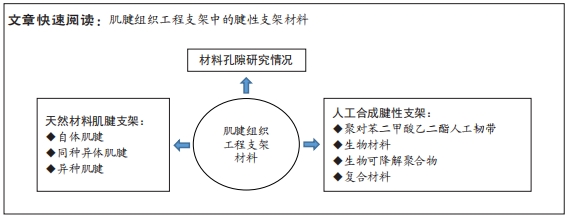
[1] PATEL S, CALDWELL JM, DOTY SB, et al. Integrating Soft and Hard Tissues via Interface Tissue Engineering. J Orthop Res. 2018;36(4):1069-1077.
[2] WHITLOCK PW, SMITH TL, POEHLING GG, et al. A naturally derived cytocompatible, and architecturally optimized scaffold for tendon and ligament regeneration. Biomaterials. 2007;28(29):4321-4329.
[3] HINSENKAMP M, MUYLLE L, EASTLUND T, et al. Adverse reactions and events related to musculoskeletal allografts: Reviewed by the World Health Organisation Project NOTIFY. Int Orthop. 2012;36:633-641.
[4] MALLICK TK, MOSQUERA A, ZINDERMAN CE, et al. Reported infections after human tissue transplantation before and after new Food and Drug Administration (FDA) regulations, United States, 2001 through June, 2010. Cell Tissue Bank. 2012;13:259-267.
[5] SEYLER TM, BRACEY DN, PLATE JF, et al. The Development of a Xenograft-Derived Scaffold for Tendon and Ligament Reconstruction Using a Decellularization and Oxidation Protocol. Arthroscopy. 2017;33(2):374-386.
[6] CHEN T, JIANG J, CHEN S. Status and headway of the clinical application of artificial ligaments. Asia Pac J Sports Med Arthrosc Rehabil Technol. 2015;2(1):15-26.
[7] LIM WL, LIAU LL, NG MH, et al. Current Progress in Tendon and Ligament Tissue Engineering. Tissue Eng Regen Med. 2019;16(6):549-571.
[8] CHEN B, WANG B, ZHANG WJ, et al. In vivo tendon engineering with skeletal muscle derived cells in a mouse model. Biomaterials. 2012;33(26):6086-6097.
[9] WHITLOCK PW, SEYLER TM, PARKS GD, et al. A novel process for optimizing musculoskeletal allograft tissue to improve safety, ultrastructural properties, and cell infiltration. J Bone Joint Surg Am. 2012;94(16):1458-1467.
[10] OZASA Y, AMADIO PC, THORESON AR, et al. Repopulation of intrasynovial flexor tendon allograft with bone marrow stromal cells: an ex vivo model. Tissue Eng Part A. 2014;20(3-4):566-574.
[11] LORD BR, EL-DAOU H, SABNIS M, et al. Biomechanical comparison of graft structures in anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2017;25(2):559-568.
[12] MA T, LI Y,YANG ST, et al. Effects of pore size in 3-D fibrous matrix on human trophoblast tissue developmen. Biotechnol Bioeng. 2000;70(6):606-618.
[13] ITOI Y, TAKATORI M, HYAKUSOKU H, et al. Comparison of readily available scaffolds for adipose tissue engineering using adipose-derived stem cells. J Plast Reconstr Aesthet Surg. 2010;63(5):858-864.
[14] MANDAL B, KUNDU S. Cell proliferation and migration in silk fibroin 3D scaffolds. Biomaterials. 2009;30(15):2956-2965.
[15] CRISTINO S, GRASSI F, TONEGUZZI S, et al. Analysis of mesenchymal stem cells grown on a three-dimensional HYAFF 11-based prototype ligament scaffold. J Biomed Mater Res A. 2005;73:275-283.
[16] GRIFFON DJ, SEDIGHI MR, SCHAEFFER DV, et al.Chitosan scaffolds: interconnective pore size and cartilage engineering. Acta Biomater. 2006; 2(3):313-320.
[17] YAMANE S, IWASAKI N, MAJIMA T, et al. Feasibility of chitosan-based hyaluronic acid hybrid biomaterial for a novel scaffold in cartilage tissue engineering. Biomaterials. 2005;26:611-619.
[18] LIN VS, LEE MC, O’NEAL S, et al. Ligament tissue engineering using synthetic biodegradable fiber scaffolds. Tissue Eng. 1999;5:443-452.
[19] LU HH, COOPER JR JA, MANUEL S, et al. Anterior cruciate ligament regeneration using braided biodegradable scaffolds:in vitro optimization studies. Biomaterials. 2005;26:4805e4816.
[20] WANG M, PEI H, ZHANG L, et al. Hepatogenesis of adipose-derived stem cells on poly-lactide-co-glycolide scaffolds: in vitro and in vivo studies. Tissue Eng Part C Methods. 2010;16:1041.
[21] LI J, TAO R, WU W, et al. 3D PLGA scaffolds improve differentiation and function of bone marrow mesenchymal stem cell-derived hepatocytes. Stem Cells Dev. 2010;19(9):1427-1436.
[22] HOYER M, DRECHSEL N, MEYER M, et al. Embroidered polymer-collagen hybrid scaffold variants for ligament tissue engineering. Mater Sci Eng C Mater Biol Appl. 2014;43:290-299.
[23] TALBOT M, BERRY G, FERNANDES J, et al.Knee dislocations:experience at the Hôpital du Sacré-Coeur de Montréal. Tissue Eng Part C Methods. 2010;16(5):1041-1050.
[24] KRUDWIG WK. Anterior cruciate ligament reconstruction using an alloplastic ligament of polyethylene terephthalate (PET-Trevira-hochfest).Follow-up study. Biomed Mater Eng. 2002;12(1):59-67.
[25] LIDÉN M, EJERHED L, SERNERT N, et al. Re: patellar tendon or semitendinosus tendon autografts for anterior cruciate ligament reconstruction: a prospective, randomized study with a 7-Year follow-up. Am J Sports Med. 2007;35(5):740-748.
[26] MASCARENHAS R, PETER B. MacDonald.Anterior cruciate ligament reconstruction: a look at prosthetics - past, present and possible future. McGill J Med. 2008;11(1):29-37.
[27] GUOAN L, RAMPRASAD P, LOUIS ED, et al. Comparison of the ACL and ACL graft forces before and after ACL reconstruction. Acta Orthop. 2006; 77(2):267-274.
[28] CAREY JL, DUNN WR, DAHM DL, et al. A systematic review of anterior cruciate ligament reconstruction with autograft compared with allograft. J Bone Joint Surg Am. 2009;91(9):2242-2250.
[29] HARNER CD, OLSON E, IRRGANG JJ, et al. Allograft versus autograft anterior cruciate ligament reconstruction: 3- to 5-year outcome. Clin Orthop. 1996; 324:134-144.
[30] PETERSON RK, SHELTON WR, BOMBOY AL. Allograft versus autograft patellar tendon anterior cruciate ligament reconstruction: a 5-year follow-up. Arthroscopy. 2001;17:9-13.
[31] POEHLING GG, CURL WW, LEE CA, et al. Analysis of outcomes of anterior cruciate ligament repair with 5-year follow-up: allograft versus autograft. Arthroscopy. 2005;21:774-785.
[32] SHELTON WR, PAPENDICK L, DUKES AD. Autograft versus allograft anterior cruciate ligament reconstruction. Arthroscopy. 1997;13:446-449.
[33] PEARSALL IV AW, HOLLIS JM, RUSSELL GV JR, et al. A biomechanical comparison of three lower extremity tendons for ligamentous recons truction about the knee. Arthroscopy. 2003;19:1091-1096.
[34] BARRETT G, STOKES D, WHITE M. Anterior cruciate ligament reconstruction in patients older than 40 years: allograft versus autograft patellar tendon. Am J Sports Med. 2005;33:1505-1512.
[35] GROSSMAN MG, ELATTRACHE NS, SHIELDS CL, et al. Revision anterior cruciate ligament reconstruction: three to nine year follow-up. Arthroscopy. 2005;21:418-423.
[36] URIBE JW, HECHTMAN KS, ZVIJAC JE, et al. Revision anterior cruciate ligament surgery: experience from Miami. Clin Orthop. 1996;325:91-99.
[37] COHEN SB, SEKIYA JK. Allograft Safety in Anterior Cruciate Ligament Reconstruction. Clin Sports Med. 2007;26(4):597-605.
[38] LEONG NL, PETRIGLIANO FA, MCALLISTER DR. Current tissue engineering strategies in anterior cruciate ligament reconstruction J Biomed Mater Res A. 2014;102(5):1614-1624.
[39] BORIE DC, CRAMER DV, PHAN-THANH L, et al. Microbiological hazards related to xenotransplantation of porcine organs into man. Infect Control Hosp Epidemiol. 1998;19(5):355-365.
[40] WEESE J. A review of equine zoonotic diseases: risks in veterinary medicine. Proc Ann Convent AAEP. 2002;48:362-369.
[41] KONAKCI KZ, BOHLE B, BLUMER R, et al. Alpha-Gal on bioprostheses: Xenograft immune response in cardiac surgery. Euro J Clin Invest. 2005; 35:17-23.
[42] BOLTON CW, BRUCHMAN WC. The GORE-TEX expanded polytetra-fluoroethylene prosthetic ligament. An in vitro and in vivo evaluation. Clin Orthop Relat Res 1985;(196):202-213.
[43] RICHMOND JC, MANSEAU CJ, PATZ R, et al. Anterior cruciate reconstruction using a Dacron ligament prosthesis. A long-term study. Am J Sports Med. 1992;20(1):24-28.
[44] RADING J, PETERSON L. Clinical experience with the Leeds-Keio artificial ligament in anterior cruciate ligament reconstruction. A prospective two-year follow-up study. Am J Sports Med. 1995;23(3):316-319.
[45] FUJIKAWA K, ISEKI F, SEEDHOM BB. Arthroscopy after anterior cruciate reconstruction with the Leeds-Keio ligament. J Bone Joint Surg Br. 1989; 71(4):566-570.
[46] CHEN T, JIANG J, CHEN S. Status and headway of the clinical application of artificial ligaments. Asia Pac J Sports Med Arthrosc Rehabil Technol. 2015;2(1):15-26.
[47] KOOB TJ, WILLIS TA, QIU YS, et al. Biocompatibility of NDGA-polymerized collagen fibers. II. Attachment, proliferation,and migration of tendon fibroblasts in vitro. J Biomed Mater Res. 2001;56:40-48.
[48] CARUSO AB, DUNN MG. Changes in mechanical properties and cellularity during long-term culture of collagen fiber ACL reconstruction scaffolds. J Biomed Mater Res A. 2005;73:388-397.
[49] DUNN MG, LIESCH JB, TIKU ML, et al. Development of fibroblast-seeded ligament analogs for ACL reconstruction. J Biomed Mater Res. 1995;29: 1363-1371.
[50] BELLINCAMPI LD, CLOSKEY RF, PRASAD R, et al. Viability of fibroblast-seeded ligament analogs after autogenous implantation. J Orthop Res. 1998;16: 414-420.
[51] JAMES R, TOTI US, LAURENCIN CT, et al. Electrospun nanofibrous scaffolds for engineering soft connective tissues. Methods Mol Biol. 2011;726: 243e258.
[52] LIN VS, LEE MC, O’NEAL S, et al. Ligament tissue engineering using synthetic biodegradable fiber scaffolds. Tissue Eng. 1999;5:443e452.
[53] LU HH, COOPER JR JA, MANUEL S, et al. Anterior cruciate ligament regeneration using braided biodegradable scaffolds: in vitro optimization studies. Biomaterials. 2005;26:4805-4816.
[54] LI WJ, LAURENCIN CT, CATERSON EJ, et al. Electrospun nanofibrous structure: A novel scaffold for tissue engineering. J Biomed Mater Res. 2002;60:613-621.
[55] MATTHEWS JA, WNEK GE, SIMPSON DG, et al. Electrospining of collagen nanofibers. Biomacromolecules. 2002;3:232-238.
[56] BOURKE SL, KOHN J, DUNN MG. Preliminary development of a novel resorbable synthetic polymer fiber scaffold for anterior cruciate ligament reconstruction. Tissue Eng. 2004;10:43-52.
[57] SURRAO DC, WALDMAN SD, AMSDEN BG. Biomimetic poly(lactide) based fibrous scaffolds for ligament tissue engineering. Acta Biomater. 2012;8(11):3997-4006.
[58] MALININ TI, LEVITT RL, BASHORE C, et al. A study of retrieved allografts used to replace anterior cruciate ligaments. Arthroscopy. 2002;18:163-170.
[59] GRIER WK, IYOHA EM, HARLEY BAC. The influence of pore size and stiffness on tenocyte bioactivity and transcriptomic stability in collagen –GAG scaffolds. J Mech Behav Biomed Mater. 2017;65:295-305.
[60] YANG J, SHI G, BEI J, et al. Fabrication and surface modification of macroporous poly(L-lactic acid) and poly(L-lactic-co-glycolic acid) (70/30) cell scaffolds for human skin fibroblast cell culture. J Biomed Mater Res. 2002;62(3):438-446.
[61] TANG Y, WONG C, WANG H, et al. Three-dimensional tissue scaffolds from interbonded poly(e-caprolactone) fibrous matrices with controlled porosity. Tissue Eng Part C Methods. 2011;17:209-218.
[62] BUTLER S, KOHLES S, THIELKE R, et al. Interstitial fluid flow in tendons or ligaments:a porous medium finite element simulation. Med Biol Eng Comput. 1997;35:742-746.
[63] RNJAK-KOVACINA J, WISE S, LI Z, et al. Tailoring the porosity and pore size of electrospun synthetic human elastin scaffolds for dermal tissue engineering. Biomaterials. 2011;32:6729-6736.
[64] MANDAL B, KUNDU S. Cell proliferation and migration in silk fibroin 3D scaffolds. Biomaterials. 2009;30:2956-2965.
[65] GU B, PARK S, KIM M, et al. Fabrication of sonicated chitosan nanofiber mat with enlarged porosity for use as hemostatic materials. Carbohydr Polym. 2003;97:65-73.
[66] MURPHY C, O’BRIEN F. Understanding the effect of mean pore size on cell activity in collageng lyco saminogly can scaffolds. Cell Adhes Migr. 2010;4:377-381.
[67] SUSSMAN EM, HALPIN MC, MUSTER J, et al. Porous implants modulate healing and induce shifts in local macrophage polarization in the foreign body reaction. Ann Biomed Eng. 2014;42:1508-1516.
[68] ARTEL A, MEHDIZADEH H, CHIU YC, et al. An agent-based model for the investigation of neovascularization within porous scaffolds. Tissue Eng Part A. 2011;17(17-18):2133-2141.
[69] MURPHY CM, HAUGH MG, O’BRIEN FJ. The effect of mean pore size on cell attachment, proliferation and migration in collagen–glycosaminoglycan scaffolds for bone tissue engineering. Biomaterials. 2010;31(3):461-466.
[70] KUBOKI Y, JIN Q, TAKITA H. Geometry of carriers controlling phenotypic expression in BMP-induced osteogenesis and chondrogenesis. J Bone Joint Surg Am. 2001;83-A Suppl 1(Pt 2):S105-115.
[71] XU MY, ZHANG HR, ZHANG L, et al. Peracetic Acid-Ethanol Processed Human Tendon Allograft: A Morphological, Biochemical, and Biomechanical Study In Vitro. Orthop Surg. 2021. doi: 10.1111/os.13030.
[72] XU HH, SIMON JR CG. Fast setting calcium phosphate-chitosan scaffold: mechanical properties and biocompatibility. Biomaterials. 2005;26:1337-1348.
[73] FREED LE, MARQUIS JC, NOHRIA A, et al. Neocartilage formation in vitro and in vivo using cells cultured on synthetic biodegradable polymers. J Biomed Mater Res. 1993;27:11-23.
[74] CHEN TW, JIANG J, CHEN SY. Status and headway of the clinical application of artificial ligaments. Asia Pac J Sports Med Arthrosc Rehabil Technol. 2015;2(1):15-26.
[75] ITOI U, TAKATORI M, HYAKUSOKU H, et al. Comparison of readily available scaffolds for adipose tissue engineering using adipose-derived stem cells. J Plast Reconstr Aesthet Surg. 2010;63:858-864.
[76] BIMAN BM, SUBHAS CK. Cell proliferation and migration in silk fibroin 3D scaffolds. Biomaterials. 2009;30(15):2956-2965.
[77] EIVAZI ZADEH Z, SOLOUK A, SHAFIEIAN M, et al. Electrospun polyurethane/carbon nanotube composites with different amounts of carbon nanotubes and almost the same fiber diameter for biomedical applications. Mater Sci Eng C Mater Biol Appl. 2021;118:111403.
[78] OLVERA D, SCHIPANI R, SATHY BN, et al. Electrospinning of highly porous yet mechanically functional microfibrillar scaffolds at the human scale for ligament and tendon tissue engineering. Biomed Mater. 2019;14:035016.
[79] GUNER MB, DALGIC AD, TEZCANER A, et al. A dual-phase scaffold produced by rotary jet spinning and electrospinning for tendon tissue engineering. Biomed Mater. 2020;15:065014.
[80] JAMES R, KESTURU G, BALIAN G, et al. Tendon:biology, biomechanics, repair, growth factors, and evolving treatment options. J Hand Surg Am. 2008;33(1):102-112.
[81] CHEN J, XU J, WANG A, et al. Scaffolds fortendonand ligament repair: review of the efficacy ofcommercial products. Expert Rev Med Devices. 2009;6(1):61-73.
[82] BAYER ML, SCHJERLING P, HERCHENHAN A, et al. Release of tensile strain on engineered human tendon tissue disturbs cell adhesions, changes matrix architecture,and induces an inflammatory phenotype. PLoS One. 2014;9(1):86078.
[83] MATHEW B, SARAH S, STEPHANIE D. Augmenting endogenous repair of soft tissues with nanofibre scaffolds. J R Soc Interface. 2018;15(141):20180019.
[84] NELLY AP, EVAN LF, LOUIS JS. Tendon basic science: Development, repair, regeneration, and healing. J Orthop Res. 2015;33(6):780-784. |