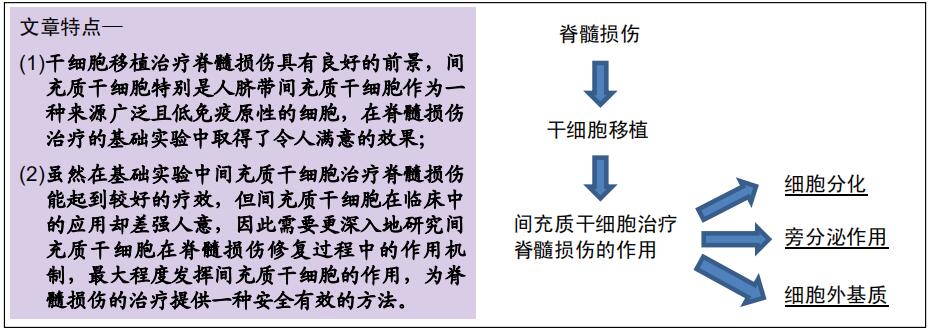
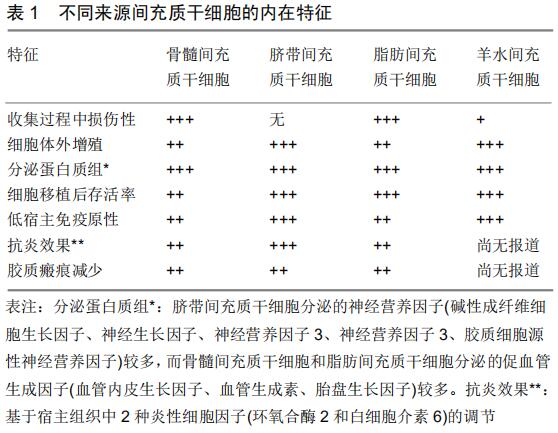
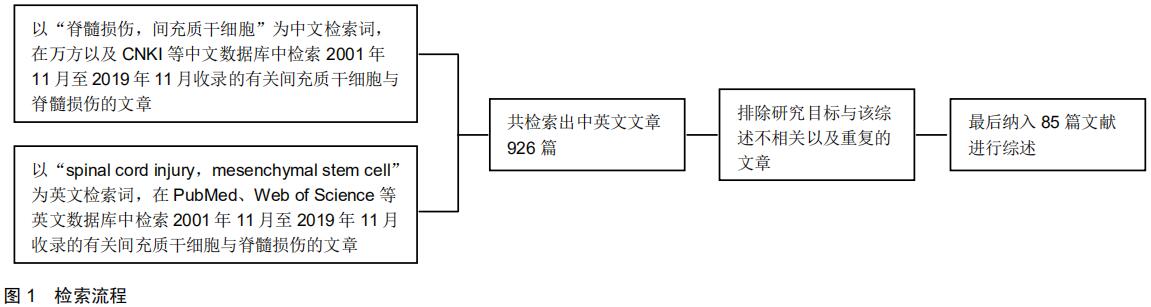
|
[1] KALRA H, SIMPSON RJ, JI H, et al. Vesiclepedia: a compendium for extracellular vesicles with continuous community annotation. PLoS Biol. 2012;10(12):e1001450.
[2] YILMAZ T, KAPTANOĞLU E. Current and future medical therapeutic strategies for the functional repair of spinal cord injury. World J Orthop. 2015;6(1):42-55.
[3] WILSON JR, TETREAULT LA, KWON BK, et al. Timing of Decompression in Patients With Acute Spinal Cord Injury: A Systematic Review. Global Spine J. 2017;7(3 Suppl): 95S-115S.
[4] COFANO F, BOIDO M, MONTICELLI M, et al. Mesenchymal Stem Cells for Spinal Cord Injury: Current Options, Limitations, and Future of Cell Therapy. Int J Mol Sci. 2019;20(11):E2698.
[5] SILVA NA, SOUSA N, REIS RL, et al. From basics to clinical: a comprehensive review on spinal cord injury. Prog Neurobiol. 2014;114: 25-57.
[6] VISMARA I, PAPA S, ROSSI F, et al. Current Options for Cell Therapy in Spinal Cord Injury. Trends Mol Med. 2017;23(9):831-849.
[7] FEHLINGS MG, NAKASHIMA H, NAGOSHI N, et al. Rationale, design and critical end points for the Riluzole in Acute Spinal Cord Injury Study (RISCIS): a randomized, double-blinded, placebo-controlled parallel multi-center trial. Spinal Cord. 2016;54(1):8-15.
[8] GAZDIC M, VOLAREVIC V, HARRELL CR, et al. Stem Cells Therapy for Spinal Cord Injury. Int J Mol Sci. 2018;19(4): E1039.
[9] GARCIA E, AGUILAR-CEVALLOS J, SILVA-GARCIA R, et al. Cytokine and Growth Factor Activation In Vivo and In Vitro after Spinal Cord Injury. Mediators Inflamm. 2016;2016:9476020.
[10] HAYTA E, ELDEN H. Acute spinal cord injury: A review of pathophysiology and potential of non-steroidal anti-inflammatory drugs for pharmacological intervention. J Chem Neuroanat.2018; 87:25-31.
[11] PAPA S, CARON I, ERBA E, et al. Early modulation of pro-inflammatory microglia by minocycline loaded nanoparticles confers long lasting protection after spinal cord injury. Biomaterials. 2016;75:13-24.
[12] PAPA S, CARON I, ROSSI F, et al. Modulators of microglia: a patent review. Expert Opin Ther Pat. 2016;26(4):427-437.
[13] SCHWAB ME, STRITTMATTER SM. Nogo limits neural plasticity and recovery from injury. Curr Opin Neurobiol. 2014;27:53-60.
[14] BELLER JA, SNOW DM. Proteoglycans: road signs for neurite outgrowth. Neural Regen Res. 2014;9(4):343-355.
[15] YUAN YM, HE C. The glial scar in spinal cord injury and repair. Neurosci Bull. 2013;29(4):421-435.
[16] SABIN KZ, JIANG P, GEARHART MD, et al. AP-1cFos/JunB/miR-200a regulate the pro-regenerative glial cell response during axolotl spinal cord regeneration. Commun Biol. 2019;2:91.
[17] VAN NIEKERK EA, TUSZYNSKI MH, LU P, et al. Molecular and Cellular Mechanisms of Axonal Regeneration After Spinal Cord Injury. Mol Cell Proteomics. 2016;15(2):394-408.
[18] TRAN AP, WARREN PM, SILVER J. The Biology of Regeneration Failure and Success After Spinal Cord Injury. Physiol Rev. 2018;98(2): 881-917.
[19] O'SHEA TM, BURDA JE, SOFRONIEW MV. Cell biology of spinal cord injury and repair. J Clin Invest. 2017;127(9):3259-3270.
[20] LIAU LL, RUSZYMAH BHI, NG MH, et al. Characteristics and clinical applications of Wharton's jelly-derived mesenchymal stromal cells. Curr Res Transl Med. 2019 Sep 19. doi: 10.1016/j.retram.2019.09.001. [Epub ahead of print]
[21] DASARI VR, VEERAVALLI KK, DINH DH. Mesenchymal stem cells in the treatment of spinal cord injuries: A review. World J Stem Cells. 2014;6(2):120-133.
[22] LU LL, LIU YJ, YANG SG, et al. Isolation and characterization of human umbilical cord mesenchymal stem cells with hematopoiesis- supportive function and other potentials. Haematologica. 2006;91(8): 1017-1026.
[23] LO B, PARHAM L. Ethical issues in stem cell research. Endocr Rev. 2009;30(3):204-213.
[24] FILIPPI M, BOIDO M, PASQUINO C, et al. Successful in vivo MRI tracking of MSCs labeled with Gadoteridol in a Spinal Cord Injury experimental model. Exp Neurol. 2016;282:66-77.
[25] ZACHAR L, BAČENKOVÁ D, ROSOCHA J. Activation, homing, and role of the mesenchymal stem cells in the inflammatory environment. J Inflamm Res. 2016;9:231-240.
[26] QU J, ZHANG H. Roles of Mesenchymal Stem Cells in Spinal Cord Injury. Stem Cells Int. 2017;2017:5251313.
[27] PELAGALLI A, NARDELLI A, LUCARELLI E, et al. Autocrine signals increase ovine mesenchymal stem cells migration through Aquaporin-1 and CXCR4 overexpression. J Cell Physiol. 2018;233(8): 6241-6249.
[28] MARQUEZ-CURTIS LA, GUL-ULUDAG H, XU P, et al. CXCR4 transfection of cord blood mesenchymal stromal cells with the use of cationic liposome enhances their migration toward stromal cell-derived factor-1. Cytotherapy. 2013;15(7):840-849.
[29] KIM Y, JO SH, KIM WH, et al. Antioxidant and anti-inflammatory effects of intravenously injected adipose derived mesenchymal stem cells in dogs with acute spinal cord injury. Stem Cell Res Ther. 2015;6:229.
[30] RAMALHO BDS, ALMEIDA FM, SALES CM, et al. Injection of bone marrow mesenchymal stem cells by intravenous or intraperitoneal routes is a viable alternative to spinal cord injury treatment in mice. Neural Regen Res. 2018;13(6):1046-1053.
[31] YANG C, WANG G, MA F, et al. Repeated injections of human umbilical cord blood-derived mesenchymal stem cells significantly promotes functional recovery in rabbits with spinal cord injury of two noncontinuous segments. Stem Cell Res Ther. 2018;9(1):136.
[32] WATSON N, DIVERS R, KEDAR R, et al. Discarded Wharton jelly of the human umbilical cord: a viable source for mesenchymal stromal cells. Cytotherapy. 2015;17(1):18-24.
[33] YOUSEFIFARD M, NASIRINEZHAD F, SHARDI MANAHEJI H, et al. Human bone marrow-derived and umbilical cord-derived mesenchymal stem cells for alleviating neuropathic pain in a spinal cord injury model. Stem Cell Res Ther. 2016;7:36.
[34] CARON I, ROSSI F, PAPA S, et al. A new three dimensional biomimetic hydrogel to deliver factors secreted by human mesenchymal stem cells in spinal cord injury. Biomaterials. 2016;75:135-147.
[35] SOBACCHI C, PALAGANO E, VILLA A, et al. Soluble Factors on Stage to Direct Mesenchymal Stem Cells Fate. Front Bioeng Biotechnol. 2017;5:32.
[36] BAEZ-JURADO E, HIDALGO-LANUSSA O, BARRERA-BAILÓN B, et al. Secretome of Mesenchymal Stem Cells and Its Potential Protective Effects on Brain Pathologies. Mol Neurobiol. 2019;56(10):6902-6927.
[37] RYAN JM, BARRY FP, MURPHY JM, et al. Mesenchymal stem cells avoid allogeneic rejection. J Inflamm (Lond). 2005;2:8.
[38] LIANG X, DING Y, ZHANG Y, et al. Paracrine mechanisms of mesenchymal stem cell-based therapy: current status and perspectives. Cell Transplant. 2014;23(9):1045-1059.
[39] VIZOSO FJ, EIRO N, CID S, et al. Mesenchymal Stem Cell Secretome: Toward Cell-Free Therapeutic Strategies in Regenerative Medicine. Int J Mol Sci. 2017;18(9): E1852.
[40] SALEHI H, AMIRPOUR N, NIAPOUR A, et al. An Overview of Neural Differentiation Potential of Human Adipose Derived Stem Cells. Stem Cell Rev Rep. 2016;12(1):26-41.
[41] CHEN S, ZHANG W, WANG JM, et al. Differentiation of isolated human umbilical cord mesenchymal stem cells into neural stem cells. Int J Ophthalmol. 2016;9(1):41-47.
[42] BORKOWSKA P, KOWALSKA J, FILA-DANILOW A, et al. Affect of antidepressants on the in vitro differentiation of rat bone marrow mesenchymal stem cells into neuronal cells. Eur J Pharm Sci. 2015;73:81-87.
[43] LEITE C, SILVA NT, MENDES S, et al. Differentiation of human umbilical cord matrix mesenchymal stem cells into neural-like progenitor cells and maturation into an oligodendroglial-like lineage. PLoS One. 2014;9(10):e111059.
[44] HERNÁNDEZ R, JIMÉNEZ-LUNA C, PERALES-ADÁN J, et al. Differentiation of Human Mesenchymal Stem Cells towards Neuronal Lineage: Clinical Trials in Nervous System Disorders. Biomol Ther (Seoul). 2019 Oct 25. doi: 10.4062/biomolther.2019.065. [Epub ahead of print]
[45] ZENG X, MA YH, CHEN YF, et al. Autocrine fibronectin from differentiating mesenchymal stem cells induces the neurite elongation in vitro and promotes nerve fiber regeneration in transected spinal cord injury. J Biomed Mater Res A. 2016;104(8):1902-1911.
[46] RAGELLE H, NABA A, LARSON BL, et al. Comprehensive proteomic characterization of stem cell-derived extracellular matrices. Biomaterials. 2017;128:147-159.
[47] JOO S, YEON KIM J, LEE E, et al. Effects of ECM protein micropatterns on the migration and differentiation of adult neural stem cells. Sci Rep. 2015;5:13043.
[48] XIAO B, RAO F, GUO ZY, et al. Extracellular matrix from human umbilical cord-derived mesenchymal stem cells as a scaffold for peripheral nerve regeneration. Neural Regen Res. 2016;11(7): 1172-1179.
[49] GU Y, LI Z, HUANG J, et al. Application of marrow mesenchymal stem cell-derived extracellular matrix in peripheral nerve tissue engineering. J Tissue Eng Regen Med. 2017;11(8):2250-2260.
[50] KOČÍ Z, VÝBORNÝ K, DUBIŠOVÁ J, et al. Extracellular Matrix Hydrogel Derived from Human Umbilical Cord as a Scaffold for Neural Tissue Repair and Its Comparison with Extracellular Matrix from Porcine Tissues. Tissue Eng Part C Methods. 2017;23(6): 333-345.
[51] CHEN ZL, STRICKLAND S. Laminin gamma1 is critical for Schwann cell differentiation, axon myelination, and regeneration in the peripheral nerve. J Cell Biol. 2003;163(4):889-899.
[52] ROGERS SL, LETOURNEAU PC, PALM SL,et al. Neurite extension by peripheral and central nervous system neurons in response to substratum-bound fibronectin and laminin. Dev Biol. 1983;98(1): 212-220.
[53] HAGGERTY AE, MARLOW MM, OUDEGA M. Extracellular matrix components as therapeutics for spinal cord injury. Neurosci Lett. 2017;652:50-55.
[54] MENEZES K, DE MENEZES JR, NASCIMENTO MA, et al. Polylaminin, a polymeric form of laminin, promotes regeneration after spinal cord injury. FASEB J. 2010;24(11):4513-4522.
[55] OPPLIGER B, JOERGER-MESSERLI MS, SIMILLION C, et al. Mesenchymal stromal cells from umbilical cord Wharton's jelly trigger oligodendroglial differentiation in neural progenitor cells through cell-to-cell contact. Cytotherapy. 2017;19(7):829-838.
[56] GUO ZY, SUN X, XU XL, et al. Human umbilical cord mesenchymal stem cells promote peripheral nerve repair via paracrine mechanisms. Neural Regen Res. 2015;10(4):651-658.
[57] SUN L, WANG F, CHEN H, et al. Co-Transplantation of Human Umbilical Cord Mesenchymal Stem Cells and Human Neural Stem Cells Improves the Outcome in Rats with Spinal Cord Injury. Cell Transplant. 2019;28(7):893-906.
[58] HUANG JH, YIN XM, XU Y, et al. Systemic Administration of Exosomes Released from Mesenchymal Stromal Cells Attenuates Apoptosis, Inflammation, and Promotes Angiogenesis after Spinal Cord Injury in Rats. J Neurotrauma. 2017;34(24):3388-3396.
[59] SABELSTRÖM H, STENUDD M, RÉU P, et al. Resident neural stem cells restrict tissue damage and neuronal loss after spinal cord injury in mice. Science. 2013;342(6158):637-640.
[60] HOFER HR, TUAN RS. Secreted trophic factors of mesenchymal stem cells support neurovascular and musculoskeletal therapies. Stem Cell Res Ther. 2016;7(1):131.
[61] TEIXEIRA FG, CARVALHO MM, SOUSA N, et al. Mesenchymal stem cells secretome: a new paradigm for central nervous system regeneration? Cell Mol Life Sci. 2013;70(20):3871-3882.
[62] SABELSTRÖM H, STENUDD M, FRISÉN J. Neural stem cells in the adult spinal cord. Exp Neurol. 2014;260:44-49.
[63] CHEN SQ, CAI Q, SHEN YY, et al. Combined use of NGF/BDNF/bFGF promotes proliferation and differentiation of neural stem cells in vitro. Int J Dev Neurosci. 2014;38:74-78.
[64] 黄斐,马广文,尹宗生,等.脑源性神经营养因子缓释胶原凝胶支架对神经干细胞生长和分化的影响[J].安徽医科大学学报, 2014,49(5):586-590.
[65] RAO JS, ZHAO C, ZHANG A, et al. NT3-chitosan enables de novo regeneration and functional recovery in monkeys after spinal cord injury. Proc Natl Acad Sci U S A. 2018;115(24):E5595-E5604.
[66] ZHANG HY, WANG ZG, WU FZ, et al. Regulation of autophagy and ubiquitinated protein accumulation by bFGF promotes functional recovery and neural protection in a rat model of spinal cord injury. Mol Neurobiol. 2013;48(3):452-464.
[67] ROSICH K, HANNA BF, IBRAHIM RK, et al. The Effects of Glial Cell Line-Derived Neurotrophic Factor after Spinal Cord Injury. J Neurotrauma. 2017;34(24):3311-3325.
[68] KITAMURA K, NAGOSHI N, TSUJI O, et al. Application of Hepatocyte Growth Factor for Acute Spinal Cord Injury: The Road from Basic Studies to Human Treatment. Int J Mol Sci. 2019;20(5): E1054.
[69] TUSZYNSKI MH, GRILL R, JONES LL, et al. NT-3 gene delivery elicits growth of chronically injured corticospinal axons and modestly improves functional deficits after chronic scar resection. Exp Neurol. 2003;181(1):47-56.
[70] ZANOTTI L, ANGIONI R, CALÌ B, et al. Mouse mesenchymal stem cells inhibit high endothelial cell activation and lymphocyte homing to lymph nodes by releasing TIMP-1. Leukemia. 2016;30(5):1143-1154.
[71] ZHAO J, CHEN N, SHEN N, et al. Transplantation of human umbilical cord blood mesenchymal stem cells to treat a rat model of traumatic brain injury. Neural Regen Res. 2012;7(10):741-748.
[72] RECORD M, SUBRA C, SILVENTE-POIROT S, et al. Exosomes as intercellular signalosomes and pharmacological effectors. Biochem Pharmacol. 2011;81(10):1171-1182.
[73] YAGHOUBI Y, MOVASSAGHPOUR A, ZAMANI M, et al. Human umbilical cord mesenchymal stem cells derived-exosomes in diseases treatment. Life Sci. 2019;233:116733.
[74] SUN G, LI G, LI D, et al. hucMSC derived exosomes promote functional recovery in spinal cord injury mice via attenuating inflammation. Mater Sci Eng C Mater Biol Appl. 2018;89:194-204.
[75] XIN H, LI Y, BULLER B, et al. Exosome-mediated transfer of miR-133b from multipotent mesenchymal stromal cells to neural cells contributes to neurite outgrowth. Stem Cells. 2012;30(7):1556-1564.
[76] YUAN X, WU Q, WANG P, et al. Exosomes Derived From Pericytes Improve Microcirculation and Protect Blood-Spinal Cord Barrier After Spinal Cord Injury in Mice. Front Neurosci. 2019;13:319.
[77] BOIDO M, GARBOSSA D, VERCELLI A. Early graft of neural precursors in spinal cord compression reduces glial cyst and improves function. J Neurosurg Spine. 2011;15(1):97-106.
[78] DAI G, LIU X, ZHANG Z, et al. Transplantation of autologous bone marrow mesenchymal stem cells in the treatment of complete and chronic cervical spinal cord injury. Brain Res. 2013;1533:73-79.
[79] EL-KHEIR WA, GABR H, AWAD MR, et al. Autologous bone marrow-derived cell therapy combined with physical therapy induces functional improvement in chronic spinal cord injury patients. Cell Transplant. 2014;23(6):729-745.
[80] GEFFNER LF, SANTACRUZ P, IZURIETA M, et al. Administration of autologous bone marrow stem cells into spinal cord injury patients via multiple routes is safe and improves their quality of life: comprehensive case studies. Cell Transplant. 2008;17(12):1277-1293.
[81] CORCIONE A, BENVENUTO F, FERRETTI E, et al. Human mesenchymal stem cells modulate B-cell functions. Blood. 2006; 107(1):367-372.
[82] DENG J, PETERSEN BE, STEINDLER DA, et al. Mesenchymal stem cells spontaneously express neural proteins in culture and are neurogenic after transplantation. Stem Cells. 2006;24(4):1054-1064.
[83] LU P, BLESCH A, TUSZYNSKI MH. Induction of bone marrow stromal cells to neurons: differentiation, transdifferentiation, or artifact? J Neurosci Res. 2004;77(2):174-191.
[84] CORTÉS-MEDINA LV, PASANTES-MORALES H, AGUILERA- CASTREJON A, et al. Neuronal Transdifferentiation Potential of Human Mesenchymal Stem Cells from Neonatal and Adult Sources by a Small Molecule Cocktail. Stem Cells Int. 2019;2019:7627148.
[85] PARK WS, AHN SY, SUNG SI, et al. Strategies to enhance paracrine potency of transplanted mesenchymal stem cells in intractable neonatal disorders. Pediatr Res. 2018;83(1-2):214-222.
|