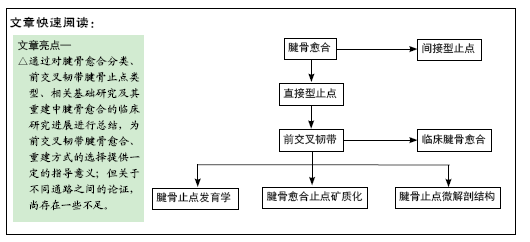
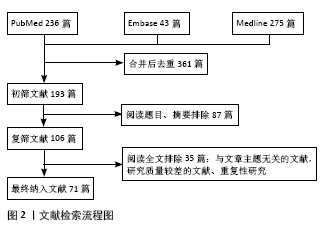
[1] MARKOLF KL, MENSCH JS, AMSTUTZ HC. Stiffness and laxity of the knee--the contributions of the supporting structures. A quantitative in vitro study. J Bone Joint Surg Am. 1976;58(5):583-594.
[2] LEVINE JW, KIAPOUR AM, QUATMAN CE, et al. Clinically relevant injury patterns after an anterior cruciate ligament injury provide insight into injury mechanisms. Am J Sports Med. 2013;41(2):385-395.
[3] LAI CCH, ARDERN CL, FELLER JA, et al. Eighty-three per cent of elite athletes return to preinjury sport after anterior cruciate ligament reconstruction: a systematic review with meta-analysis of return to sport rates, graft rupture rates and performance outcomes. Br J Sports Med. 2018;52(2):128-138.
[4] AGEL J, ROCKWOOD T, KLOSSNER D. Collegiate ACL Injury Rates Across 15 Sports: National Collegiate Athletic Association Injury Surveillance System Data Update (2004-2005 Through 2012-2013) . Clin J Sport Med. 2016;26(6):518-523.
[5] ØIESTAD BE, ENGEBRETSEN L, STORHEIM K, et al. Knee osteoarthritis after anterior cruciate ligament injury: a systematic review. Am J Sports Med. 2009;37(7):1434-1443.
[6] LOHMANDER LS, ENGLUND PM, DAHL LL, et al. The long-term consequence of anterior cruciate ligament and meniscus injuries: osteoarthritis. Am J Sports Med. 2007;35(10):1756-1769.
[7] MIHELIC R, JURDANA H, JOTANOVIC Z, et al. Long-term results of anterior cruciate ligament reconstruction: a comparison with non-operative treatment with a follow-up of 17-20 years. Int Orthop. 2011;35(7):1093-1097.
[8] COX CL, HUSTON LJ, DUNN WR, et al. Are articular cartilage lesions and meniscus tears predictive of IKDC, KOOS, and Marx activity level outcomes after anterior cruciate ligament reconstruction? A 6-year multicenter cohort study. Am J Sports Med. 2014;42(5):1058-1067.
[9] OIESTAD BE, HOLM I, AUNE AK, et al. Knee function and prevalence of knee osteoarthritis after anterior cruciate ligament reconstruction: a prospective study with 10 to 15 years of follow-up. Am J Sports Med. 2010;38(11):2201-2210.
[10] RISBERG MA, OIESTAD BE, GUNDERSON R, et al. Changes in Knee Osteoarthritis, Symptoms, and Function After Anterior Cruciate Ligament Reconstruction: A 20-Year Prospective Follow-up Study. Am J Sports Med. 2016;44(5):1215-1224.
[11] GIOVE TP, MILLER SJ, KENT BE, et al. Non-operative treatment of the torn anterior cruciate ligament. J Bone Joint Surg Am. 1983;65(2): 184-192.
[12] MAHAPATRA P, HORRIAT S, ANAND BS. Anterior cruciate ligament repair - past, present and future. J Exp Orthop. 2018;5(1):20.
[13] FOSTER TE, WOLFE BL, RYAN S, et al. Does the graft source really matter in the outcome of patients undergoing anterior cruciate ligament reconstruction? An evaluation of autograft versus allograft reconstruction results: a systematic review. Am J Sports Med. 2010; 38(1):189-199.
[14] SAMUELSEN BT, WEBSTER KE, JOHNSON NR, et al. Hamstring Autograft versus Patellar Tendon Autograft for ACL Reconstruction: Is There a Difference in Graft Failure Rate? A Meta-analysis of 47,613 Patients. Clin Orthop Relat Res. 2017;475(10):2459-2468.
[15] XIE X, LIU X, CHEN Z, et al. A meta-analysis of bone-patellar tendon-bone autograft versus four-strand hamstring tendon autograft for anterior cruciate ligament reconstruction. Knee. 2015;22(2):100-110.
[16] MOHTADI N, CHAN D, BARBER R, et al. A Randomized Clinical Trial Comparing Patellar Tendon, Hamstring Tendon, and Double-Bundle ACL Reconstructions: Patient-Reported and Clinical Outcomes at a Minimal 2-Year Follow-up. Clin J Sport Med. 2015;25(4):321-331.
[17] PARK SH, CHOI YJ, MOON SW, et al. Three-Dimensional Bio-Printed Scaffold Sleeves With Mesenchymal Stem Cells for Enhancement of Tendon-to-Bone Healing in Anterior Cruciate Ligament Reconstruction Using Soft-Tissue Tendon Graft. Arthroscopy. 2018; 34(1):166-179.
[18] LU H, CHEN C, XIE S, et al. Tendon Healing in Bone Tunnel after Human Anterior Cruciate Ligament Reconstruction: A Systematic Review of Histological Results. J Knee Surg. 2019;32(5):454-462.
[19] CHEN CH. Graft healing in anterior cruciate ligament reconstruction. Sports Med Arthrosc Rehabil Ther Technol. 2009;1(1): 21.
[20] BUEHLER MJ, YUNG YC. Deformation and failure of protein materials in physiologically extreme conditions and disease. Nature Mat. 2009; 8(3):175-188.
[21] DENG X H, LEBASCHI A, CAMP CL, et al. Expression of Signaling Molecules Involved in Embryonic Development of the Insertion Site Is Inadequate for Reformation of the Native Enthesis: Evaluation in a Novel Murine ACL Reconstruction Model. J Bone Joint Surg Am. 2018;100(15):e102.
[22] BLITZ E, SHARIR A, AKIYAMA H, et al. Tendon-bone attachment unit is formed modularly by a distinct pool of Scx- and Sox9-positive progenitors. Development. 2013;140(13):2680-2690.
[23] BRENT AE, BRAUN T, TABIN CJ. Genetic analysis of interactions between the somitic muscle, cartilage and tendon cell lineages during mouse development. Development. 2005;132(3):515-528.
[24] SUGIMOTO Y, TAKIMOTO A, AKIYAMA H, et al. Scx+/Sox9+ progenitors contribute to the establishment of the junction between cartilage and tendon/ligament. Development. 2013;140(11):2280-2288.
[25] BLITZ E, VIUKOV S, SHARIR A, et al. Bone ridge patterning during musculoskeletal assembly is mediated through SCX regulation of Bmp4 at the tendon-skeleton junction. Dev Cell. 2009;17(6):861-873.
[26] KULYK WM, RODGERS BJ, GREER K, et al. Promotion of embryonic chick limb cartilage differentiation by transforming growth factor-β. Dev Biol. 1989;135(2):424-430.
[27] CARRINGTON J L, CHEN P, YANAGISHITA M, et al. Osteogenin (bone morphogenetic protein-3) stimulates cartilage formation by chick limb bud cells in vitro. Dev Biol.1991;146(2):406-415.
[28] VERRECCHIA F, MAUVIEL A. Transforming growth factor-β signaling through the Smad pathway: role in extracellular matrix gene expression and regulation. J Invest Dermatol. 2002;118(2):211-215.
[29] PRYCE BA, WATSON SS, MURCHISON ND, et al. Recruitment and maintenance of tendon progenitors by TGFβ signaling are essential for tendon formation. Development. 2009;136(8):1351-1361.
[30] LORDA-DIEZ CI, MONTERO JA, MARTINEZ-CUE C, et al. Transforming growth factors β coordinate cartilage and tendon differentiation in the developing limb mesenchyme. J Biol Chem. 2009;284(43): 29988-29996.
[31] OVCHINNIKOV DA, SELEVER J, WANG Y, et al. BMP receptor type IA in limb bud mesenchyme regulates distal outgrowth and patterning. Dev Biol. 2006;295(1):103-115.
[32] HOSSEINI A, HOGG D. The effects of paralysis on skeletal development in the chick embryo. II. Effects on histogenesis of the tibia. J Anat. 1991; 177:169-178.
[33] EDOM-VOVARD F, SCHULER B, BONNIN MA, et al. Fgf4 positively regulates scleraxis and tenascin expression in chick limb tendons. Dev Biol. 2002;247(2):351-366.
[34] ROSEN CJ. Primer on the metabolic bone diseases and disorders of mineral metabolism. John Wiley Sons,2009.
[35] KARSENTY G, WAGNER EF. Reaching a genetic and molecular understanding of skeletal development. Dev Cell. 2002;2(4):389-406.
[36] APOSTOLAKOS J, DURANT TJ, DWYER CR, et al. The enthesis: a review of the tendon-to-bone insertion. Muscles Ligaments Tendons J. 2014;4(3): 333-342.
[37] BENJAMIN M, TOUMI H, RALPHS JR, et al. Where tendons and ligaments meet bone: attachment sites (‘entheses’) in relation to exercise and/or mechanical load. J Anat. 2006;208(4):471-490.
[38] THAMBYAH A, LEI Z, BROOM N. Microanatomy of the medial collateral ligament enthesis in the bovine knee. Anat Rec (Hoboken). 2014;297(12):2254-2261.
[39] ROSSETTI L, KUNTZ LA, KUNOLD E, et al. The microstructure and micromechanics of the tendon-bone insertion. Nat Mater. 2017;16(6): 664-670.
[40] ZHAO L, LEE PVS, ACKLAND DC, et al. Microstructure Variations in the Soft-Hard Tissue Junction of the Human Anterior Cruciate Ligament. Anat Rec (Hoboken). 2017;300(9):1547-1559.
[41] DAI C, GUO L, YANG L, et al. Regional fibrocartilage variations in human anterior cruciate ligament tibial insertion: a histological three-dimensional reconstruction. Connect Tissue Res. 2015;56(1):18-24.
[42] ZHAO L, THAMBYAH A, BROOM ND. A multi-scale structural study of the porcine anterior cruciate ligament tibial enthesis. J Anat. 2014;224(6): 624-633.
[43] BEAULIEU ML, CAREY GE, SCHLECHT SH, et al. On the heterogeneity of the femoral enthesis of the human ACL: microscopic anatomy and clinical implications. J Exp Orthop. 2016;3(1):14.
[44] QU D, SUBRAMONY SD, BOSKEY AL, et al. Compositional mapping of the mature anterior cruciate ligament-to-bone insertion. J Orthop Res. 2017;35(11):2513-2523.
[45] KUNTZ LA, ROSSETTI L, KUNOLD E, et al. Biomarkers for tissue engineering of the tendon-bone interface. PLoS One. 2018;13(1): e0189668.
[46] XU K, KUNTZ LA, FOEHR P, et al. Efficient decellularization for tissue engineering of the tendon-bone interface with preservation of biomechanics. PLoS One. 2017;12(2):e0171577.
[47] LUETKEMEYER CM, MARCHI BC, ASHTON-MILLER JA, et al. Femoral entheseal shape and attachment angle as potential risk factors for anterior cruciate ligament injury. J Mech Behav Biomed Mater. 2018;88:313-321.
[48] BEAULIEU ML, CAREY GE, SCHLECHT SH, et al. Quantitative comparison of the microscopic anatomy of the human ACL femoral and tibial entheses. J Orthop Res. 2015;33(12):1811-1817.
[49] CHEN J, KIM J, SHAO W, et al. An Anterior Cruciate Ligament Failure Mechanism. Am J Sports Med. 2019;47(9):2067-2076.
[50] SCHLECHT SH, MARTIN CT, OCHOCKI DN, et al. Morphology of Mouse Anterior Cruciate Ligament-Complex Changes Following Exercise During Pubertal Growth. J Orthop Res. 2019;37(9):1910-1919.
[51] SUZUKI D, OTSUBO H, ADACHI T, et al. Functional Adaptation of the Fibrocartilage and Bony Trabeculae at the Attachment Sites of the Anterior Cruciate Ligament. Clin Anat. 2020;33(7):988-996.
[52] EINHORN TA, BUCKWALTER JA, O’KEEFE RJ. Orthopaedic basic science: foundations of clinical practice. Amer Acad Orthop. 2007.
[53] HEXTER AT, THANGARAJAH T, BLUNN G, et al. Biological augmentation of graft healing in anterior cruciate ligament reconstruction: a systematic review. Bone Joint J. 2018;100-b(3):271-284.
[54] HAO ZC, WANG SZ, ZHANG XJ, et al. Stem cell therapy: a promising biological strategy for tendon-bone healing after anterior cruciate ligament reconstruction. Cell Prolif. 2016;49(2):154-162.
[55] ZHOU Y, ZHANG J, YANG J, et al. Kartogenin with PRP promotes the formation of fibrocartilage zone in the tendon-bone interface. J Tissue Eng Regen Med. 2017;11(12):3445-3456.
[56] ZHANG Y, YU J, ZHANG J, et al. Simvastatin With PRP Promotes Chondrogenesis of Bone Marrow Stem Cells In Vitro and Wounded Rat Achilles Tendon-Bone Interface Healing In Vivo. Am J Sports Med. 2019; 47(3):729-739.
[57] MURRAY IR, GEESLIN AG, GOUDIE EB, et al. Minimum Information for Studies Evaluating Biologics in Orthopaedics (MIBO): Platelet-Rich Plasma and Mesenchymal Stem Cells. J Bone Joint Surg Am. 2017; 99(10):809-819.
[58] AGIR I, AYTEKIN MN, KUCUKDURMAZ F, et al. The effect of platelet-rich plasma in bone-tendon integration. Adv Clin Exp Med. 2017;26(2): 193-199.
[59] TIAN X, JIANG H, CHEN Y, et al. Baicalein Accelerates Tendon-Bone Healing via Activation of Wnt/beta-Catenin Signaling Pathway in Rats. BioMed Res Int. 2018;2018:3849760.
[60] HU J, YAO B, YANG X, et al. The immunosuppressive effect of Siglecs on tendon-bone healing after ACL reconstruction. Med Hypotheses. 2015; 84(1):38-39.
[61] MUTSUZAKI H, NAKAJIMA H, NOMURA S, et al. Differences in placement of calcium phosphate-hybridized tendon grafts within the femoral bone tunnel during ACL reconstruction do not influence tendon-to-bone healing. J Orthop Surg Res. 2017;12(1):80.
[62] SUN Z, WANG X, LING M, et al. Acceleration of tendon-bone healing of anterior cruciate ligament graft using intermittent negative pressure in rabbits. J Orthop Surg Res. 2017;12(1):60.
[63] SONG F, JIANG D, WANG T, et al. Mechanical Loading Improves Tendon-Bone Healing in a Rabbit Anterior Cruciate Ligament Reconstruction Model by Promoting Proliferation and Matrix Formation of Mesenchymal Stem Cells and Tendon Cells. Cell Physiol Biochem. 2017; 41(3):875-889.
[64] WANG J, XU J, SONG B, et al. Magnesium (Mg) based interference screws developed for promoting tendon graft incorporation in bone tunnel in rabbits. Acta Biomaterialia. 2017;63:393-410.
[65] LI X, CHENG R, SUN Z, et al. Flexible bipolar nanofibrous membranes for improving gradient microstructure in tendon-to-bone healing. Acta Biomaterialia. 2017;61:204-216.
[66] TEUSCHL AH, TANGL S, HEIMEL P, et al. Osteointegration of a Novel Silk Fiber-Based ACL Scaffold by Formation of a Ligament-Bone Interface. Am J Sports Med. 2019;47(3):620-627.
[67] DRURY JL, MOONEY DJ. Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials. 2003;24(24):4337-4351.
[68] WIIG ME, AMIEL D, VANDEBERG J, et al. The early effect of high molecular weight hyaluronan (hyaluronic acid) on anterior cruciate ligament healing: an experimental study in rabbits. J Orthop Res. 1990;8(3):425-434.
[69] BERRY SM, GREEN MH. Hyaluronan: a potential carrier for growth factors for the healing of ligamentous tissues. Wound Repair Regen. 1997;5(1):33-38.
[70] FLEMING BC, MAGARIAN EM, HARRISON SL, et al. Collagen scaffold supplementation does not improve the functional properties of the repaired anterior cruciate ligament. J Orthop Res. 2010;28(6):703-709.
[71] JOSHI SM, MASTRANGELO AN, MAGARIAN EM, et al. Collagen-platelet composite enhances biomechanical and histologic healing of the porcine anterior cruciate ligament. Am J Sports Med. 2009;37(12): 2401-2410. |