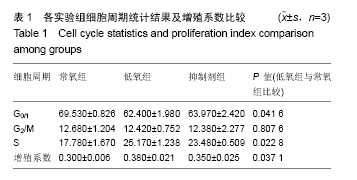
| [1] Fayyad-Kazan H, Faour WH, Badran B, et al. The immunomodulatory properties of human bone marrow-derived mesenchymal stromal cells are defined according to multiple immunobiological criteria. Inflamm Res. 2016;65(6):501-510.[2] Ge L, Jiang M, Duan D, et al. Secretome of Olfactory Mucosa Mesenchymal Stem Cell, a Multiple Potential Stem Cell. Stem Cells Int. 2016;2016:1243659.[3] 葛丽特,卓毅,段答,等.人嗅黏膜间充质干细胞的生物学特性[J]. 中南大学学报:医学版, 2015, 40(1): 53-58.[4] Jiang Y, Jahagirdar BN, Reinhardt RL, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002;418(6893):41-49.[5] Rodesch F, Simon P, Donner C, et al. Oxygen measurements in endometrial and trophoblastic tissues during early pregnancy. Obstet Gynecol. 1992;80(2):283-285.[6] Ren H, Cao Y, Zhao Q, et al. Proliferation and differentiation of bone marrow stromal cells under hypoxic conditions. Biochem Biophys Res Commun. 2006;347(1):12-21.[7] Forristal CE, Wright KL, Hanley NA, et al. Hypoxia inducible factors regulate pluripotency and proliferation in human embryonic stem cells cultured at reduced oxygen tensions. Reproduction. 2010;139(1):85-97.[8] Prasad SM, Czepiel M, Cetinkaya C, et al. Continuous hypoxic culturing maintains activation of Notch and allows long-term propagation of human embryonic stem cells without spontaneous differentiation. Cell Prolif. 2009;42(1):63-74.[9] Bas E, Van De Water TR, Lumbreras V, et al. Adult human nasal mesenchymal-like stem cells restore cochlear spiral ganglion neurons after experimental lesion. Stem Cells Dev. 2014;23(5):502-514.[10] Shafiee A, Kabiri M, Ahmadbeigi N, et al. Nasal septum-derived multipotent progenitors: a potent source for stem cell-based regenerative medicine. Stem Cells Dev. 2011;20(12):2077-2091.[11] Murrell W, Féron F, Wetzig A, et al. Multipotent stem cells from adult olfactory mucosa. Dev Dyn. 2005;233(2):496-515.[12] Black IB, Woodbury D. Adult rat and human bone marrow stromal stem cells differentiate into neurons. Blood Cells Mol Dis. 2001;27(3):632-636.[13] Lee JH, Kemp DM. Human adipose-derived stem cells display myogenic potential and perturbed function in hypoxic conditions. Biochem Biophys Res Commun. 2006;341(3): 882-888.[14] Wang Y, Yang J, Li H, et al. Hypoxia promotes dopaminergic differentiation of mesenchymal stem cells and shows benefits for transplantation in a rat model of Parkinson's disease. PLoS One. 2013;8(1):e54296.[15] Nekanti U, Dastidar S, Venugopal P, et al. Increased proliferation and analysis of differential gene expression in human Wharton's jelly-derived mesenchymal stromal cells under hypoxia. Int J Biol Sci. 2010;6(5):499-512.[16] Semenza GL, Wang GL. A nuclear factor induced by hypoxia via de novo protein synthesis binds to the human erythropoietin gene enhancer at a site required for transcriptional activation. Mol Cell Biol. 1992;12(12): 5447-5454.[17] Yu Z, Tang L, Chen L, et al. Role for HIF-1α and Downstream Pathways in Regulating Neuronal Injury after Intracerebral Hemorrhage in Diabetes. Cell Physiol Biochem. 2015;37(1): 67-76.[18] Wang L, Jiang M, Duan D, et al. Hyperthermia-conditioned OECs serum-free-conditioned medium induce NSC differentiation into neuron more efficiently by the upregulation of HIF-1 alpha and binding activity. Transplantation. 2014; 97(12):1225-1232.[19] Du F, Zhu L, Qian ZM, et al. Hyperthermic preconditioning protects astrocytes from ischemia/reperfusion injury by up-regulation of HIF-1 alpha expression and binding activity. Biochim Biophys Acta. 2010;1802(11):1048-1053.[20] Das R, Jahr H, van Osch GJ, et al. The role of hypoxia in bone marrow-derived mesenchymal stem cells: considerations for regenerative medicine approaches. Tissue Eng Part B Rev. 2010;16(2):159-168.[21] Fehrer C, Brunauer R, Laschober G, et al. Reduced oxygen tension attenuates differentiation capacity of human mesenchymal stem cells and prolongs their lifespan. Aging Cell. 2007;6(6):745-757.[22] Tsai CC, Chen YJ, Yew TL, et al. Hypoxia inhibits senescence and maintains mesenchymal stem cell properties through down-regulation of E2A-p21 by HIF-TWIST. Blood. 2011; 117(2):459-469.[23] Sasabe M, Machida Y. Signaling Pathway that Controls Plant Cytokinesis. Enzymes. 2014;35:145-165.[24] Cai TY, Zhu W, Chen XS, et al. Fibroblast growth factor 2 induces mesenchymal stem cells to differentiate into tenocytes through the MAPK pathway. Mol Med Rep. 2013;8(5):1323-1328.[25] Huang F, Fang ZF, Hu XQ, et al. Overexpression of miR-126 promotes the differentiation of mesenchymal stem cells toward endothelial cells via activation of PI3K/Akt and MAPK/ERK pathways and release of paracrine factors. Biol Chem. 2013;394(9):1223-1233.[26] Gu W, Song L, Li XM, et al. Mesenchymal stem cells alleviate airway inflammation and emphysema in COPD through down-regulation of cyclooxygenase-2 via p38 and ERK MAPK pathways. Sci Rep. 2015;5:8733.[27] Mei Y, Bian C, Li J, et al. miR-21 modulates the ERK-MAPK signaling pathway by regulating SPRY2 expression during human mesenchymal stem cell differentiation. J Cell Biochem. 2013 Jun;114(6):1374-1384.[28] Tsai CC, Su PF, Huang YF, et al. Oct4 and Nanog directly regulate Dnmt1 to maintain self-renewal and undifferentiated state in mesenchymal stem cells. Mol Cell. 2012;47(2): 169-182.[29] Chen TL, Zhu GL, Wang JA, et al. Apoptosis of bone marrow mesenchymal stem cells caused by hypoxia/reoxygenation via multiple pathways. Int J Clin Exp Med. 2014;7(12): 4686-4697.[30] Cao W, Guo XW, Chen K, et al. Inhibition of hypoxia and serum deprivation-induced apoptosis by salvianolic acid in rat mesenchymal stem cells. J Tradit Chin Med. 2012;32(2): 222-228.[31] Zhu W, Chen J, Cong X, et al. Hypoxia and serum deprivation-induced apoptosis in mesenchymal stem cells. Stem Cells. 2006;24(2):416-425. |
.jpg)

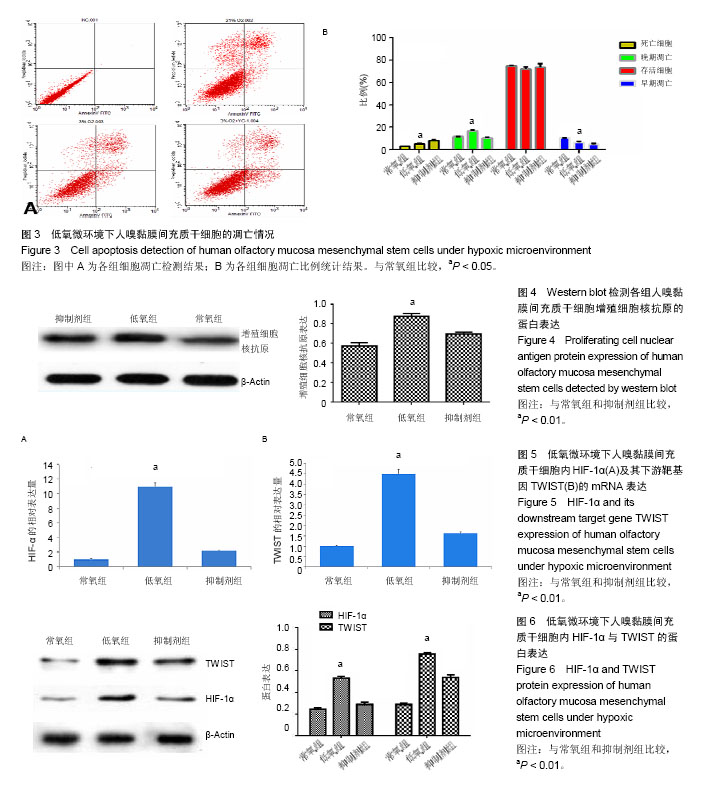
.jpg)