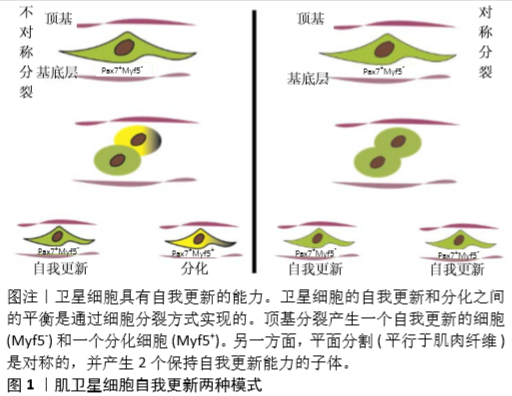
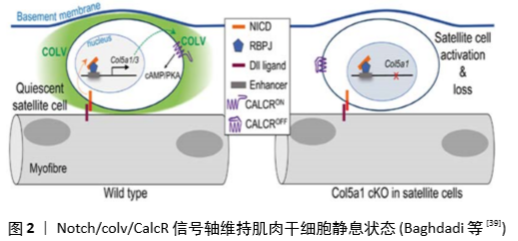
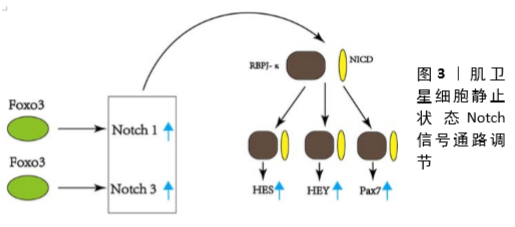
[1] TIERNEY MT, SACCO A. Satellite Cell Heterogeneity in Skeletal Muscle Homeostasis. Trends Cell Biol. 2016;26(6):434-444.
[2] ANDES-KOBACK M, KEATING CD. Complete budding and asymmetric division of primitive model cells to produce daughter vesicles with different interior and membrane compositions. J Am Chem Soc. 2011; 133(24):9545-9555.
[3] HOU GY, ZHOU HL, CAO T, et al. Expression and variation of Myf5 and MyoD1 genes in different tissues of Wuzhishan pigs. Genet Mol Res. 2015;14(2):3729-3735.
[4] BOWLIN KM, EMBREE LJ, GARRY MG,et al. Kbtbd5 is regulated by MyoD and restricted to the myogenic lineage. Differentiation. 2013; 86(4-5):184-191.
[5] MILEWSKA M, GRABIEC K, GRZELKOWSKA-KOWALCZYK K. Interactions of proliferation and differentiation signaling pathways in myogenesis. Postepy Hig Med Dosw (Online). 2014;68:516-526.
[6] QAHAR M, TAKUMA Y, MIZUNOYA W, et al. Semaphorin 3A promotes activation of Pax7, Myf5, and MyoD through inhibition of emerin expression in activated satellite cells. FEBS Open Bio. 2016;6(6):529-539.
[7] D’AGOSTINO M, TORCINARO A, MADARO L, et al. Role of mir-200c in myogenic differentiation impairment via p66shc: implication in skeletal muscle regeneration of dystrophic mdx mice. Oxid Med Cell Longev. 2018;2018:4814696.
[8] ZANOU N, GAILLY P. Skeletal muscle hypertrophy and regeneration: interplay between the myogenic regulatory factors (MRFs) and insulin-like growth factors (IGFs) pathways. Cell Mol Life Sci. 2013; 70(21):4117-4130.
[9] SJÖSTRÖM M, THORNELL LE, CEDERGREN E. The application of cryo microtomy in the study of the fine structure of myofilaments. J Microscopy. 2011;99(2):193-204.
[10] PADILLA-BENAVIDES T, NASIPAK BT, IMBALZANO AN. Brg1 Controls the Expression of Pax7 to Promote Viability and Proliferation of Mouse Primary Myoblasts. J Cell Physiol. 2015;230(12):2990-2997.
[11] JANG YJ, SON HJ, KIM JS, et al. Coffee consumption promotes skeletal muscle hypertrophy and myoblast differentiation. Food Funct. 2018; 9(2):1102-1111.
[12] PANDURANGAN M, MOORTHY H, Sambandam R, et al. Effects of stress hormone cortisol on the mRNA expression of myogenenin, MyoD, Myf5, PAX3 and PAX7. Cytotechnology. 2014;66(5):839-844.
[13] MASHINCHIAN O, PISCONTI A, MOAL EL, et al. The Muscle Stem Cell Niche in Health and Disease. Curr Top Dev Biol. 2018;126:23-65.
[14] BENTZINGER CF, WANG YX, DUMONT NA, et al. Cellular dynamics in the muscle satellite cell niche. Embo Rep. 2013;14(12):1062-1072.
[15] GIORDANI L, INCITTI T, MAGLI A, et al. Incitti T, Magli A, Darabi R, et al. Pluripotent stem cell-derived myogenic progenitors remodel their molecular signature upon in vivo engraftment. Proc Natl Acad Sci U S A. 2019;116(10):4346-4351.
[16] GIORDANI L, PARISI A, LE GRAND F. Satellite Cell Self-Renewal. Curr Top Dev Biol. 2018;126:177-203.
[17] FILIPPIN LI, MOREIRA AJ, MARRONI NP, et al. Nitric oxide and repair of skeletal muscle injury. Nitric Oxide. 2009;21(3-4):157-163.
[18] EBNER F, SEDLYAROV V, TASCIYAN S, et al. The RNA-binding protein tristetraprolin schedules apoptosis of pathogen-engaged neutrophils during bacterial infection. J Clin Invest. 2017;127(6):2051-2065.
[19] LV J, ZOU L, ZHAO L, et al. Leukotriene B₄-leukotriene B₄ receptor axis promotes oxazolone-induced contact dermatitis by directing skin homing of neutrophils and CD8⁺ T cells. Immunology. 2015;146(1):50‐58.
[20] NEDERVEEN JP, JOANISSE S, SNIJDERS T, et al. The influence of capillarization on satellite cell pool expansion and activation following exercise-induced muscle damage in healthy young men. J Physiol. 2018;596(6):1063-1078.
[21] YANAY N, ELBAZ M, KONIKOV-ROZENMAN J, et al. Pax7, Pax3 and Mamstr genes are involved in skeletal muscle impaired regeneration of dy2J/dy2J mouse model of Lama2-CMD. Hum Mol Genet. 2019; 28(20):3369-3390.
[22] BURM JS, HWANG J, LEE YK. A New Option for the Reconstruction of Primary or Recurrent Ischial Pressure Sores: Hamstring-Adductor Magnus Muscle Advancement Flap and Direct Closure. Ann Plast Surg. 2018;80(4):400-405.
[23] BOLDRIN L, MORGAN JE. Modulation of the host skeletal muscle niche for donor satellite cell grafting. Methods Mol Biol. 2013;1035:179-190.
[24] ROCHETEAU P, GAYRAUD-MOREL B, SIEGL-CACHEDENIER I, et al. A Subpopulation of Adult Skeletal Muscle Stem Cells Retains All Template DNA Strands after Cell Division. Cell. 2012;148(1-2):125.
[25] INSINGA A, CICALESE A, FARETTA M, et al. DNA damage in stem cells activates p21, inhibits p53, and induces symmetric self-renewing divisions. Proc Natl Acad Sci U S A. 2013;110(10):3931‐3936.
[26] ONO Y, MASUDA S, NAM HS, et al. Slow-dividing satellite cells retain long-term self-renewal ability in adult muscle. J Cell Sci. 2012; 125(5): 1309-1317.
[27] CHAKKALAKAL JV, CHRISTENSEN J, XIANG W, et al. Early forming labelretaining muscle stem cells require p27kip1 for maintenance of the primitive state. Development. 2014;141(8):1649-1659.
[28] KOKI K, RYOSUKE D, MAKOTO H, et al. The Ror1 receptor tyrosine kinase plays a critical role in regulating satellite cell proliferation during regeneration of injured muscle. J Biol Chem. 2017;292(38): 1593915951.
[29] QAHAR M, TAKUMA Y, MIZUNOYA W, et al. Semaphorin 3A promotes activation of Pax7, Myf5, and MyoD through inhibition of emerin expression in activated satellite cells. FEBS Open Bio. 2016;6(6):529-539.
[30] YANAY N, ELBAZ M, KONIKOV-ROZENMAN J, et al. Pax7, Pax3 and Mamstr genes are involved in skeletal muscle impaired regeneration of dy2J/dy2J mouse model of Lama2-CMD. Hum Mol Genet. 2019; 28(20):3369‐3390.
[31] ADDICKS GC, BRUN CE, SINCENNES MC, et al. MLL1 is required for PAX7 expression and satellite cell self-renewal in mice. Nat Commun. 2019;10(1):4256.
[32] SOLEIMANI VD. Transcriptional dominance of Pax7 in adult myogenesis is due to high-affinity recognition of homeodomain motifs. Dev Cell. 2012;22:1208-1220.
[33] LEPPER C, PARTRIDGE TA, FAN CM. An absolute requirement for Pax7-positive satellite cells in acute injury-induced skeletal muscle regeneration. Development. 2011;138(17):3639-3646.
[34] DICK SA, CHANG NC, DUMONT NA, et al. Caspase 3 cleavage of Pax7 inhibits self-renewal of satellite cells. Proc Natl Acad Sci U S A. 2015;112(38):E5246-E5252.
[35] LINKHART TA, CLEGG CH, HAUSCHKA SD. Control of mouse myoblast commitment to terminal differentiation by mitogens. J Supramol Struct. 1980;14(4):483-498.
[36] MOHAN A, ASAKURA A. CDK inhibitors for muscle stem cell differentiation and self-renewal. J Phys Fit Sports Med. 2017;6(2):65-74.
[37] THERET M, GSAIER L, SCHAFFER B, et al. AMPKα1-LDH pathway regulates muscle stem cell self-renewal by controlling metabolic homeostasis. EMBO J. 2017;36(13):1946-1962.
[38] ZISMANOV, CHICHKOV, VICTOR, et al. Phosphorylation of eIF2α Is a translational control mechanism regulating muscle stem cell quiescence and self-renewal. Cell Stem Cell. 2016;18(1):79-90.
[39] BAGHDADI MB, CASTEL D, MACHADO L, et al. Reciprocal signalling by Notch-Collagen V-CALCR retains muscle stem cells in their niche. Nature. 2018;557(7707):714-718.
[40] VERMA M, ASAKURA Y, MURAKONDA BSR, et al. Muscle Satellite Cell Cross-Talk with a Vascular Niche Maintains Quiescence via VEGF and Notch Signaling. Cell Stem Cell. 2018;23(4):530-543.
[41] YARTSEVA V, GOLDSTEIN LD, RODMAN J, et al. Heterogeneity of Satellite Cells Implicates DELTA1/NOTCH2 Signaling in Self-Renewal. Cell Rep. 2020;30(5):1491-1503.e6.
[42] FUJIMAKI S, SEKO D, KITAJIMA Y, et al. Notch1 and Notch2 Coordinately Regulate Stem Cell Function in the Quiescent and Activated States of Muscle Satellite Cells. Stem Cells. 2018;36(2):278-285.
[43] CASTEL D, BAGHDADI MB, MELLA S, et al. Small-RNA sequencing identifies dynamic microRNA deregulation during skeletal muscle lineage progression. Sci Rep. 2018;8(1):4208.
[44] GOPINATH SD, WEBB AE, BRUNET A, et al. FOXO3 promotes quiescence in adult muscle stem cells during the process of self-renewal. Stem Cell Rep. 2014;2(4):414-426.
[45] CHAKKALAKAL JV, JONES KM, BASSON MA, et al. The aged niche disrupts muscle stem cell quiescence. Nature. 2012;490(7420):355-360.
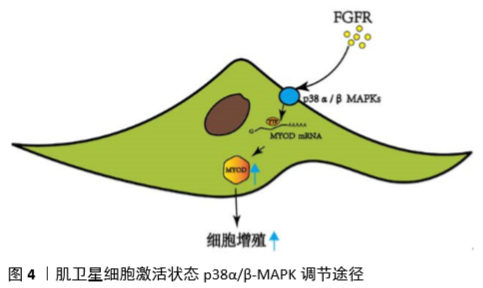
[46] TROY A, CADWALLADER A, FEDOROV Y, et al. Coordination of Satellite Cell Activation and Self-Renewal by Par-Complex-Dependent Asymmetric Activation of p38α/β MAPK. Cell Stem Cell. 2012;11(4): 541-553.
[47] Bernet JD, Doles JD, Hall JK, et al. P38 MAPK signaling underlies a cell autonomous loss of stem cell self-renewal in aged skeletal muscle. Nature Med. 2014;20(3):265-271.
[48] MELISSA AH, JASON DD, SANDRA LC, et al. Post-transcriptional regulation of satellite cell quiescence by TTP-mediated mRNA decay. ELife. 2015;4(4):e03390.
[49] BRIEN P, PUGAZHENDHI D, WOODHOUSE S, et al. p38α MAPK regulates adult muscle stem cell fate by restricting progenitor proliferation during postnatal growth and repair. Stem Cells. 2013;31(8):1597-1610.
[50] PAPACONSTANTINOU J, WANG CZ, ZHANG M, et al. Attenuation of p38α MAPK stress response signaling delays the in vivo aging of skeletal muscle myofibers and progenitor cells. Aging (Albany NY). 2015;7(9):718-733.
[51] BERNET JD, DOLES JD, HALL JK, et al. p38 MAPK signaling underlies a cell-autonomous loss of stem cell self-renewal in skeletal muscle of aged mice. Nat Med. 2014;20(3):265-271.
[52] ZHANG J, HE L, YANG Z, et al. Lithium chloride promotes proliferation of neural stem cells in vitro, possibly by triggering the Wnt signaling pathway. Anim Cells Syst (Seoul). 2018;23(1):32-41.
[53] LU X, CHEN X, XING J, et al. miR-140-5p regulates the odontoblastic differentiation of dental pulp stem cells via the Wnt1/β-catenin signaling pathway. Stem Cell Res Ther. 2019;10(1):226.
[54] TAKATA H, TERADA K, OKA H, et al. Involvement of Wnt4 signaling during myogenic proliferation and differentiation of skeletal muscle. Dev Dyn. 2007;236(10):2800-2807.
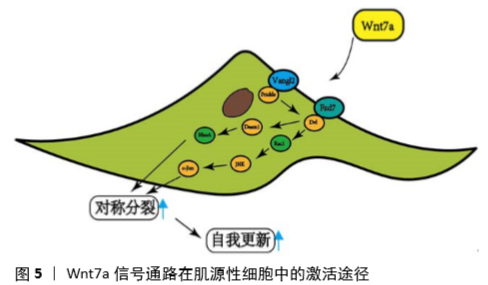
[55] VON MALTZAHN J, BENTZINGER CF, RUDNICKI MA. Wnt7a-Fzd7 signalling directly activates the Akt/mTOR anabolic growth pathway in skeletal muscle. Nat Cell Biol. 2011;14(2):186-191.
[56] BENTZINGER C, WANG Y, VONMALTZAHN J, et al. Fibronectin Regulates Wnt7a Signaling and Satellite Cell Expansion. Cell Stem Cell. 2013; 12(1):75-87.
[57] SONIA AM, Frédéric A, DESPOINA M, et al. SOXF factors regulate murine satellite cell self-renewal and function through inhibition of β-catenin activity. ELife. 2018;7:e26039.
[58] KNOBLICH JA. Asymmetric cell division: recent developments and their implications for tumour biology. Nat Rev Mol Cell Biol. 2010; 11(12):849-860.
[59] BENEDETTI A, FIORE PF, MADARO L, et al. Targeting PKCθ Promotes Satellite Cell Self-Renewal. Int J Mol Sci. 2020;21(7):E2419.
[60] SU Y, YU Y, LIU C, et al. Fate decision of satellite cell differentiation and self-renewal by miR-31-IL34 axis. Cell Death Differ. 2020;27(3): 949-965. |