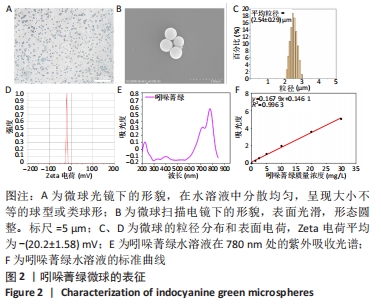
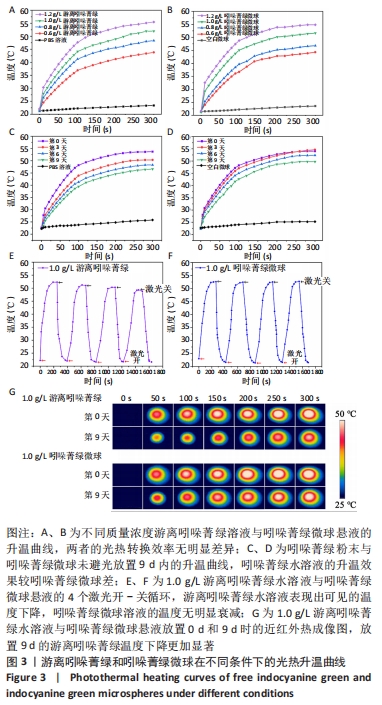
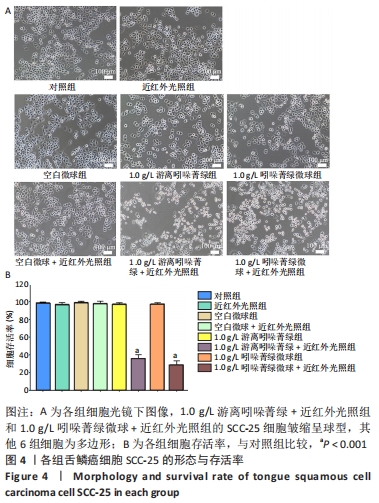
[1] YANAMOTO S, DENDA Y, OTA Y, et al. Postoperative adjuvant therapy for patients with loco-regionally advanced oral squamous cell carcinoma who are at high risk of recurrence. Int J Oral Maxillofac Surg. 2020;49(7):848-853.
[2] KORDZINSKA-CISEK I, GRZYBOWSKA-SZATKOWSKA L. Complications of radio- and radiochemotherapy in patients undergoing major salivary gland cancer surgery. Otolaryngol Pol. 2019;73(3):26-31.
[3] LIU Y, BHATTARAI P, DAI Z, et al. Photothermal therapy and photoacoustic imaging via nanotheranostics in fighting cancer. Chem Soc Rev. 2019;48(7):2053-2108.
[4] LONG S, XU Y, ZHOU F, et al. Characteristics of temperature changes in photothermal therapy induced by combined application of indocyanine green and laser. Oncol Lett. 2019;17(4):3952-3959.
[5] CHEN J, LI Q, WANG F, et al. Biosafety, Nontoxic Nanoparticles for VL-NIR Photothermal Therapy Against Oral Squamous Cell Carcinoma. ACS Omega. 2021;6(17):11240-11247.
[6] LAU CT, AU DM, WONG K. Application of indocyanine green in pediatric surgery. Pediatr Surg Int. 2019;35(10):1035-1041.
[7] HIROHASHI K, ANAYAMA T, WADA H, et al. Lung cancer photothermal ablation by low-power near-infrared laser and topical injection of indocyanine green. Interact Cardiovasc Thorac Surg. 2019;29(5):693-698.
[8] LIU C, RUAN C, SHI R, et al. A near infrared-modulated thermosensitive hydrogel for stabilization of indocyanine green and combinatorial anticancer phototherapy. Biomater Sci. 2019;7(4):1705-1715.
[9] HWANG J, JIN J. Attachable Hydrogel Containing Indocyanine Green for Selective Photothermal Therapy against Melanoma. Biomolecules (Basel, Switzerland). 2020;10(8):1124.
[10] SAXENA V, SADOQI M, SHAO J. Degradation kinetics of indocyanine green in aqueous solution. J Pharm Sci. 2003;92(10):2090-2097.
[11] OTT P. Hepatic elimination of indocyanine green with special reference to distribution kinetics and the influence of plasma protein binding. Pharmacol Toxicol. 1998;83 Suppl 2:1-48.
[12] WU L, FANG S, SHI S, et al. Hybrid polypeptide micelles loading indocyanine green for tumor imaging and photothermal effect study. Biomacromolecules. 2013;14(9):3027-3033.
[13] MIRANDA D, WAN C, KILIAN HI, et al. Indocyanine green binds to DOTAP liposomes for enhanced optical properties and tumor photoablation. Biomater Sci. 2019;7(8):3158-3164.
[14] WANG Y, NIU C, FAN S, et al. Indocyanine Green Loaded Modified Mesoporous Silica Nanoparticles as an Effective Photothermal Nanoplatform. Int J Mol Sci. 2020;21(13):4789.
[15] MAKADIA HK, SIEGEL SJ. Poly Lactic-co-Glycolic Acid (PLGA) as Biodegradable Controlled Drug Delivery Carrier. Polymers (Basel). 2011;3(3):1377-1397.
[16] TAN H, HUANG D, LAO L, et al. RGD modified PLGA/gelatin microspheres as microcarriers for chondrocyte delivery. J Biomed Mater Res B Appl Biomater. 2009;91(1):228-238.
[17] RAFIEI P, HADDADI A. Docetaxel-loaded PLGA and PLGA-PEG nanoparticles for intravenous application: pharmacokinetics and biodistribution profile. Int J Nanomedicine. 2017;12:935-947.
[18] CHOI S, LEE SH, PARK S, et al. Indocyanine Green-Loaded PLGA Nanoparticles Conjugated with Hyaluronic Acid Improve Target Specificity in Cervical Cancer Tumors. Yonsei Med J. 2021;62(11):1042-1051.
[19] CHEN HH, LU IL, LIU TI, et al. Indocyanine green/doxorubicin-encapsulated functionalized nanoparticles for effective combination therapy against human MDR breast cancer. Colloids Surf B Biointerfaces. 2019;177:294-305.
[20] SAXENA V, SADOQI M, SHAO J. Indocyanine green-loaded biodegradable nanoparticles: preparation, physicochemical characterization and in vitro release. Int J Pharm. 2004;278(2):293-301.
[21] LI Y, PEI Y, ZHANG X, et al. PEGylated PLGA nanoparticles as protein carriers: synthesis, preparation and biodistribution in rats. J Control Release. 2001;71(2):203-211.
[22] LAN M, ZHU L, WANG Y, et al. Multifunctional nanobubbles carrying indocyanine green and paclitaxel for molecular imaging and the treatment of prostate cancer. J Nanobiotechnology. 2020;18(1):121.
[23] HOUTHOOFD S, VUYLSTEKE M, MORDON S, et al. Photodynamic therapy for atherosclerosis. The potential of indocyanine green. Photodiagnosis Photodyn Ther. 2020;29:101568.
[24] YOON HJ, LEE HS, LIM JY, et al. Liposomal Indocyanine Green for Enhanced Photothermal Therapy. ACS Appl Mater Interfaces. 2017; 9(7):5683-5691.
[25] SAXENA V, SADOQI M, SHAO J. Enhanced photo-stability, thermal-stability and aqueous-stability of indocyanine green in polymeric nanoparticulate systems. J Photochem Photobiol B. 2004;74(1):29-38.
[26] Reinhart MB, Huntington CR, Blair LJ, et al. Indocyanine Green: Historical Context, Current Applications, and Future Considerations. Surg Innov. 2016;23(2):166-175.
[27] 余柳丹,罗一帆,陈小菊,等.吲哚菁绿稳定性及光敏性和声敏性研究[J].化学研究与应用,2016,28(3):400-403.
[28] LIM HJ, CHIOW A, LEE LS, et al. Novel method of intraoperative liver tumour localisation with indocyanine green and near-infrared imaging. Singapore Med J. 2021;62(4):182-189.
[29] WANG Q, SHEN M, LI W, et al. Controlled-release of fluazinam from biodegradable PLGA-based microspheres. J Environ Sci Health B. 2019; 54(10):810-816.
[30] LIU Z, YE W, ZHENG J, et al. Hierarchically electrospraying a PLGA@chitosan sphere-in-sphere composite microsphere for multi-drug-controlled release. Regen Biomater. 2020;7(4):381-390.
[31] GAIGNAUX A, REEFF J, DE VRIESE C, et al. Evaluation of the degradation of clonidine-loaded PLGA microspheres. J Microencapsul. 2013;30(7): 681-691.
[32] HAN S, ZHANG X, LI M. Progress in research and application of PLGA embolic microspheres. Front Biosci (Landmark Ed). 2016;21:931-940.
[33] ABULATEEFEH SR, ALKAWAREEK MY, ABDULLAH FR, et al. Preparation of Aqueous Core-Poly(d,l-Lactide-co-Glycolide) Shell Microcapsules With Mononuclear Cores by Internal Phase Separation: Optimization of Formulation Parameters. J Pharm Sci. 2017;106(4):1136-1142.
[34] PALAZZO I, LAMPARELLI EP, CIARDULLI MC, et al. Supercritical emulsion extraction fabricated PLA/PLGA micro/nano carriers for growth factor delivery: Release profiles and cytotoxicity. Int J Pharm. 2021;592:120108.
[35] SHI NQ, ZHOU J, WALKER J, et al. Microencapsulation of luteinizing hormone-releasing hormone agonist in poly (lactic-co-glycolic acid) microspheres by spray-drying. J Control Release. 2020;321:756-772.
[36] Ohta S, Matsuura M, Kawashima Y, et al. Facile fabrication of PEG-coated PLGA microspheres via SPG membrane emulsification for the treatment of scleroderma by ECM degrading enzymes. Colloids Surf B Biointerfaces. 2019;179:453-461.
[37] WU B, WU L, HE Y, et al. Engineered PLGA microspheres for extended release of brexpiprazole: in vitro and in vivo studies. Drug Dev Ind Pharm. 2021;47(6):1001-1010.
[38] GASPAR MC, PAIS A, SOUSA J, et al. Development of levofloxacin-loaded PLGA microspheres of suitable properties for sustained pulmonary release. Int J Pharm. 2019;556:117-124.
[39] BUTREDDY A, GADDAM RP, KOMMINENI N, et al. PLGA/PLA-Based Long-Acting Injectable Depot Microspheres in Clinical Use: Production and Characterization Overview for Protein/Peptide Delivery. Int J Mol Sci. 2021;22(16):8884.
[40] ZHAI P, CHEN XB, SCHREYER DJ. PLGA/alginate composite microspheres for hydrophilic protein delivery. Mater Sci Eng C Mater Biol Appl. 2015; 56:251-259.
[41] PARK JS, YANG HN, JEON SY, et al. The use of anti-COX2 siRNA coated onto PLGA nanoparticles loading dexamethasone in the treatment of rheumatoid arthritis. Biomaterials. 2012;33(33):8600-8612.
[42] LIU R, MA GH, WAN YH, et al. Influence of process parameters on the size distribution of PLA microcapsules prepared by combining membrane emulsification technique and double emulsion-solvent evaporation method. Colloids Surf B Biointerfaces. 2005;45(3-4):144-153.
[43] KONAN YN, CERNY R, FAVET J, et al. Preparation and characterization of sterile sub-200 nm meso-tetra(4-hydroxylphenyl)porphyrin-loaded nanoparticles for photodynamic therapy. Eur J Pharm Biopharm. 2003; 55(1):115-124.
[44] JAQUE D, MARTINEZ ML, DEL RB, et al. Nanoparticles for photothermal therapies. Nanoscale. 2014;6(16):9494-9530.
|