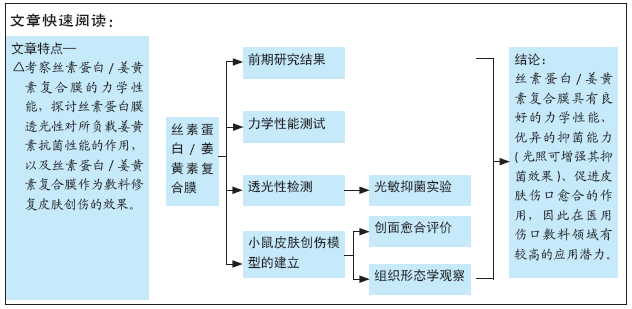
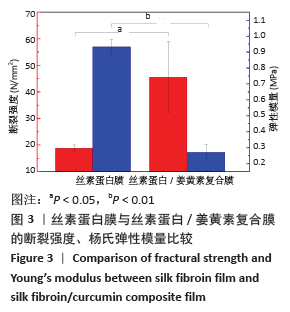
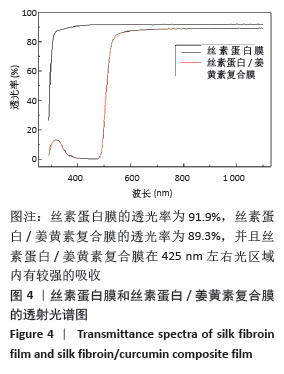
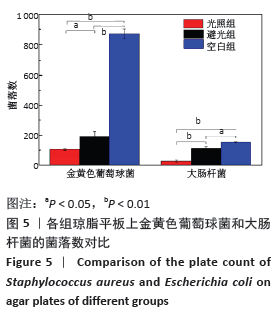
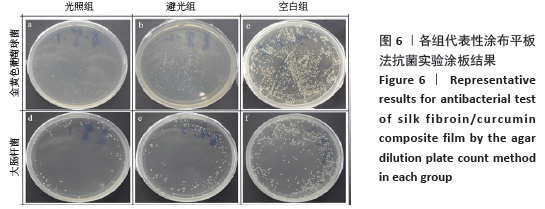
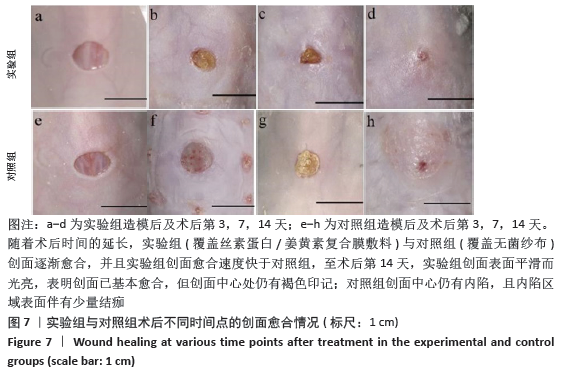
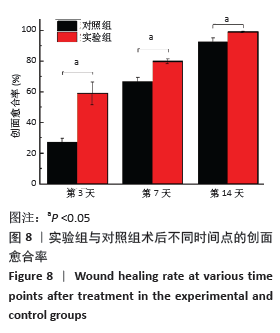
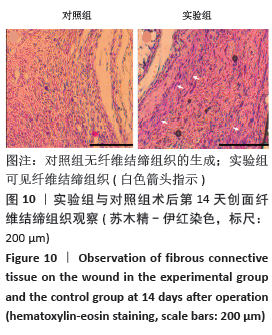
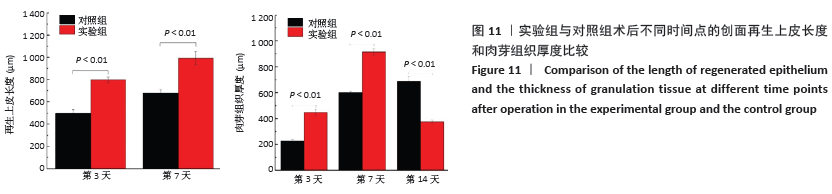
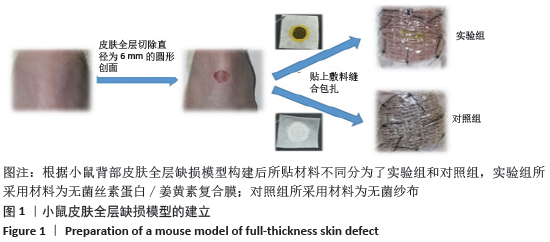
[1] MCGILL M, GRANT JM, KAPLAN DL, et al. Enzyme-mediated conjugation of peptides to silk fibroin for facile hydrogel functionalization. Ann Biomed Eng. 2020;48(7):1905-1915.
[2] YANG N, QI P, REN J, et al. Polyvinyl alcohol/silk fibroin/borax hydrogel ionotronics: a highly stretchable, self-healable, and biocompatible sensing platform. ACS Appl Mater Inter. 2019;11(26):23632-23638.
[3] LI DW, HE J, HE FL, et al. Silk fibroin/chitosan thin film promotes osteogenic and adipogenic differentiation of rat bone marrow-derived mesenchymal stem cells. J Biomater Appl. 2018;32(9): 088532821875776.
[4] GUAN Y, SUN F, ZHANG X, et al. Silk fibroin hydrogel promote burn wound healing through regulating TLN1 expression and affecting cell adhesion and migration. J Mater Sci Mater Med. 2020;31:48.
[5] YANG C, CHEN S, SU H, et al. Biocompatible, small-sized and well-dispersed gold nanoparticles regulated by silk fibroin fiber from Bombyx mori cocoons. Front Mater Sci. 2019;13(2):126-132.
[6] RAMESHBABU AP, BANKOTI K, DATTA S, et al. Bioinspired 3D porous human placental derived extracellular matrix/silk fibroin sponges for accelerated bone regeneration. Mat Sci Eng C-Mater. 2020;113: 110990.
[7] GOMAA F, FAWAL E, MARWA M, et al. Hydroxyethyl cellulose hydrogel for wound dressing: Fabrication, characterization and in vitro evaluation. Int J Biol Macromol. 2018;111:649-659.
[8] MIGUEL SP, SIMÕES D, MOREIRA AF, et al. Production and characterization of electrospun silk fibroin based asymmetric membranes for wound dressing applications. Int J Biol Macromol. 2019;121:524-535.
[9] NOURMOHAMMADI J, HADIDI M, NAZARPAK MH, et al. Physicochemical and antibacterial characterization of nanofibrous wound dressing from silk fibroin-polyvinyl alcohol-elaeagnus angustifolia extract. Fiber Polym. 2020; 21(3):456-464.
[10] ZHANG X, CHEN Z, BAO H, et al. Fabrication and characterization of silk fibroin/curcumin sustained-release film. Mater. 2019;12(20):3340.
[11] WU CN, FUH SC, LIN SP, et al. Tempo-oxidized bacterial cellulose pellicle with silver nanoparticles for wound dressing. Biomacromolecules. 2018;19(2):544-554.
[12] SIMÕES D, MIGUEL SP, RIBEIRO MP, et al. Recent advances on antimicrobial wound dressing: A review. Eur J Pharm Biopharm. 2018; 127:130-141.
[13] PATIL S, SINGH N. Antibacterial silk fibroin scaffolds with green synthesized silver nanoparticles for osteoblast proliferation and human mesenchymal stem cell differentiation. Colloid Surface B. 2019;176:150-155.
[14] KARATEPE UY, OZDEMIR T. Improving mechanical and antibacterial properties of PMMA via polyblend electrospinning with silk fibroin and polyethyleneimine towards dental applications. Bioact Mater. 2020;5(3):510-515.
[15] MAITI P, PALADUGU L, DUNBAR GL. Solid lipid curcumin particles provide greater anti-amyloid, anti-inflammatory and neuroprotective effects than curcumin in the 5xFAD mouse model of Alzheimer’s disease. BMC Neurosci. 2018;19(1):7-25.
[16] TSUDA T. Curcumin as a functional food-derived factor: degradation products, metabolites, bioactivity, and future perspectives. Food Funct. 2018;9(2):705-714.
[17] DE OLIVEIRA EF, TOSATI JV, TIKEKAR RV, et al. Antimicrobial activity of curcumin in combination with light against Escherichia coli O157:H7 and Listeria innocua : Applications for fresh produce sanitation. Postharvest Biol Technol. 2018;137:86-94.
[18] ZINELIS S, BRANTLEY W. Orthodontic applications of biomaterials. Chapter 3 - Structure/property relationships in orthodontic ceramics. Woodhead Publishing, 2017:61-71.
[19] PÉREZ L, CABRERA I, SANTIAGO AA, et al. Effect of the Al–CNT interlayer on the tensile elastic modulus of Al matrix composites with random dispersion of CNTs. J Braz Soc Mech Sci. 2018;40(11):550.
[20] BORTHAGARAY G. Inorganic frameworks as smart nanomedicines.Chapter 8 - Silver-containing nanoparticles in the research of new antimicrobial agents against ESKAPE pathogens. William Andrew Publishing. 2018:317-386.
[21] MARSLIN G, SELVAKESAVAN RK, FRANKLIN G, et al. Antimicrobial activity of cream incorporated with silver nanoparticles biosynthesized from Withania somnifera. Int J Nanomed. 2014;10:5955-5963.
[22] MARIANELLI C, PETRUCCI P, COMELLI MC, et al. Silver sucrose octasulfate (IASOS) as a valid active ingredient into a novel vaginal gel against human vaginal pathogens: in vitro antimicrobial activity assessment. PLoS One. 2014;9(6):97791-97798.
[23] VIEIRA AC, VICENTE AF, PEREZ R, et al. Chloral hydrate anesthesia and lens opacification in mice. Curr Eye Res. 2009;34(5):355-359.
[24] BERMUDEZ MA, VICENTE AF, ROMERO MC, et al. Time course of cold cataract development in anesthetized mice. Curr Eye Res. 2011; 36(3):278-284.
[25] HUANG Y, SHI F, WANG L, et al. Preparation and evaluation of Bletilla striata polysaccharide/carboxymethyl chitosan/carbomer 940 hydrogel for wound healing. Int J Biol Macomol. 2019;132:729-737.
[26] FAN Z, LIU B, WANG J, et al. A novel wound dressing based on Ag/Graphene polymer hydrogel: effectively kill bacteria and accelerate wound healing. Adv Fun Mater. 2014;24(25):3933-3943.
[27] BORRERO-LOPEZ O, HOFFMAN M. Measurement of fracture strength in brittle thin films. Surf Coat Tech. 2014;254:1-10.
[28] GADHAVE RV, MAHANWAR PA, GADEKAR PT. Effect of glutaraldehyde on thermal and mechanical properties of starch and polyvinyl alcohol blends. Des Monomers Polym. 2019;22(1):164-170.
[29] ARIANITA A, CAHYANINGTYAS, AMALIA B, et al. Effect of glutaraldehyde to the mechanical properties of chitosan/nanocellulose. J Phys Conf Ser. 2019;1317:012045.
[30] LEE SY, MOHAN DJ, KANG IA, et al. Nanocellulose reinforced PVA composite films: effects of acid treatment and filler loading. Fiber Polym. 2009;10(1):77-82.
[31] TONG WY, BIN ABDULLAH AYK, BINTI ROZMAN NAS, et al. Antimicrobial wound dressing film utilizing cellulose nanocrystal as drug delivery system for curcumin. Cellulose. 2018;25:631-638.
[32] PRIYADARSINI KI. The chemistry of curcumin: from extraction to therapeutic agent. Molecules. 2014;19(12):20091-20112.
[33] LIANG J, ZHANG X, CHEN Z, et al. Thiol-ene click reaction initiated rapid gelation of PEGDA/silk fibroin hydrogels. Polymers. 2019;11(12):2102.
[34] SCHREML S, SZEIMIES RM, PRANTL L, et al. Wound healing in the 21st century. J Am Acad Dermatol. 2010;63:866-881.
[35] SALIM MN, SILVIA M, ALIZA D, et al. Efficacy of jatropha curcas latex cream in the epithelialization phase of wound healing in mice skin. E3S Web Conf. 2020;151:01038.
[36] XU R, LUO G, XIA H, et al. Novel bilayer wound dressing composed of silicone rubber with particular micropores enhanced wound re-epithelialization and contraction. Biomaterials. 2015;40:1-11.
[37] SEIFERT AW, MADEN M. New insights into vertebrate skin regeneration. Int Rev Cell Mol Biol. 2014;310:129-169.
[38] CLARK RAF. Principles of Tissue Engineering. Chapter 76 - Wound Repair: Basic Biology to Tissue Engineering. Academic Press. 2014: 1595-1617.
[39] JONES K. Fibrotic response to biomaterials and all associated sequence of fibrosis. Host Response Biomater. 2015:189-237.
[40] BALAKRISHNAN B, MOHANTY M, UMASHANKAR PR, et al. Evaluation of an in situ forming hydrogel wound dressing based on oxidized alginate and gelatin. Biomaterials. 2005;26(32):6335-6342.
[41] BUSH K, GERTZMAN AA. Skin tissue engineering and regenerative medicine. Chapter 5 - Process development and manufacturing of human and animal acellular dermal matrices. Academic Press. 2016: 83-108.
|