梅, 胡旭麟, 高 鸿, 黄毅娜. 聚三亚甲基碳酸酯/β-磷酸三钙微球支架修复大鼠骨缺损[J]. 中国组织工程研究, 2023, 27(12): 1856-1862. |
| [11] | 胡金龙, 全桦红, 王静成, 张 佩, 张家乐, 陈鹏涛, 梁 远. 温敏性水凝胶Pluronic F127载硫化铜纳米颗粒修复大鼠感染性创面[J]. 中国组织工程研究, 2023, 27(12): 1927-1931. |
| [12] | 仵博宇, 叶 开, 陈家瀚, 王江华, 乌日开西·艾依提, 蒋厚峰, 滕 勇. 3D打印聚醚醚酮/羟基磷灰石复合物的生物相容性评价[J]. 中国组织工程研究, 2023, 27(12): 1932-1937. |
| [13] | 任文燕, 刘 雪, 王一宇. 石墨烯及衍生物治疗牙周病:有利于成骨分化及牙周支持组织的重建[J]. 中国组织工程研究, 2023, 27(12): 1954-1960. |
| [14] | 王月升, 高 炜, 彭 诚. 口腔种植体材料表面处理及其抗菌、诱导成骨的能力[J]. 中国组织工程研究, 2023, 27(12): 1961-1968. |
| [15] | 王伟伟, 欧志学, 周 毅, 李 统. 铜死亡基因在骨关节炎免疫浸润中的生物信息学分析[J]. 中国组织工程研究, 2023, 27(11): 1669-1676. |
然而,骨折术后发生感染一直是困扰全球骨科医生的难题[5]。由于社会经济因素的原因,年轻人的高能量损伤骨折、老年人的低能量损伤骨折的病例越来越多[6-7]。据统计,世界上每年有超过1 400万例院内感染,60%与医疗器械有关,有资料显示美国每年骨科植入物发生的感染约占整体骨科手术的4.3%[8]。而德国的数据则表明2018年骨折相关感染为每10万居民发生10.7例,患病率从2008年的1.05%升高到2018年的1.23%,并且发现感染发生率随年龄的增加而递增,60岁以上的老人尤其明显[9]。近期对急慢性骨折相关感染综合回顾发现,经过系统治疗的病例感染复发率为6%-9%,而最终3%-5%的病例不可避免地进行了截肢手术[10]。虽然手术操作的严格无菌要求、术后感染指标的监控及预防性抗生素的使用有效避免了植入物相关感染情况的发生,但是基于每年庞大的骨科手术患者,感染病例的发生似乎仍然有增无减。尤其是老龄化社会的加剧使得发生骨质疏松骨折并患有糖尿病、冠心病等基础病的老年人数量与日俱增[11]。而现有的治疗内植物相关感染时常在植入物表面的细菌生物膜形成之后进行,因此存在一定滞后性。而且,通常对发生感染的病例会首先采用足量广谱抗生素进行全身抗感染治疗,但当感染局部未达到有效抗菌浓度时治疗效果并不明显且可能形成耐药菌株,如果感染情况进一步发展,植入物的取出将不可避免,这也标志着手术的失败[12-13]。一旦发生植入物相关感染,则会带给患者及家属巨大的身心压力和经济负担,据统计发生感染的病例约是非感染病例治疗费用的4.8倍,高达73 500人民币左右[14-15]。因此研究具有良好抗菌性能的钛合金材料作为植入物尽可能避免此类事件的发生是有必要的,而其中含铜(Copper,Cu)钛合金一直是研究的热点方向。该文主要阐述加入金属铜的钛合金材料抗菌性能的最新研究进展,并对其未来进行展望。
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 第一作者在2022年3月进行检索。
1.1.2 检索文献时限 检索2001年1月至2022年3月发表的相关文献。
1.1.3 检索数据库 PubMed及Web of Science数据库。
1.1.4 检索词 以“Titanium(Ti),Copper(Cu),Antibacterial,Surface modification,Coating”为英文检索词。
1.1.5 检索文献类型 研究性原著、综述及病例报告。
1.1.6 手工检索情况 通过阅读题目及摘要初步筛选,排除与研究无关的文献。
1.1.7 文献检索策略 以PubMed数据库检索为例,见图1。

1.1.8 文献检索量 初检得到相关文献412篇。
1.2 入选标准
纳入标准:①与医用钛合金加入铜的抗菌性能研究密切相关的文章;②与测试加铜后的钛合金细胞毒性及生物力学性能有关的文章;③同一领域的相关性研究选择近年发表的文章。
排除标准:重复性研究;相关性差及无关研究。
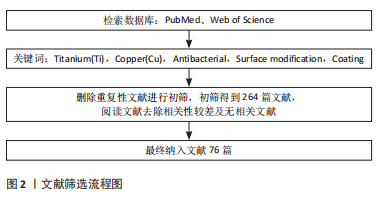
1.3 资料提取 根据检索词检索到相关文献后,删除重复性文献进行初筛,初筛得到264篇文献,阅读文献题目及摘要,按照纳入排除标准筛选后最终共纳入76篇文献进行分析和整理。文献检索流程图,见图2。
选择铜加入钛合金使其具备抗菌能力还需要解决以下问题:①内植物抗菌能力随铜元素含量的提升而提高,然而会对材料生物力学性能及生物相容性产生一定影响,尤其在伤口经外科处理后多保持无菌或少菌状态,因此不能一味的追求抗菌效率,而选择合适的铜质量占比及相应的制备加工工艺达到综合性能平衡是才是研究的重点。②钛合金的表面改性虽然会避免铜对合金的直接影响,但多数制备工艺会造成合金表面的微损伤,这种微损伤是否会对内植物植入人体造成不利影响有待进一步验证。③合金的抗菌性能依赖于铜离子释放产生抗菌效果,而如何控制铜离子的释放速率使其保持较长时间的抗菌能力以及避免铜离子早期大量突然释放的“突释现象”造成的不良反应也需要解决。目前,大量实验表明含铜的钛合金基本对抗菌功能的需求,而且在复杂的内外界环境影响下其稳定性是值得信赖的,相信在不久的未来,通过对所需铜含量进一步分析、制备工艺的完善、综合性能的优化,含铜钛合金在抗菌生物材料领域十分值得期待。
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
 #br#
#br#
文题释义:
医用钛合金:用于制造植入人体内的医疗器件、假体、辅助治疗设备的钛合金,生物力学性能接近人骨,还具备耐疲劳、耐腐蚀及良好的生物相容性等优势。细菌生物膜:细菌黏附于表面分泌多糖基质、纤维蛋白、脂质蛋白等,将其自身包绕其中而形成的大量细菌聚集膜样物,是为适应自然环境有利生存的一种生命现象。
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
钛及其合金材料因具有良好的生物相容性及优异的力学性能被广泛应用于包括骨科在内的多个医学领域。钛所具备的生物惰性使其在少量氧气存在情况下产生氧化膜,可以使其在体内出现疲劳、抗压甚至出现裂缝的情况下仍然不会与复杂的人体环境发生反应;而且钛不易被磁化,不会妨碍做核磁共振等影像学检查。所以以纯钛、Ti6Al4V合金为代表的钛基合金作为内固定板、螺钉等器械在目前骨科手术中具有不可替代的作用。
作为内植物研究热点之一,广泛的应用所带来的相关感染问题成为不可忽视的一部分,而且治疗感染有目共睹的代价及难度更加凸显了合金本身具备抗菌性能的重要性。而铜作为人体必须的微量元素、钛β相的稳定元素以及抗菌机制清楚的金属元素十分贴合这一需求。采取不同的制备方法将优异且机制清楚的抗菌金属铜加入其中,在不影响生物力学性能的同时可使其具备一定的抗菌能力。该文主要阐述加入金属铜的钛合金材料抗菌性能的最新研究进展,并对其未来进行展望。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||