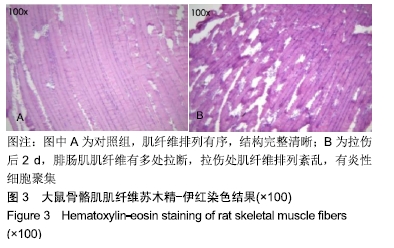
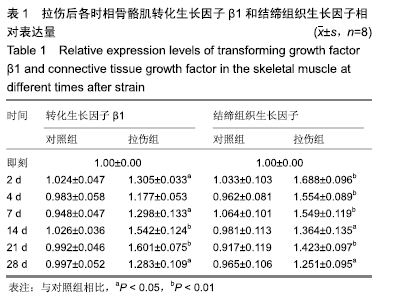
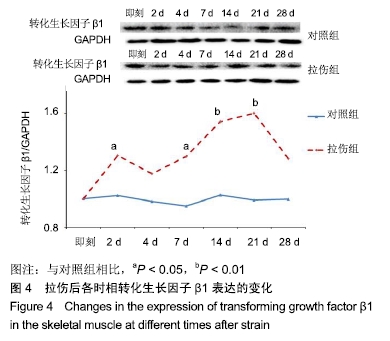
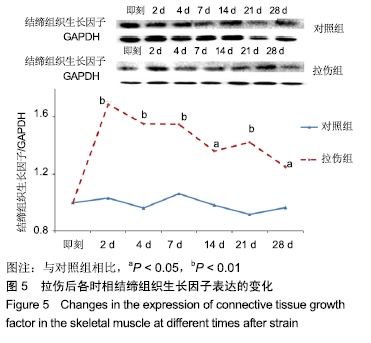
[1] LAI S, PANARESE A, LAWRENCE R, et al. A murine model of robotic training to evaluate skeletal muscle recovery after Injury. Med Sci Sports Exerc. 2017;49(4):840-847.
[2] PARK JK, KI MR, LEE EM, et al. Losartan improves adipose tissue-derived stem cell niche by inhibiting transforming growth factor-β and fibrosis in skeletal muscle injury. Cell Transplant.2012;21(11):2407-2424.
[3] 刘洪柏,区丽明.富血小板血浆联合转化生长因子-β1抗体对大鼠骨骼肌损伤修复的研究[J].生物医学工程与临床,2018,22(2): 187-190.
[4] 马晓燕,闫渊,崔峥.肾衰饮对TGF-β1诱导的下游囚子CTGF及FN的调控作用[J].中华医药学刊,2015,33(7):1543-1545.
[5] 董丽霞,蒋小岗,李梦姣,等.黄岑素对TGF_β1体外诱导肺成纤维细胞向肌成纤维细胞转化的影响[J].中国药理学通报,2013, 29(3):406-412.
[6] SBINALE FG, VILLARREAL F. Targeting matrix metalloproteinases in heart disease: Lessons from endogenous inhibitors. Biochem Pharmacol.2014;90(1):7-13.
[7] 于学平,孙晓伟,邹伟,等.头针对脑缺血再灌注大鼠脑组织中组织金属蛋白酶抑制因子-1表达调控机制的动态研究[J].中国康复医学杂志,2017,32(3):283-287.
[8] ZHU J, LI Y, LU A, et al. Follistatin improves skeletal muscle healing after injury and disease through an interaction with muscle regeneration, angiogenesis, and fibrosis. Am J Pathol. 2011;179(2):915-930.
[9] GUITART M, LLORETA J, MAÑAS-GARCIA L, et al. Muscle regeneration potential and satellite cell activation profile during recovery following hindlimb immobilization in mice. J Cell Physiol. 2018;233(5):4360-4372.
[10] PEAKE JM, NEUBAUER O, DELLA GATTA PA, et al. Muscle damage and inflammation during recovery from exercise. J Appl Physiol (1985). 2017;122(3):559-570.
[11] BAOGE L, VAN DEN STEEN E, RIMBAUT S, et al. Treatment of skeletal muscle injury: a review. ISRN Orthop. 2012;2012: 689012.
[12] LEPPER C, PARTRIDGE TA, FAN CM. An absolute requirement for Pax7-positive satellite cells in acute injury-induced skeletal muscle regeneration.Development. 2011;138(17):3639-3646.
[13] DUCHESNE E, DUFRESNE SS, DUMONT NA. Impact of inflammation and anti-inflammatory modalities on skeletal muscle healing: from fundamental research to the clinic.Phys Ther. 2017;97(8):807-817.
[14] MAHDY MAA. Skeletal muscle fibrosis: an overview. Cell Tissue Res. 2019;375(3):575-588.
[15] JÄRVINEN TAH, JÄRVINEN M, KALIMO H. Regeneration of injured skeletal muscle after the injury. Muscles Ligaments Tendons J. 2014;3(4):337.
[16] DUEWEKE JJ, AWAN TM, MENDIAS CL. Regeneration of skeletal muscle after eccentric injury. J Sport Rehabil.2017; 26(2):171-179.
[17] 刘镕,赵琴平,董惠芬,等. TGF-β信号传导通路及其生物学功能[J].中国寄生虫病防治杂志,2014, 9(1):77-83.
[18] MORALES MG, GUTIERREZ J, CABELLO-VERRUGIO C, et al. Reducing CTGF/CCN2 slows down mdx muscle dystrophy and improves cell therapy. Hum Mol Genet. 2013;22(24): 4938-4951.
[19] 石斌豪,徐宗佩,樊官伟.基于TGF-β1/Smads信号通路治疗心肌梗死后心肌纤维化的研究进展[J].中国药理学通报,2018,34(1): 5-8.
[20] 王鹏. TGF-β1介导TGF-β1/Smad及ERK/MAPK通路协同促进大鼠骨髓炎瘢痕形成的机制研究[D].济南:山东大学, 2017.
[21] BIERNACKA A, DOBACZEWSKI M, FRANGOGIANNIS NG. TGF-β signaling in fibrosis. Growth Factors. 2011;29(5): 196-202
[22] BALAH A, EZZAT O, AKOOL ES. Vitamin E inhibits cyclosporin A-induced CTGF and TIMP-1 expression by repressing ROS-mediated activation of TGF-β/Smad signaling pathway in rat liver. Int Immunopharmacol.2018;65:493-502.
[23] ISMAEEL A, KIM JS, KIRK JS, et al. Role of Transforming Growth Factor-β in Skeletal Muscle Fibrosis: A Review. Int J Mol Sci. 2019;20(10). pii: E2446.
[24] LI Y, HUARD J. Differentiation of muscle-derived cells into myofibroblasts in injured skeletal muscle. Am J Pathol. 2002; 161(3):895-907.
[25] SHEN W, LI Y, TANG Y, et al. NS-398, a cyclooxygenase-2- specific inhibitor, delays skeletal muscle healing by decreasing regeneration and promoting fibrosis. Am J Pathol. 2005;167(4):1105-1117.
[26] LI Y, NEGISHI S, SAKAMOTO M, et al. The use of relaxin improves healing in injured muscle. Ann N Y Acad Sci. 2005; 1041:395-397.
[27] NEGISHI S, LI Y, USAS A, et al. The effect of relaxin treatment on skeletal muscle injuries. Am J Sports Med. 2005; 33(12):1816-1824.
[28] LIU S, SHI-WEN X, ABRAHAM DJ, et al. CCN2 is required for bleomycin-induced skin fibrosis in mice. Arthritis Rheum. 2011; 63(1):239-246.
[29] SONG H, REN J. Protocatechuic acid attenuates angiotensin II‐induced cardiac fibrosis in cardiac fibroblasts through inhibiting the NOX4/ROS/p38 signaling pathway. Phytother Res. 2019;33(9):2440-2447.
[30] MORALES MG, ACUÑA MJ, CABRERA D, et al. The pro-fibrotic connective tissue growth factor (CTGF/CCN2) correlates with the number of necrotic-regenerative foci in dystrophic muscle. J Cell Commun Signal. 2018;12(1): 413-421.
[31] VIAL C, GUTIÉRREZ J, SANTANDER C, et al. Decorin interacts with connective tissue growth factor (CTGF)/CCN2 by LRR12 inhibiting its biological activity. J Biol Chem. 2011; 286(27):24242-24252.
[32] MORALES MG, CABRERA D, CÉSPEDES C, et al. Inhibition of the angiotensin-converting enzyme decreases skeletal muscle fibrosis in dystrophic mice by a diminution in the expression and activity of connective tissue growth factor (CTGF/CCN-2). Cell Tissue Res. 2013;353(1):173-187.
[33] KULAR L, RIVAT C, LELONGT B, et al. NOV/CCN3 attenuates inflammatory pain through regulation of matrix metalloproteinases-2 and-9. J Neuroinflammation. 2012;9:36.
[34] NUAMNAICHATI N, SATO VH, MOONGKARNDI P, et al. Sustained β-AR stimulation induces synthesis and secretion of growth factors in cardiac myocytes that affect on cardiac fibroblast activation. Life Sci. 2018;193: 257-269.
[35] WANG X, MCLENNAN SV, ALLEN TJ, et al. Regulation of pro-inflammatory and pro-fibrotic factors by CCN2/CTGF in H9c2 cardiomyocytes. J Cell Commun Signal. 2010;4(1): 15-23.
[36] TSUCHIYA M, KALURUPALLE S, KUMAR P, et al. RPTOR, a novel target of miR-155, elicits a fibrotic phenotype of cystic fibrosis lung epithelium by upregulating CTGF. RNA Biology. 2016;13(9):837-847.
[37] CABELLO-VERRUGIO C, ACUÑA MJ, MORALES MG, et al. Fibrotic response induced by angiotensin-II requires NAD(P)H oxidase-induced reactive oxygen species (ROS) in skeletal muscle cells. Biochem Biophys Res Commun. 2011; 410(3):665-670.
[38] TALL EG. TGF-β-stimulated CTGF production enhanced by collagen and associated with biogenesis of a novel 31-kDa CTGF form in human corneal fibroblasts. Invest Ophth Vis Sci,2010;51(10):5002-5011.
[39] 潘锐焕,孙景波,黄燕. MMP/TIMP平衡与缺血性脑卒中的关系[J]. 时珍国医国药,2013,24(4):972-973+976.
[40] JUNG P, ZIMOWSKA M. Matrix metalloproteinase in development, physiology and degenerative processes of skeletal muscles. Post Biochem.2016;62(1): 25-35.
[41] PRIHADI JC, SUGANDI S, SIREGAR NC, et al. Imbalance in extracellular matrix degradation in urethral stricture. Res Rep Urol. 2018;10:227.
[42] ALAMEDDINE HS, MORGAN JE. Matrix metalloproteinases and tissue inhibitor of metalloproteinases in inflammation and fibrosis of skeletal muscles. J Neuromuscul Dis. 2016;3(4): 455-473.
[43] LIN SW, GAO ZX, LIN LR, et al. Treponema pallidum enhances human monocyte migration and invasion by dysregulating the MMP/TIMP balance. Int Immunopharmacol. 2019;75:105744.
[44] JAYASOORIYA PR, NADEEKA JAYASINGHE KA, MUDIYANSELAGE TILAKARATNE W. Relationship between thickness of fibrosis and epithelial dysplasia in oral submucous fibrosis. J Investig Clin Dent. 2011;2(3):171-175.
[45] DAI JP, ZHU DX, SHENG JT, et al. Inhibition of tanshinone IIA, salvianolic acid A and salvianolic acid B on areca nut extract-induced oral submucous fibrosis in vitro. Molecules. 2015;20(4):6794-6807.
[46] YANG L, LIU R, WANG X, et al. Imbalance between matrix metalloproteinase‐1 (MMP‐1) and tissue inhibitor of metalloproteinase‐1 (TIMP‐1) contributes to bladder compliance changes in rabbits with partial bladder outlet obstruction (PBOO). BJU Int. 2013;112(4):E391-397.
[47] LAN HY. Diverse roles of TGF-β/Smads in renal fibrosis and inflammation. Int J Biol Sci. 2011;7(7): 1056.
[48] ULRICH D, LICHTENEGGER F, EBLENKAMP M, et al. Matrix metalloproteinases, tissue inhibitors of metalloproteinases, aminoterminal propeptide of procollagen type III, and hyaluronan in sera and tissue of patients with capsular contracture after augmentation with Trilucent breast implants. Plast Reconstr Surg. 2004;114(1):229-236.
[49] 王世强,常芸,马晓雯,等.不同强度耐力运动对大鼠心肌胶原蛋白的影响及MMP-1/TIMP-1的调节作用[J].中国体育科技,2015, 51(5):60-66.
[50] WRIGHT KJ, THOMAS MM, BETIK AC, et al. Exercise training initiated in late middle age attenuates cardiac fibrosis and advanced glycation end-product accumulation in senescent rats. Exp Gerontol.2014;50:9-18.
|