Odontoblasts are the main cells for synthesis and secretion of dentin matrix. Because odontoblasts are difficult to obtain and easy to age and die after passage
in vitro, current studies on the function of odontoblasts mainly focus on the application of differentiation characteristics of dental pulp cells and dental papilla cells
[11-14]. SHED which is isolated by Miura
et al using enzymatic digestion, has become a rookie in tissue engineering research because of their strong proliferation ability and multi-directional differentiation potential.
During tooth development , formation and repair after pulp damage, a variety of growth factors have been discovered. These growth factors provide a microenvironment for cell survival, proliferation and differentiation. The complex interactions between growth factors regulate various cell activities, and play an important role in the growth and differentiation of organs and tissues. TGF-β superfamily is a superfamily of regulating cell growth and differentiation, which is not only directly involved in the signal transduction between the mesenchyma and epithelium, and also control the formation of body’s odontoblasts and osteoblasts in the embryonic stage, playing an important role in odontoblast growth, migration and differentiation[15-17]. In the TGF-β superfamily, TGF-β3 is mainly secreted by mesenchymal cells, and exerts a critical role in the differentiation and growth of mesenchymal cell lines. During tooth development, the dentin, dental pulp, and periodontal tissue are all derived from mesenchymal tissue. Therefore, this study investigated the odontoblast-like differentiation of SHED, so as to further explore the action mechanism and clinical value of SHED in the induction of dentin formation.
Studies have shown that TGF-β, human basic fibroblast growth factor, insulin-like growth factor, bone morphogenetic proteins and fibronectin can induce the in vitro differentiation of dental papilla cells or dental pulp cells in vitro into odontoblasts under tissue culture conditions[18-19]. Huojia et al [20] found that recombinant TGF-β3 can induce ectopic calcification of dental pulp cells from mouse dental germ, improve the levels of osteocalcin and collagen type I mRNA and protein in dental pulp cells, and induce the expressions of dentin sialoprotein, dentin matrix protein, osteocalcin, osteonectin, osteopontin in primary dental pulp cells, indicating that TGF-β3 can induce ectopic mineralization of dental pulp cells by upregulating osteocalcin and collagen type I, and thereby promoting the differentiation of dental pulp cells into odontoblasts.
However, a growth factor alone appears to have some shortcomings in cell induction, such as low efficiency and instable differentiation. Heparin is shown to have a special affinity with many biologically important proteins. Consequently, heparin is associated with cell adhesion, recognition, migration, and regulating the activity of various enzymes. There are a lot of affinity growth factors for heparin, including fibroblast growth factor, vascular endothelial growth factor, hepatocyte growth factor and TGF. These factors bind tightly to heparin PHS molecule in a specific extracellular matrix. Studies have shown that heparin can provide a plurality of cell binding sites for TGF-β superfamily in intracellular matrix[21], prevent TGF-β from proteolytic hydrolysis and chemical inactivation, effectively protect the activity of TGF-β, and stabilize the molecular conformation[22]. Begue-Kirm and co-workers reported in the experiments of culturing dental papilla tissue that TGF-β1 combined with heparin could promote the polarization of dental papilla cells, and these cells transcribed dentin sialophosphoprotein and secreted predentin, showing a differentiation state of odontoblasts[23].
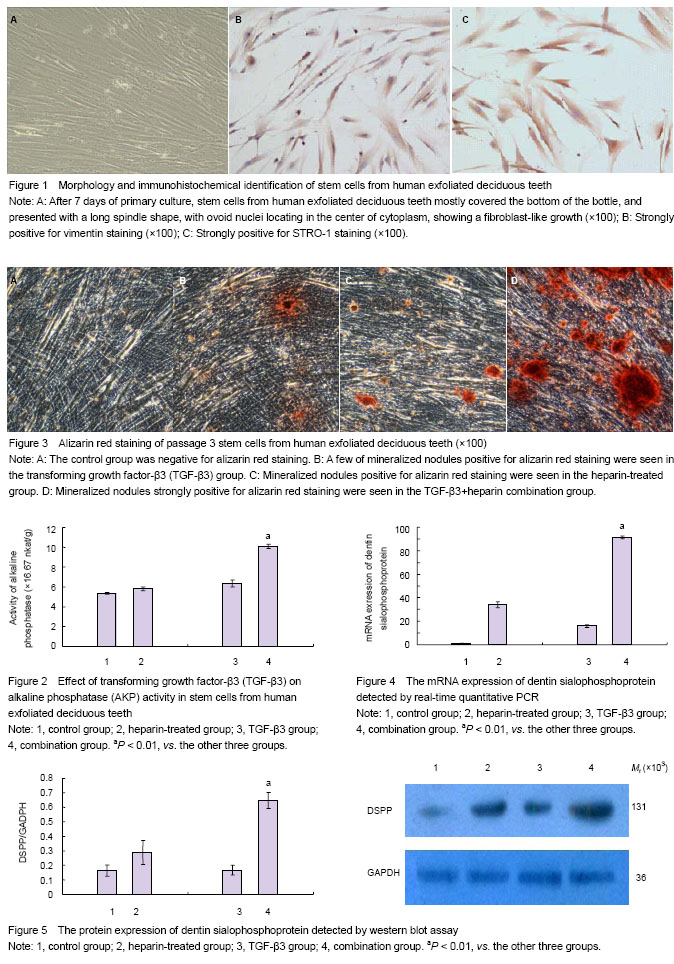
Therefore, in this study, we isolated and purified SHED according to the Gronthos’s method, and identified SHED using STRO-1 and vimentin staining. There were four groups: TGF-β3+heparin combination group, TGF-β3 alone group, heparin-treated group and control group. Passage 3 cells were collected and cultured with 25 μg/L recombinant TGF-β3, 10 U/mL heparin or their combination. Alizarin red staining, alkaline phosphatase detection, real-time quantitative PCR and western blot assay were used for observing the differentiation of SHED into odontoblasts. We found that SHED induced by TGF-β3 alone was only weakly positive for alizarin red staining, and the mRNA expression of dentin sialophosphoprotein in TGF-β3-induced SHED was slightly higher than that in the control group, indicating TGF-β3 alone cannot obviously accelerate the odontoblast-like differentiation of SHED. However, the combined use of TGF-β3 and heparin could significantly increase the AKP activity and expressions of dentin sialophosphoprotein at mRNA and protein levels as compared with the control group, moreover, cells in the combination group were strongly positive for alizarin red staining. Therefore, we believe that the combined use of TGF-β3 and heparin can remarkably facilitate the differentiation of SHED into odontoblasts, probably because heparin can protect TGF-β3 to form a stable complex, thereby preventing the enzyme hydrolysis or chemical inactivation of TGF-β3, enhancing the active role of TGF-β3 in the odontoblast-like differentiation of SHED.
Taken together, TGF-β3 is an important cytokine, which specifically binds to TGF-βR III (CD105), a cell surface receptor, and produces a major biological function. TGF-β3 can be stably combined with heparin to promote the growth, proliferation and odontoblast-like differentiation of dental pulp cells, thereby providing an important role in dental pulp repair.
.jpg)