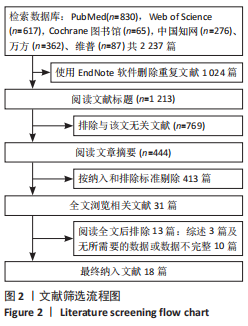
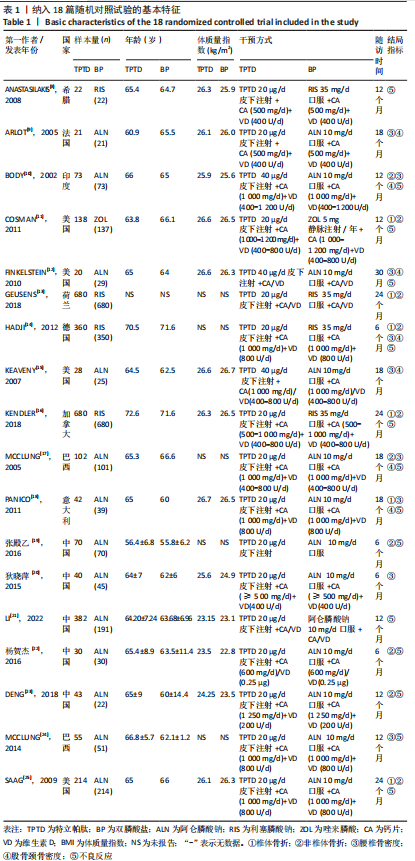
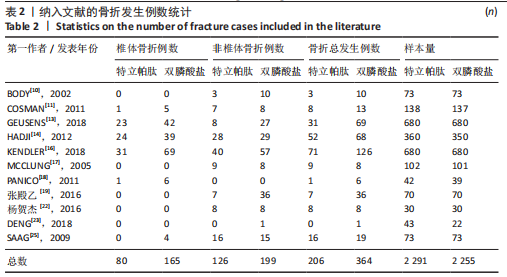
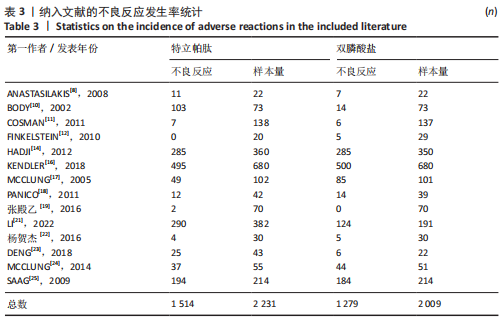
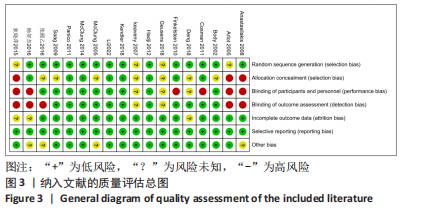
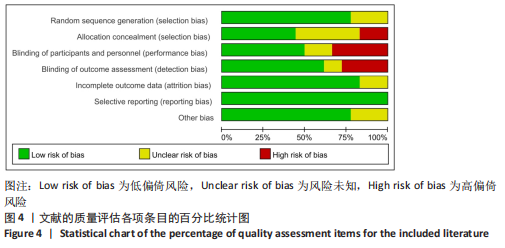
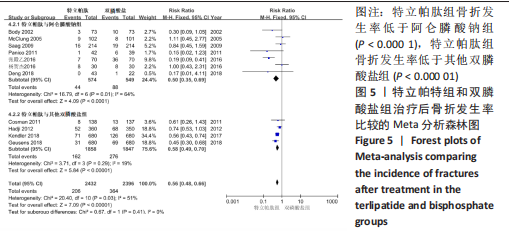
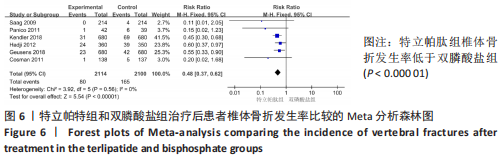
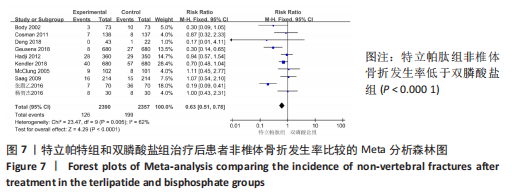
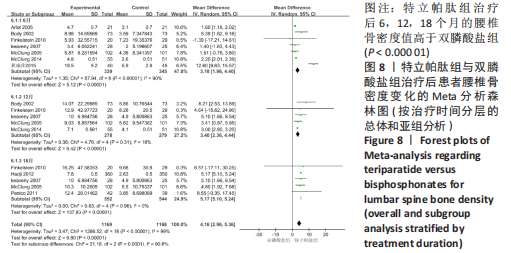
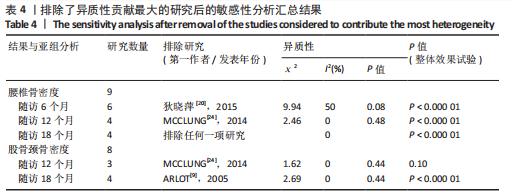
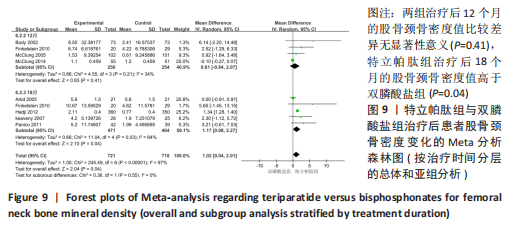
[1] 马远征,王以朋,刘强,等.中国老年骨质疏松症诊疗指南(2018)[J].中国骨质疏松杂志,2018, 24(12):1541-1567.
[2] HADJI P, JACOB L, KOSTEV K. Gender- and age-related treatment compliance in patients with osteoporosis in Germany. Patient Prefer Adher. 2016;10:2379-2385.
[3] SI L, WINZENBERG TM, JIANG Q, et al. Projection of osteoporosis-related fractures and costs in China: 2010-2050. Osteoporos Int. 2015;26(7):1929-1937.
[4] 夏维波,章振林,林华,等.原发性骨质疏松症诊疗指南(2017)[J].中国骨质疏松杂志,2019, 25(3):281-309.
[5] MCCONNELL M, SHIEH A. Polypharmacy in osteoporosis treatment. Clin Geriatr Med. 2022;38(4):715-726.
[6] CHANG B, QUAN Q, LI Y, et al. Treatment of osteoporosis, with a focus on 2 monoclonal antibodies. Med Sci Monit. 2018;24:8758-8766.
[7] COLLINGE C, FAVELA J. Use of teriparatide in osteoporotic fracture patients. Injury. 2016;47: S36-S38.
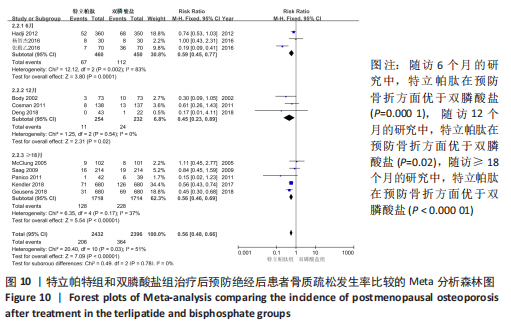
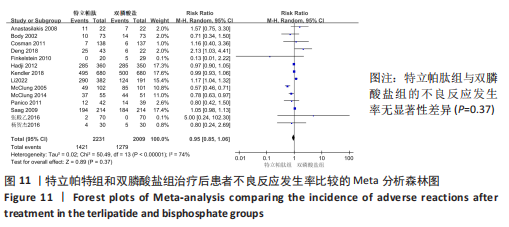
[8] ANASTASILAKIS AD, GOULIS DG, POLYZOS SA, et al. Head-to-head comparison of risedronate vs. teriparatide on bone turnover markers in women with postmenopausal osteoporosis: a randomised trial. Int J Clin Pract. 2008;62(6):919-924.
[9] ARLOT M, MEUNIER PJ, BOIVIN G, et al. Differential effects of teriparatide and alendronate on bone remodeling in postmenopausal women assessed by histomorphometric parameters. J Bone Miner Res. 2005;20(7):1244-1253.
[10] BODY JJ, GAICH GA, SCHEELE WH, et al. A Randomized double-blind trial to compare the efficacy of teriparatide [recombinant human parathyroid hormone (1-34)] with alendronate in postmenopausal women with osteoporosis. J Clin Endocrinol Metab. 2002;87(10):4528-4535.
[11) COSMAN F, ERIKSEN EF, RECKNOR C, et al. Effects of intravenous zoledronic acid plus subcutaneous teriparatide [rhPTH(1-34)] in postmenopausal osteoporosis. J Bone Miner Res. 2011;26(3):503-511.
[12] FINKELSTEIN JS, WYLAND JJ, LEE H, et al. Effects of teriparatide,alendronate, or both in women with postmenopausal osteoporosis. J Clin Endocrinol Metab. 2010;95(4):1838-1845.
[13] GEUSENS P, MARIN F, KENDLER DL, et al. Effects of teriparatide compared with risedronate on the risk of fractures in subgroups of postmenopausal women with severe osteoporosis: the VERO trial. J Bone Miner Res. 2018;33(5):783-794.
[14] HADJI P, ZANCHETTA JR, RUSSO L, et al. The effect of teriparatide compared with risedronate on reduction of back pain in postmenopausal women with osteoporotic vertebral fractures. Osteoporos Int. 2012;23(8):2141-2150.
[15] KEAVENY TM, DONLEY DW, HOFFMANN PF, et al. Effects of teriparatide and alendronate on vertebral strength as assessed by finite element modeling of QCT scans in women with osteoporosis. J Bone Miner Res. 2007;22(1):149-157.
[16] KENDLER DL, MARIN F, ZERBINI C AF, et al. Effects of teriparatide and risedronate on new fractures in post-menopausal women with severe osteoporosis (VERO): a multicentre, double-blind, double-dummy,randomised controlled trial. Lancet. 2018; 391(10117):230-240.
[17] MCCLUNG MR, SAN MARTIN J, MILLER PD, et al. Opposite bone remodeling effects of teriparatide and alendronate in increasing bone mass. Arch Intern Med. 2005;165(15):1762-1768.
[18] PANICO A, LUPOLI GA, MARCIELLO F, et al. Teriparatide vs. Alendronate as a treatment for osteoporosis: changes in biochemical markers of bone turnover, BMD and quality of life. Med Sci Monit. 2011;17(8):CR442-CR448.
[19] 张殿乙,李雅男,张楠,等.特立帕肽联合阿仑膦酸钠治疗绝经后骨质疏松症的临床研究[J].现代药物与临床,2016,31(8):1231-1235.
[20] 狄晓萍,狄维鹏,任婷,等.特立帕肽与阿仑膦酸钠对绝经后骨质疏松患者的疗效对比[J].宁夏医学杂志,2015,37(7):648-650.
[21) LI M, ZHANG Z, XUE Q, et al. Efficacy of generic teriparatide and alendronate in Chinese postmenopausal women with osteoporosis: a prospective study. Arch Osteoporos. 2022;17(1):103.
[22] 杨贺杰,周辉,施建勤,等.特立帕肽与阿仑膦酸钠治疗绝经后骨质疏松症的效果比较[J].浙江临床医学,2016,18(5):912-913.
[23] DENG J, FENG ZP, LI Y, et al. Efficacy and safety of recombinant human parathyroid hormone (1-34) are similar to those of alendronate in the treatment of postmenopausal osteoporosis. Medicine. 2018; 97(47):e13341.
[24] MCCLUNG MR, GRAUER A, BOONEN S, et al. Romosozumab in postmenopausal women with low bone mineral density. N Engl J Med. 2014;370(5):412-220.
[25] SAAG KG, ZANCHETTA JR, DEVOGELAER JP, et al. Effects of teriparatide versus alendronate for treating glucocorticoid-induced osteoporosis thirty-six-month results of a randomized,double-blind,controlled trial. Arthritis Rheum. 2009; 60(11):3346-3355.
[26] CAMACHO PM, PETAK SM, BINKLEY N, et al. American association of clinical endocrinologists/american college of endocrinology clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis-2020 update. Endocr Pract. 2020; 26(Suppl 1):1-46.
[27] 季逸超,张林林,杨惠林,等.特立帕肽与唑来膦酸治疗绝经后骨质疏松性椎体骨折的疗效对比研究[J].中华骨与关节外科杂志,2021, 14(12):1011-1015.
[28] TAKADA J, DINAVAHI R, MIYAUCHI A, et al. Relationship between P1NP, a biochemical marker of bone turnover, and bone mineral density in patients transitioned from alendronate to romosozumab or teriparatide: a post hoc analysis of the STRUCTURE trial. J Bone Miner Metab. 2020;38(3):310-315.
[29] MCCARTHY J, DAVIS A. Diagnosis and management of vertebral compression fractures. Am Fam Physician. 2016,94(1):44-50.
[30] YOLCU Y,ALVI M,WANDERMAN N, et al. Effect of teriparatide use on bone mineral density and spinal fusion: a narrative review of animal models. Int J Neurosci. 2019;129(8):814-820.
[31) CHEN Q,GUO M,MA X M, et al. Adherence to teriparatide treatment and risk of fracture: a systematic review and meta-analysis. Horm Metab Res. 2019;51(12):785-791.
[32] SATO C, MIYAKOSHI N, KASUKAWA Y, et al. Teriparatide and exercise improve bone,skeletal muscle,and fat parameters in ovariectomized and tail-suspended rats. J Bone Miner Metab. 2021;39(3):385-395.
[33] LANGDAHL BL, SILVERMAN S, FUJIWARA S, et al. Real-world effectiveness of teriparatide on fracture reduction in patients with osteoporosis and comorbidities or risk factors for fractures:Integrated analysis of 4 prospective observational studies. Bone. 2018;116:58-66.
[34] 包呼和,孙官文,王剑,等.特立帕肽和双膦酸盐序贯治疗高龄骨质疏松症患者的疗效观察[J].实用临床医药杂志,2021,25(16):95-98.
[35] MIYAUCHI A, MATSUMOTO T, SUGIMOTO T, et al. Effects of teriparatide on bone mineral density and bone turnover markers in Japanese subjects with osteoporosis at high risk of fracture in a 24-month clinical study: 12-month, randomized, placebo-controlled, double-blind and 12-month open-label phases. Bone. 2010;47(3):493-502.
[36] COHEN A, SHIAU S, NAIR N, et al. Effect of teriparatide on bone remodeling and density in premenopausal idiopathic osteoporosis: a phase II trial. J Clin Endocrinol Metab. 2020;105(10):e3540-e3556.
[37] HASSAN N, GREGSON CL, TOBIAS JH. Anabolic treatments for osteoporosis in postmenopausal women. Fac Rev. 2021;10:44.
[38] DIEZ-PEREZ A, MARIN F, ERIKSEN EF, et al. Effects of teriparatide on hip and upper limb fractures in patients with osteoporosis: a systematic review and meta-analysis. Bone. 2019;120:1-8.
[39] SAAG KG, SHANE E, BOONEN S, et al. Teriparatide or alendronate in glucocorticoid-induced osteoporosis. N Engl J Med. 2007;357(20):2028-2039.
[40] LINDSAY R, KREGE JH, MARIN F, et al. Teriparatide for osteoporosis:importance of the full course. Osteoporos Int. 2016;27(8):2395-2410.
[41) LANGDAHL B. Treatment of postmenopausal osteoporosis with bone-forming and antiresorptive treatments:Combined and sequential approaches. Bone. 2020;139:115516.
[42] SILVERMAN S, MILLER P, SEBBA A, et al. The direct assessment of nonvertebral fractures in community experience (DANCE) study: 2-year nonvertebral fragility fracture results. Osteoporos Int. 2013;24(8):2309-2317. |