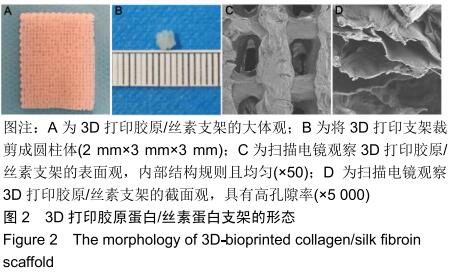
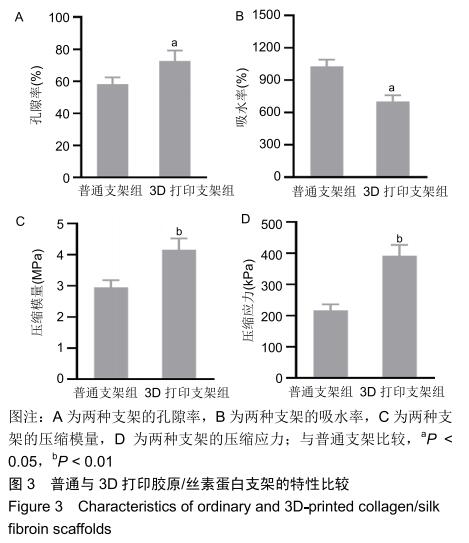
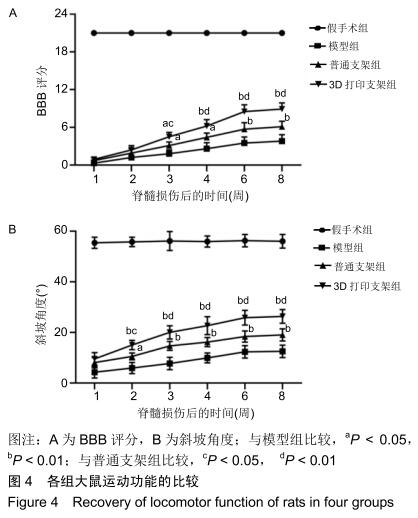
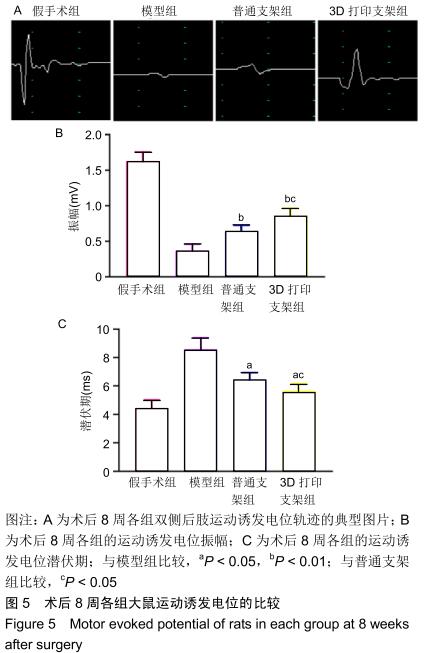
中国组织工程研究 ›› 2020, Vol. 24 ›› Issue (28): 4547-4554.doi: 10.3969/j.issn.2095-4344.2297
• 复合支架材料 composite scaffold materials • 上一篇 下一篇
弥散张量成像预测3D打印支架促进脊髓损伤后运动功能恢复
刘晓银1,2,钟 琳3,郑 博4,魏 攀5,代 晨2,胡良聪6,王甜甜6,梁小龙7,张 赛2,王晓丽2
1天津医科大学,天津市 300070;2中国人民武装警察部队特色医学中心,天津市 300162;3昆明医科大学第二附属医院,云南省昆明市 650101;4金堂县第二人民医院,四川省成都市 610404;5成都市龙泉驿区第一人民医院,四川省成都市 610100;6华中科技大学同济医学院附属协和医院,湖北省武汉市 430022;7德阳市人民医院,四川省德阳市 618000
Diffusion tensor imaging predicting locomotor function recovery with 3D printing scaffold after spinal cord injury
Liu Xiaoyin1, 2, Zhong Lin3, Zheng Bo4, Wei Pan5, Dai Chen2, Hu Liangcong6, Wang Tiantian6, Liang Xiaolong7, Zhang Sai2, Wang Xiaoli2
1Tianjin Medical University, Tianjin 300070, China; 2Characteristic Medical Center of People’s Armed Police Forces, Tianjin 300162, China; 3Second Affiliated Hospital of Kunming Medical University, Kunming 650101, Yunan Province, China; 4Second People's Hospital of Jintang County, Chengdu 610404, Sichuan Province, China; 5First People's Hospital of Longquanyi District, Chengdu 610100, Sichuan Province, China; 6Union Hospital Affiliated to Tongji Medical College of Huazhong University of Science and Technology, Wuhan 430022, Hubei Province, China; 7Deyang People's Hospital, Deyang 618000, Sichuan Province, China
摘要:
文题释义:
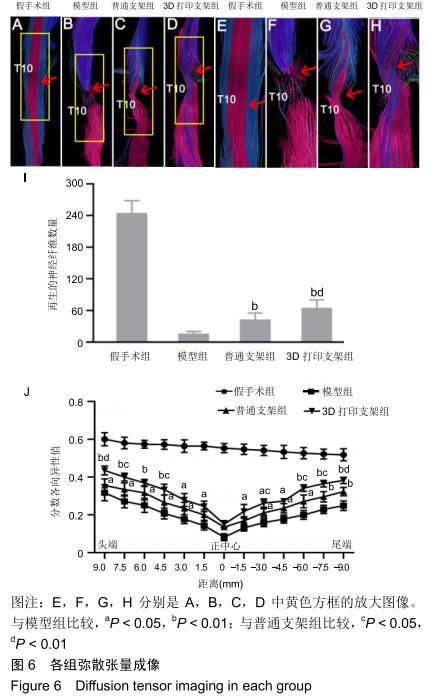
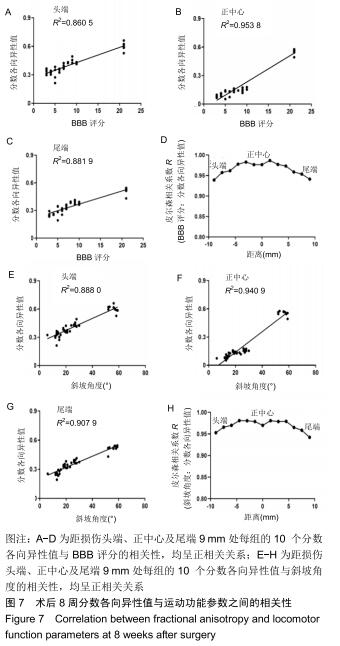
分数各向异性:通常用于提供关于推断组织特征和组织生理状态的定量信息,反映扩散的各向异性。分数各向异性值来自弥散张量成像数据,并且与运动结果大致相符,可用于评估脊髓损伤严重程度。分数各向异性值是脊髓损伤后敏感的特异性生物标志物,可量化脊髓损伤严重程度。实际上分数各向异性值反映白质的完整性,包括轴突结构损伤和脱髓鞘。分数各向异性值减少反映的是神经变性和轴突损失。损伤恢复越好分数各向异性值则越高。相比之下弥散张量成像比常规MRI检测脊髓损伤的恢复程度更敏感。分数各向异性值与损伤处的神经再生程度相关。
弥散张量纤维束成像:作为一个定性指标可直观地追踪脊髓损伤处的神经纤维束,清晰地观察轴突束的损伤。结合定量弥散张量成像扫描,弥散张量纤维束成像可有效显示脊髓损伤处神经纤维的变化,有效显示移植部位神经纤维束的变化。弥散张量纤维束成像结果能追踪到脊髓损伤大鼠损伤处的局部纤维,弥散张量纤维束成像图像可直观地表明脊髓损伤,展示脊髓损伤部位的脊髓连接。
背景:弥散张量成像作为一种基于MRI相对较新的方法,目前已成为神经影像学领域检查诊断的重要手段。
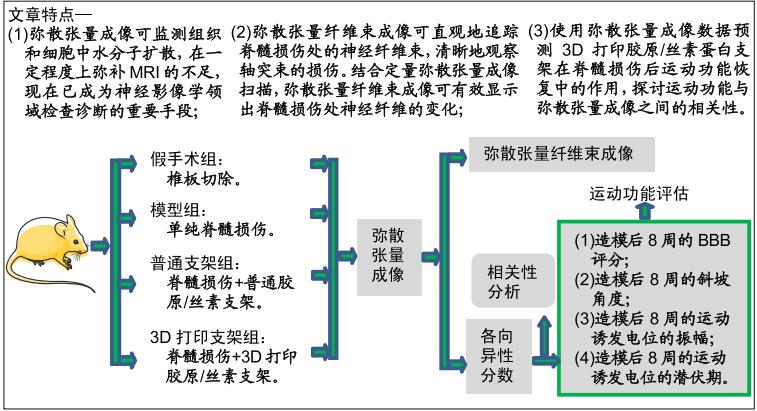
目的:使用弥散张量成像数据预测3D打印胶原/丝素蛋白支架在脊髓损伤后运动功能恢复中的作用,并探讨运动功能与弥散张量成像之间的相关性。
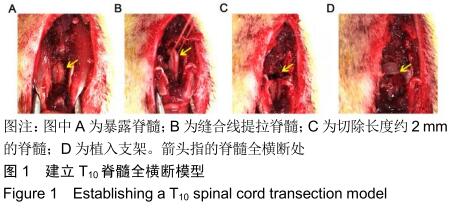
方法:制备普通胶原/丝素蛋白支架和3D打印胶原/丝素蛋白支架。取成年雌性SD大鼠40只(由中国人民解放军军事医学科学院实验动物中心提供),随机分为4组,每组10只:假手术组仅切除T10椎板;模型组制作T10脊髓全横断损伤模型;普通支架组、3D打印支架组建立T10脊髓全横断损伤模型后分别植入普通胶原/丝素蛋白支架和3D打印胶原/丝素蛋白支架。术后1,2,3,4,6,8周进行后肢BBB评分与斜板实验,术后8周进行后肢电生理检测,评估运动功能;术后8周进行腰椎弥散张量成像检查,并分析弥散张量成像参数与大鼠运动功能的相关性。动物实验获得中国人民武装警察部队特色医学中心研究动物伦理委员会的批准(伦理号:27653/58)。
结果与结论:①自术后3周起,3D打印支架组的BBB评分高于模型组、普通支架组(P < 0.05或P < 0.01);自术后2周起,3D打印支架组的斜板实验角度高于模型组、普通支架组(P < 0.05或P < 0.01);②3D打印支架组的运动诱发电位振幅大于模型组、普通支架组(P < 0.05或P < 0.01),运动诱发电位潜伏期短于模型组、普通支架组(P < 0.05或P < 0.01);③弥散张量成像显示,造模3组的神经纤维轨迹不规则,均缺乏神经纤维的连续性,但3D打印支架组的断端再生神经纤维束数量多于模型组、普通支架组(P < 0.01);3D打印支架组距离脊髓损伤正中心9,7.5,4.5,-3,-6,-7.5,-9 mm处的分数各向异性值均高于模型组、普通支架组 (P < 0.05或P < 0.01);④术后8周,BBB评分、斜坡角度、运动诱发电位振幅、运动诱发电位潜伏期与从大鼠头到尾的弥散张量成像分数各向异性值之间存在正相关;⑤结果表明,弥散张量成像可被用作有效预测指标来评估实验和临床病例中脊髓损伤的神经系统修复。
ORCID: 0000-0001-9409-4201(刘晓银)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程
中图分类号: