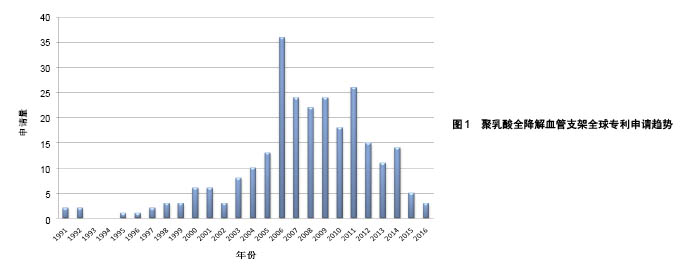
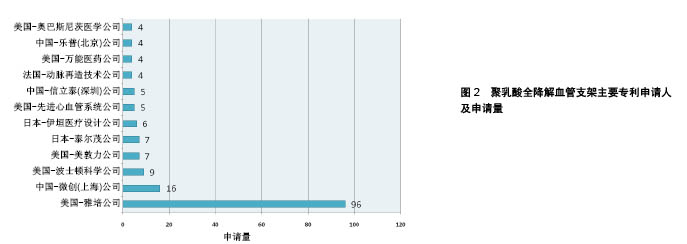
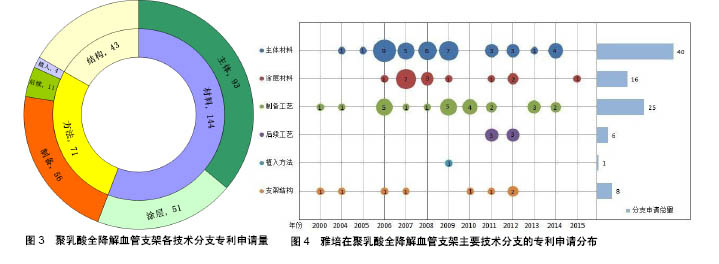
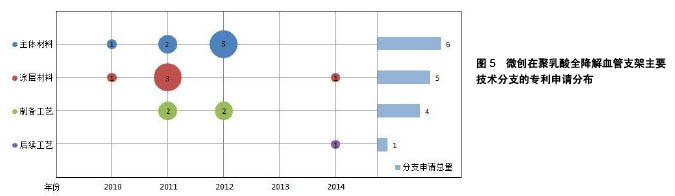
| [1] 世界卫生组织官方网站http://www.who.int/mediacentre/factsheets/fs317/zh[2] Lloyd-Jones D,Adams RJ,Brown TM,et al.Heart disease and stroke statistics-2009 update: a report from the American Heart Association statistics committee and stroke statistics subcommittee. Circulation. 2009;119:480-486.[3] 甄珍,奚廷斐,郑玉峰.全降解冠脉支架研究进展[J].新材料产业, 2011, 13(8):8-12.[4] Serruys PW,Luijten HE,Beatt KJ,et al.Incidence of restenosis after successful coronary angioplasty: A time-related phenomenon: A quantitative angiographic study in 342 consecutive patients at 1, 2, 3, and 4 months. Circulation. 1988;77:361-371.[5] Gruentzig AR,King SB,Schlumpf M,et al.Longterm follow-up after percutaneous transluminal coronary angioplasty: The early Zurich experience.N Engl J Med.1987;316:1127-1132.[6] Serruys PW,de Jaegere P,Kiemeneij F,et al.A comparison of balloon-expandable-stent implantation with balloon angioplasty in patients with coronary artery disease: Benestent Study Group.N Engl J Med.1994;331:489-495.[7] Martin DM,Boyle FJ.Drug-eluting stents for coronary artery disease: A review.Med Eng Phys. 2011;33(2):148-163.[8] Lemos PA,Serruys PW,van Domburg RT,et al.Unrestricted utilization of sirolimus-eluting stents compared with conventional bare stent implantation in the “real world”: The Rapamycin-Eluting stent Evaluated At Rotterdam Cardiology Hospital (RESEARCH) registry. Circulation.2004;109:190-195.[9] Di Mario C,Gil R,Camenzind E,et al.Quantitative assessment with intracoronary ultrasound of the mechanisms of restenosis after percutaneous transluminal coronary angioplasty and directional coronary atherectomy.Am J Cardiol.1995;75:772-777.[10] Yamawaki T,Shimokawa H,Kozai T,et al.Intramural delivery of a specific tyrosine kinase inhibitor with biodegradable stent suppresses the restenotic changes of the coronary artery in pigs in vivo.J Am Coll Cardiol.1998;32:780-786.[11] Eggebrecht H,Rodermann J,Hunold P,et al.Images in cardiovascular medicine: Novel magnetic resonance-compatible coronary stent: The absorbable magnesiumalloy stent. Circulation. 2005;112: e303-e304.[12] Tamai H,Igaki K,Kyo E,et al.Initial and 6-month results of biodegradable poly-l-lactic acid coronary stents in humans. Circulation. 2000;102:399-404.[13] Vogt F,Stein A,Rettemeier G,et al.Long-term assessment of a novel biodegradable paclitaxel-eluting coronary polylactide stent.Eur Heart J. 2004;25:1330-1340.[14] Ormiston JA,Webster MW,Armstrong G.First-in-human implantation of a fully bioabsorbable drug-eluting stent:the BVS poly-L-lactic acid everolimus-eluting coronary stent.Catheter Cardiovasc Interv. 2007; 69:128-131[15] ABBOTT CARDIOVASCULAR SYSTEMS INC.Coating stent e.g. self-expandable stents, by applying composition including polymer component and solvent to stent substrate, and exposing polymer component to temperature equal to/greater than glass transition temperature of component:美国,US8632845B2[16] ABBOTT LAB VASCULAR ENTERPRISES LTD.Apparatus, useful for stenting, comprises stent (having proximal and distal ends and a lumen extending between them, and tubular body) and a biocompatible material layer attached to a portion of stent between the proximal and distal ends:美国,EP2266502A1[17] ABBOTT CARDIOVASCULAR SYSTEMS INC, Implantable medical device such as stent for treating diseases associated with lumen in mammals e.g. atherosclerotic stenosis, has structural element containing composite having polymer nanoparticles dispersed within polymer matrix:美国,WO2008002479A2[18] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent, e.g. for maintaining vascular patency and/or drug delivery, has structural unit including continuous phase containing matrix polymer, and discrete phases containing modifier polymer with discrete phase segments:美国,US2008081063A1[19] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent useful in the treatment of artherosclerosis comprises a body consisting of composite containing a polymer, and radiopaque metallic particles mixed or dispersed within the polymer:美国,WO2008016696A2[20] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent comprises stent body containing structural elements, each element contains luminal surface, abluminal surface and sidewall surface, prohealing coating, and antiproliferative coating, where the body is made of bioabsorbable material:美国,US8709071B1[21] ABBOTT CARDIOVASCULAR SYSTEMS INC.Polymeric stent for being implanted in bodily lumen for treating polymer surface, has functional group miscible or compatible with coating polymer selected as poly-L-lactide-co-glycolide, polypropylene glycol, or polyethylene glycol:美国,US2009148591A1[22] ABBOTT CARDIOVASCULAR SYSTEMS INC.Method for fabricating implantable medical device e.g. stent from deformed polymer tube, involves treating portion of tube with solvent, radially and axially deforming tube and fabricating stent from deformed and treated tube:美国,US2014265060A1[23] ABBOTT CARDIOVASCULAR SYSTEMS INC.Polymeric tubular implantable stent e.g. self-expandable stent for use during treatment of atherosclerotic stenosis in blood vessels, has tube annealed at temperature in temperature range of above glass transition temperature of polymer:美国,US2014084515A1[24] 上海微创医疗器械(集团)有限公司.一种生物可降解支架:中国, CN102371006A[P].2012-03-14.[25] 上海微创医疗器械(集团)有限公司.一种生物可降解支架及其制备工艺:中国,CN102327652A[P].2012-01-25.[26] 上海微创医疗器械(集团)有限公司.一种可降解聚酯支架及其制备工艺:中国,CN103877624A[P].2014-06-25.[27] 上海微创医疗器械(集团)有限公司.一种生物可降解聚合物支架的制备工艺:中国,CN103876869A[P].2014-06-25.[28] SCIMED LIFE SYSTEMS INC IN.Stent, useful for inserting into body lumen such as blood vessel, has tubular main body having biodegradable matrix containing collagen IV and laminin and biodegradable strengthening material:美国,US2002099434 A1[29] UNIV TEXAS.Biodegradable polymer fiber scaffold implants with controlled release of therapeutic agents, promote cell migration use for drug delivery, stents, arteriogenesis, bone fractures, muscle grafts, wound dressings, and optic disorders:美国,WO0110421 A1[30] ABBOTT CARDIOVASCULAR SYSTEMS INC.Implantable medical device, e.g. stent, comprises structural element including continuous phase comprised of first polymer, and discrete phase comprised of second polymer that has discrete phase segments and anchor segments:美国,US2007231365 A1[31] ABBOTT CARDIOVASCULAR SYSTEMS INC.Implantable medical device e.g. stent fabricated from polymer blend comprises biodegradable matrix polymer blended with biodegradable star-block copolymers having three arms, where the arms include inner segments and outer segments:美国,US2014046006 A1[32] BIOSENSORS INT GROUP.Biodegradable polymer stent such as bile duct stent useful in blood vessels e.g. aorta, comprises an aliphatic polyester polymer, and a phosphate salt including phosphate buffer salt, citrate buffer salt and sodium chloride:百慕大,US2011301259 A1[33] XTENT INC.Implant, useful e.g. in tissue, comprises structure (having material property at normal body temperature) adapted for implantation; and particles (which are adapted to convert incident radiation into heat energy) dispersed in structure:美国,US2008199510 A1[34] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent making method by forming tube from melt processed blend of (L-lactide) and poly(D-lactic acid) comprising stereocomplex crystallites by quenching to temperature below glass transition temperature of poly (L-lactide):美国,WO2010019478 A1[35] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent for treating diseased section of blood vessel comprises specific amount of L-lactide monomer mixed/dispersed/dissolved within polymeric scaffolding having poly(L-lactide) with specific crystallinity and number average molecular weight:美国,WO2011011242 A2[36] ABBOTT CARDIOVASCULAR SYSTEMS INC.Fabricating bioresorbable stent scaffold i.e. endoprosthesis stent, to treat atherosclerotic stenosis in blood vessels or heart valve, by providing a tube made of a bioresorbable polymer, exposing into solvent, and fabricating scaffold:美国,US2014030422 A1[37] ABBOTT CARDIOVASCULAR SYSTEMS INC.Medical device implantable within peripheral vessel of body to deliver anti-restenosis drug to treat peripheral vascular disease, comprises cylindrical radially expandable body made of polymer formulation and anti-restenosis drug:美国,US2014277398 A1[38] SAHAJANAND MEDICAL TECHNOLOGIES PVT LTD IN.Composition, useful for treating e.g. restenosis, comprises first polymer having pores; nanoparticles (comprising second polymer and active agent) dispersed within pores; and heparin covalently bonded to the polymer:印度, US2007212388A1[39] BOSTON SCI SCIMED INC.Block or graft copolymer used in coating of implantable medical device, e.g. stent, comprises first block including poly(lactide-glycolide) copolymer and second block comprising, e.g. polylactide homopolymer:美国,WO2009149277A1[40] ABBOTT CARDIOVASCULAR SYSTEMS INC.Implantable medical device useful as e.g. self-expandable stents, balloon-expandable stents, stent-grafts, and implantable cardiac pacemakers, comprises structural element including a bioceramic/copolymer composite:美国,US2007282434A1[41] XTENT INC.Implant, useful e.g. in tissue, comprises structure (having material property at normal body temperature) adapted for implantation; and particles (which are adapted to convert incident radiation into heat energy) dispersed in structure:美国, US2008199510A1[42] ARTERIAL REMODELING TECHNOLOGIES INC.Reduction of sharp surface and/or irregularity of polymeric stent, involves immersing polymeric stent into bath containing solvent and biodegradable polymer:美国,US2007050018A1[43] ABBOTT CARDIOVASCULAR SYSTEMS INC.Fabricating stent useful for bodily lumen involves forming a biodegradable polymer with a polymerization initiated with an acid containing group, and fabricating the stent from the formed polymer which includes acid group from the initiator:美国,WO2008011048A2[44] ABBOTT CARDIOVASCULAR SYSTEMS INC.Implantable medical device e.g. vascular stent comprises blend of high molecular weight polyester and low molecular weight polyester containing the blend and acidic moiety:美国,US2009324670A1[45] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent useful for delivering drug or therapeutic agent for treating atherosclerosis, comprises scaffolding made of poly(L-lactide) and several particles dispersed throughout scaffolding:美国,US2012290071A1[46] TERUMO CORP.Stent for detaining in constricted/closed tubular cavities e.g. blood vessels and useful in treating various diseases, is formed using fiber composition containing biodegradable polymer and carbon fiber having preset length:日本,JP2005168646A[47] ADVANCED CARDIOVASCULAR SYSTEM.Medical article for use as medical implant comprises implant comprising polymeric material that comprises plasticizing agent capable of increasing strain-to-failure in the polymeric material while placing the implant in a subject:美国, US2006058868A1[48] ADVANCED CARDIOVASCULAR SYSTEM.Fabrication of implantable medical device by forming suspension solution including fluid, matrix polymer, and bioceramic particles, forming composite mixture, and mixing composite mixture with modifier polymer and additional matrix polymer:美国,US2011057339A1[49] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent making method by forming tube from melt processed blend of (L- lactide) and poly(D-lactic acid) comprising stereocomplex crystallites by quenching to temperature below glass transition temperature of poly (L-lactide):美国,JP2011530437A[50] TERUMO CORP.In-vivo absorptive stent useful for preventing reocclusion of lesion site in e.g. blood vessel and trachea, contains mixture of in-vivo absorptive aliphatic polyester and aromatic compound having 1 or more aromatic rings:日本,WO2011096241A1 |
.jpg)

.jpg)