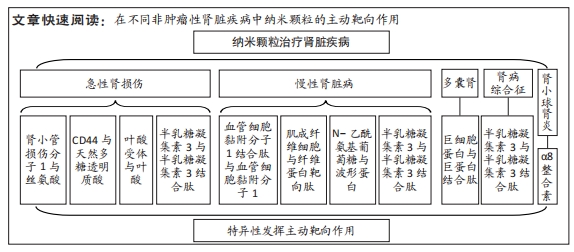
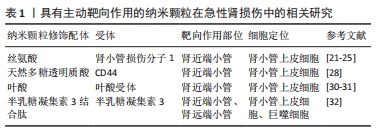
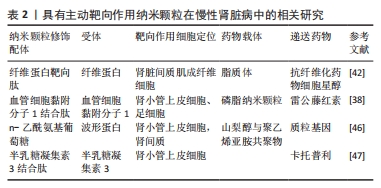
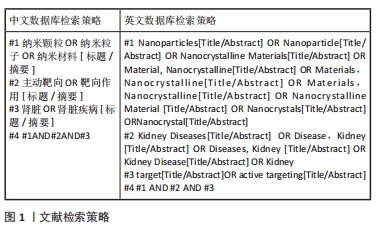
[1] BONNER R, ALBAJRAMI O, HUDSPETH J, et al. Diabetic kidney disease. Prim Care. 2020;47(4):645-659.
[2] KOVESDY CP. Epidemiology of chronic kidney disease: an update 2022. Kidney Int Suppl. 2022;12(1):7-11.
[3] WANNER N, BECHTEL-WALZ W. Epigenetics of kidney disease. Cell Tissue Res. 2017;369(1):75-92.
[4] KIM BY, RUTKA JT, CHAN WC. Nanomedicine. N Engl J Med. 2010;363(25): 2434-2443.
[5] PATRA JK, DAS G, FRACETO LF, et al. Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnology. 2018; 16(1):1-33.
[6] WANG J, MASEHI-LANO JJ, CHUNG EJ. Peptide and antibody ligands for renal targeting: nanomedicine strategies for kidney disease. Biomater Sci. 2017;5(8):1450-1459.
[7] 肖贵英,李昕,邓阳,等.抗菌药物的肾毒性机制及危险因素研究进展[J].中国抗生素杂志,2022,47(8):779-785.
[8] 伍敏,刘必成,程守勤.合理使用解热镇痛药警惕不良反应发生[N].健康报,2023-02-06(008). doi:10.28415/n.cnki.njika.2023.000320.
[9] ZHAO J, WANG X, YANG Y, et al. Upregulated miR-101 inhibits acute kidney injury-chronic kidney disease transition by regulating epithelial-mesenchymal transition. Hum Exp Toxicol. 2020;39(12):1628-1638.
[10] BRUNO BJ, MILLER GD, LIM CS. Basics and recent advances in peptide and protein drug delivery. Ther Deliv. 2013;4(11):1443-1467.
[11] ARSLAN FB, OZTURK K, CALIS S. Antibody-mediated drug delivery. Int J Pharmaceut. 2021;596:120268.
[12] GISBERT-GARZARÁN M, VALLET-REGÍ M. Nanoparticles for Bio-Medical Applications. Nanomaterials (Basel). 2022;12(7):1189.
[13] MARY SM, RAMAKRISHNAN M, SUDALAIMANI PAULPANDIAN SD, et al. Application of nanoparticles in Dentistry. Bioinformation. 2023;19(1):14-18.
[14] PEARCE AK, O’REILLY RK. Insights into active targeting of nanoparticles in drug delivery: advances in clinical studies and design considerations for cancer nanomedicine. Bioconjug Chem. 2019;30(9):2300-2311.
[15] 李珍,刘宏宝.纳米粒子肾靶向的影响因素及机制[J].中国组织工程研究,2023,27(3):453-460.
[16] HUANG Y, WANG J, JIANG K, et al. Improving kidney targeting: The influence of nanoparticle physicochemical properties on kidney interactions. J Control Release. 2021;334:127-137.
[17] HE L, WEI Q, LIU J, et al. AKI on CKD: heightened injury, suppressed repair, and the underlying mechanisms. Kidney Int. 2017;92(5):1071-1083.
[18] YAN MT, CHAO CT, LIN SH. Chronic kidney disease: Strategies to retard progression. Int J Mol Sci. 2021;22(18):10084.
[19] CHAWLA LS, EGGERS PW, STAR RA, et al. Acute kidney injury and chronic kidney disease as interconnected syndromes. N Engl J Med. 2014;371(1): 58-66.
[20] ABUELO JG. Normotensive ischemic acute renal failure. New Engl J Med. 2007;357(8):797-805.
[21] POSTON JT, KOYNER JL. Sepsis associated acute kidney injury. BMJ. 2019; 364:k4891.
[22] BUGGER H, PFEIL K. Mitochondrial ROS in myocardial ischemia reperfusion and remodeling. Biochim Biophys Acta Mol Basis Dis. 2020;1866(7):165768.
[23] ICHIMURA T, ASSELDONK EJ, HUMPHREYS BD, et al. Kidney injury molecule-1 is a phosphatidylserine receptor that confers a phagocytic phenotype on epithelial cells. J Clin Invest. 2008;118(5):1657-1668.
[24] MATSUURA S, KATSUMI H, SUZUKI H, et al. l-Serine-modified polyamidoamine dendrimer as a highly potent renal targeting drug carrier. Proc Natl Acad Sci U S A. 2018;115(41):10511-10516.
[25] LIU D, SHU G, JIN F, et al. ROS-responsive chitosan-SS31 prodrug for AKI therapy via rapid distribution in the kidney and long-term retention in the renal tubule. Sci Adv. 2020;6(41):eabb7422.
[26] LI H, TAN X, LIU Y, et al. Ruxolitinib-loaded black phosphorus nanosheets actively target proximal tubule cells to ameliorate nephrotic syndrome by reducing inflammation and oxidative stress. Nano Today. 2022;47:101631.
[27] KONG L, FAN D, ZHOU L, et al. The influence of modified molecular (D/L-serine) chirality on the theragnostics of PAMAM-based nanomedicine for acute kidney injury. J Mater Chem B. 2021;9(43):9023-9030.
[28] HERRERA M, BUSSOLATI B, BRUNO S, et al. Exogenous mesenchymal stem cells localize to the kidney by means of CD44 following acute tubular injury. Kidney Int. 2007;72(4):430-441.
[29] RAMPANELLI E, DESSING MC, CLAESSEN N, et al. CD44-deficiency attenuates the immunologic responses to LPS and delays the onset of endotoxic shock-induced renal inflammation and dysfunction. PLoS One. 2013;8(12): e84479.
[30] WANG Y, PU M, YAN J, et al. 1,2-Bis(2-aminophenoxy)ethane-N,N,N’,N’-tetraacetic Acid Acetoxymethyl Ester Loaded Reactive Oxygen Species Responsive Hyaluronic Acid-Bilirubin Nanoparticles for Acute Kidney Injury Therapy via Alleviating Calcium Overload Mediated Endoplasmic Reticulum Stress. ACS Nano. 2023;17(1):472-491.
[31] DOLMAN M, HARMSEN S, STORM G, et al. Drug targeting to the kidney: Advances in the active targeting of therapeutics to proximal tubular cells. Adv Drug Deliv Rev. 2010;62(14):1344-1357.
[32] KNIGHT SF, KUNDU K, JOSEPH G, et al. Folate receptor-targeted antioxidant therapy ameliorates renal ischemia-reperfusion injury. J Am Soc Nephrol. 2012;23(5):793-800.
[33] XIE D, WANG J, HU G, et al. Kidney-Targeted Delivery of Prolyl Hydroxylase Domain Protein 2 Small Interfering RNA with Nanoparticles Alleviated Renal Ischemia/Reperfusion Injury. J Pharmacol Exp Ther. 2021;378(3):235-243.
[34] LI HY, YANG S, LI JC, et al. Galectin 3 inhibition attenuates renal injury progression in cisplatin-induced nephrotoxicity. Biosci Rep. 2018;38(6): BSR20181803.
[35] STEVENS PE, LEVIN A, KIDNEY DISEASE: IMPROVING GLOBAL OUTCOMES CHRONIC KIDNEY DISEASE GUIDELINE DEVELOPMENT WORK GROUP MEMBERS. Evaluation and management of chronic kidney disease: synopsis of the kidney disease: improving global outcomes 2012 clinical practice guideline. Ann Intern Med. 2013;158(11):825-830.
[36] AMMIRATI AL. Chronic kidney disease. Rev Assoc Med Bras. 2020;66(Suppl 1): s03-s09.
[37] CHARLES C, FERRIS AH. Chronic kidney disease. Prim Care. 2020;47(4): 585-595.
[38] GLASSOCK RJ, WARNOCK DG, DELANAYE P. The global burden of chronic kidney disease: estimates, variability and pitfalls. Nat Rev Nephrol. 2017; 13(2):104-114.
[39] LV JC, ZHANG LX. Prevalence and disease burden of chronic kidney disease. Adv Exp Med Biol. 2019;1165:3-15.
[40] WU Q, WANG J, WANG Y, et al. Targeted delivery of celastrol to glomerular endothelium and podocytes for chronic kidney disease treatment. Nano Res. 2022;15(4):3556-3568.
[41] LIU F, GOU R, HUANG J, et al. Effect of anaphylatoxin C3a, C5a on the tubular epithelial-myofibroblast transdifferentiation in vitro. Chin Med J. 2011;124(23):4039-4045.
[42] WEISKIRCHEN R, WEISKIRCHEN S, TACKE F. Organ and tissue fibrosis: Molecular signals, cellular mechanisms and translational implications. Mol Aspects Med. 2019;65:2-15.
[43] NASTASE MV, ZENG-BROUWERS J, WYGRECKA M, et al. Targeting renal fibrosis: mechanisms and drug delivery systems. Adv Drug Deliv Rev. 2018; 129:295-307.
[44] LI R, LI Y, ZHANG J, et al. Targeted delivery of celastrol to renal interstitial myofibroblasts using fibronectin-binding liposomes attenuates renal fibrosis and reduces systemic toxicity. J Control Release. 2020;320:32-44.
[45] SHARP CN, DOLL MA, MEGYESI J, et al. Subclinical kidney injury induced by repeated cisplatin administration results in progressive chronic kidney disease. Am J Physiol Renal. 2018;315(1):F161-F172.
[46] VANSTHERTEM D, GOSSIAUX A, DECLEVES AE, et al. Expression of nestin, vimentin, and NCAM by renal interstitial cells after ischemic tubular injury. Biomed Res Int. 2010;2010:193259.
[47] ASO SI, ISE H, TAKAHASHI M, et al. Effective uptake of N-acetylglucosamine-conjugated liposomes by cardiomyocytes in vitro.J Control Release. 2007; 122(2):189-198.
[48] MATHEW AP, UTHAMAN S, BAE EH, et al. Vimentin Targeted Nano-gene Carrier for Treatment of Renal Diseases. J Korean Med Sci. 2021;36(49): e333.
[49] GENG Q, SUN X, GONG T, et al. Peptide-drug conjugate linked via a disulfide bond for kidney targeted drug delivery. Bioconjug Chem. 2012;23(6): 1200-1210.
[50] CHENG HT, HUANG HC, LEE TY, et al. Delivery of sorafenib by myofibroblast-targeted nanoparticles for the treatment of renal fibrosis. J Control Release. 2022;346:169-179.
[51] CORNEC-LE GALL E, ALAM A, PERRONE RD. Autosomal dominant polycystic kidney disease. Lancet. 2019;393(10174):919-935.
[52] ZOU Z, TAO T, LI H, et al. mTOR signaling pathway and mTOR inhibitors in cancer: progress and challenges. Cell Biosci. 2020;10(1):1-11.
[53] LI A, FAN S, XU Y, et al. Rapamycin treatment dose-dependently improves the cystic kidney in a new ADPKD mouse model via the mTORC 1 and cell-cycle-associated CDK 1/cyclin axis. J Cell Mol Med. 2017;21(8):1619-1635.
[54] ZHENG Y, JIANG Y. mTOR inhibitors at a glance. Mol Cell Pharmacolo. 2015; 7(2):15-20.
[55] MACKEIGAN JP, KRUEGER DA. Differentiating the mTOR inhibitors everolimus and sirolimus in the treatment of tuberous sclerosis complex. Neuro Oncol. 2015;17(12):1550-1559.
[56] JIANG K, HUANG Y, CHUNG EJ. Combining metformin and drug-loaded kidney-targeting micelles for polycystic kidney disease. Cell Mol Bioeng. 2023;16(1):55-67.
[57] COX A, TUNG M, LI H, et al. In vitro delivery of mTOR inhibitors by kidney-targeted micelles for autosomal dominant polycystic kidney disease. SLAS Technol. 2023;28(4):223-229.
[58] CHAUDESAYGUES E, GRASSE M, MARCHAND L, et al. Nephrotic syndrome revealed by pulmonary embolism: About four cases. Ann Cardiol Angeiol (Paris). 2014;63(5):385-388.
[59] COUSER WG. Pathogenesis of glomerular damage in glomerulonephritis. Nephrol Dial Transplant. 1998;13(suppl_1):10-15.
[60] FLOEGE J, RAUEN T, TANG SC. Current treatment of IgA nephropathy. Semin Immunopathol. 2021;43:717-728.
[61] SCINDIA Y, DESHMUKH U, THIMMALAPURA PR, et al. Anti-alpha8 integrin immunoliposomes in glomeruli of lupus-susceptible mice: a novel system for delivery of therapeutic agents to the renal glomerulus in systemic lupus erythematosus. Arthritis Rheum. 2008;58(12):3884-3891.
[62] ZHOU L, YE Z, ZHANG E, et al. Co-delivery of dexamethasone and captopril by α8 integrin antibodies modified liposome-plga nanoparticle hybrids for targeted anti-inflammatory/anti-fibrosis therapy of glomerulonephritis. Int J Nanomedicine. 2022;17:1531-1547. |