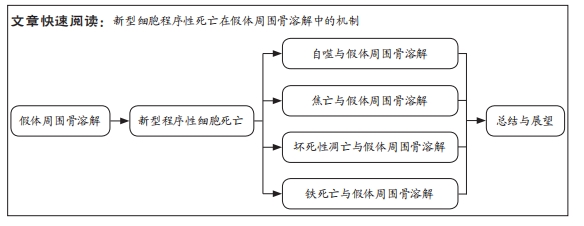
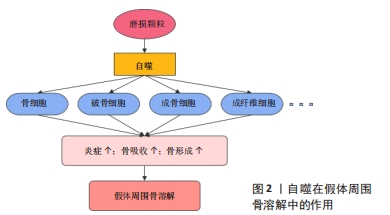
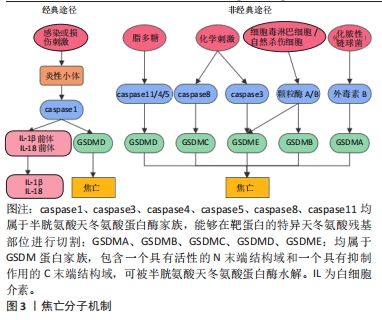
[1] SINGH JA, YU S, CHEN L, et al. Rates of Total Joint Replacement in the United States: Future Projections to 2020–2040 Using the National Inpatient Sample. J Rheumatol. 2019;46(9):1134-1140.
[2] BURKE NG, GIBBONS JP, CASSAR-GHEITI AJ, et al. Total hip replacement—the cause of failure in patients under 50 years old? Ir J Med Sci. 2019; 188(3):879-883.
[3] CHARETTE RS, SLOAN M, DEANGELIS RD, et al. Higher Rate of Early Revision Following Primary Total Knee Arthroplasty in Patients Under Age 55: A Cautionary Tale. J Arthroplasty. 2019;34(12):2918-2924.
[4] WEBER M, RENKAWITZ T, VOELLNER F, et al. Revision Surgery in Total Joint Replacement Is Cost-Intensive. Biomed Res Int. 2018;2018:8987104.
[5] HUNT LP, WHITEHOUSE MR, BESWICK A, et al. Implications of Introducing New Technology: Comparative Survivorship Modeling of Metal-on-Metal Hip Replacements and Contemporary Alternatives in the National Joint Registry. J Bone Joint Surg Am. 2018;100(3):189-196.
[6] CHALMERS BP, SYKU M, JOSEPH AD, et al. High Rate of Re-Revision in Patients Less Than 55 Years of Age Undergoing Aseptic Revision Total Knee Arthroplasty. J Arthroplasty. 2021;36(7):2348-2352.
[7] GALLO J, GOODMAN SB, KONTTINEN YT, et al. Osteolysis around total knee arthroplasty: A review of pathogenetic mechanisms. Acta Biomater. 2013; 9(9):8046-8058.
[8] GALLO J, GOODMAN SB, KONTTINEN YT, et al. Particle disease: Biologic mechanisms of periprosthetic osteolysis in total hip arthroplasty. Innate Immun. 2013;19(2):213-224.
[9] GOODMAN SB, GALLO J. Periprosthetic Osteolysis: Mechanisms, Prevention and Treatment. J Clin Med. 2019;8(12):2091.
[10] BEDOUI S, HEROLD MJ, STRASSER A. Emerging connectivity of programmed cell death pathways and its physiological implications. Nat Rev Mol Cell Biol. 2020;21(11):678-695.
[11] 刘国印, 赵建宁, 王瑞. 磨损微粒诱导细胞凋亡与无菌性松动的研究进展[J]. 中国骨伤,2013,26(9):791-796.
[12] 蒋营军, 吴连国. 人工关节置换术后磨损颗粒与假体周围骨溶解的研究进展[J]. 中国骨伤,2016,29(10):968-972.
[13] DIKIC I, ELAZAR Z. Mechanism and medical implications of mammalian autophagy. Nat Rev Mol Cell Biol. 2018;19(6):349-364.
[14] MIZUSHIMA N, KOMATSU M. Autophagy: renovation of cells and tissues. Cell. 2011;147(4):728-741.
[15] NAH J, ZABLOCKI D, SADOSHIMA J. The role of autophagic cell death in cardiac disease. J Mol Cell Cardiol. 2022;173:16-24.
[16] HURLEY JH, YOUNG LN. Mechanisms of Autophagy Initiation. Annual Review of Biochemistry. 2017;86(1):225-244.
[17] KIM YC, GUAN KL. mTOR: a pharmacologic target for autophagy regulation. J Clin Invest. 2015;125(1):25-32.
[18] KLIONSKY DJ, PETRONI G, AMARAVADI RK, et al. Autophagy in major human diseases. EMBO J. 2021;40(19):e108863.
[19] 蔡燕, 施勤, 赵环, 等. 聚甲基丙烯酸甲酯颗粒诱导骨溶解实验研究[J]. 重庆医学,2013,42(34):4160-4161+4165.
[20] 严嘉琦, 张云, 刘方舒, 等. 磷酸三钙磨损颗粒诱导小鼠假体周围骨细胞损伤的作用[J]. 中国应用生理学杂志,2018,34(1):83-87.
[21] XIAN G, CHEN W, GU M, et al. Titanium particles induce apoptosis by promoting autophagy in macrophages via the PI3K/Akt signaling pathway. J Biomed Mater Res A. 2020;108(9):1792-1805.
[22] WANG L, GAO Z, ZHANG J, et al. Netrin-1 regulates ERK1/2 signaling pathway and autophagy activation in wear particle-induced osteoclastogenesis. Cell Biol Int. 2021;45(3):612-622.
[23] CHEN W, XIAN G, GU M, et al. Autophagy inhibitors 3-MA and LY294002 repress osteoclastogenesis and titanium particle-stimulated osteolysis. Biomater Sci. 2021;9(14):4922-4935.
[24] CHEN J, YAO Y, WANG Y, et al. Autophagy triggered by the ROS/ERK signaling pathway protects mouse embryonic palatal cells from apoptosis induced by nicotine. Environ Sci Pollut Res Int. 2022;29(54):81909-81922.
[25] CHU B, CHEN S, ZHENG X, et al. Nepetin inhibits osteoclastogenesis by inhibiting RANKL-induced activation of NF-κB and MAPK signalling pathway, and autophagy. J Cell Mol Med. 2020;24(24):14366-14380.
[26] 董凡赫, 寿今豪, 陈宇峰, 等. 重楼皂苷I通过抑制自噬减轻磷酸三钙磨损颗粒诱导成骨细胞损伤的实验研究[J]. 中草药,2020,51(9):2501-2508.
[27] ZHANG Q, ZHANG XF. Hyperoside decreases the apoptosis and autophagy rates of osteoblast MC3T3-E1 cells by regulating TNF-like weak inducer of apoptosis and the p38mitogen activated protein kinase pathway. Mol Med Rep. 2019;19(1):41-50.
[28] ZHANG Z, FU X, XU L, et al. Nanosized Alumina Particle and Proteasome Inhibitor Bortezomib Prevented inflammation and Osteolysis Induced by Titanium Particle via Autophagy and NF-κB Signaling. Sci Rep. 2020;10(1): 5562.
[29] WU W, WANG L, MAO YQ, et al. Impaired Autophagy in the Fibroblasts by Titanium Particles Increased the Release of CX3CL1 and Promoted the Chemotactic Migration of Monocytes. Inflammation. 2020;43(2):673-685.
[30] LI D, WANG C, LI Z, et al. Nano-sized Al2O3 particle-induced autophagy reduces osteolysis in aseptic loosening of total hip arthroplasty by negative feedback regulation of RANKL expression in fibroblasts. Cell Death Dis. 2018; 9(8):840.
[31] COOKSON BT, BRENNAN MA. Pro-inflammatory programmed cell death. Trends in Microbiology. 2001;9(3):113-114.
[32] GALLUZZI L, VITALE I, AARONSON SA, et al. Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018;25(3):486-541.
[33] XUE W, CUI D, QIU Y. Research Progress of Pyroptosis in Alzheimer’s Disease. Front Mol Neurosci. 2022;15:872471.
[34] MAN SM, KARKI R, KANNEGANTI TD. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol Rev. 2017;277(1):61-75.
[35] NEWTON K, WICKLIFFE KE, MALTZMAN A, et al. Activity of caspase-8 determines plasticity between cell death pathways. Nature. 2019;575(7784): 679-682.
[36] WANG Y, GAO W, SHI X, et al. Chemotherapy drugs induce pyroptosis through caspase-3 cleavage of a gasdermin. Nature. 2017;547(7661):99-103.
[37] ZHONG Y, KINIO A, SALEH M. Functions of NOD-Like Receptors in Human Diseases. Front Immunol. 2013;4:333.
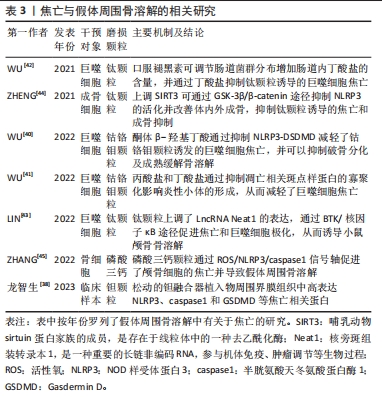
[38] 龙智生, 扶流祥, 龚飞鹏, 等. 细胞焦亡相关蛋白在钽融合器松动周围组织中的表达及意义[J]. 中国组织工程研究,2023,27(25):4057-4062.
[39] SON HS, LEE J, LEE HI, et al. Benzydamine inhibits osteoclast differentiation and bone resorption via down-regulation of interleukin-1β expression. Acta Pharm Sin B. 2020;10(3):462-474.
[40] WU Y, TENG Y, ZHANG C, et al. The ketone body β-hydroxybutyrate alleviates CoCrMo alloy particles induced osteolysis by regulating NLRP3 inflammasome and osteoclast differentiation. J Nanobiotechnology. 2022; 20:120.
[41] WU YL, ZHANG CH, TENG Y, et al. Propionate and butyrate attenuate macrophage pyroptosis and osteoclastogenesis induced by CoCrMo alloy particles. Mil Med Res. 2022;9:46.
[42] WU Y, HE F, ZHANG C, et al. Melatonin alleviates titanium nanoparticles induced osteolysis via activation of butyrate/GPR109A signaling pathway. J Nanobiotechnology. 2021;19:170.
[43] LIN S, WEN Z, LI S, et al. LncRNA Neat1 promotes the macrophage inflammatory response and acts as a therapeutic target in titanium particle-induced osteolysis. Acta Biomater. 2022;142:345-360.
[44] ZHENG K, BAI J, LI N, et al. Protective effects of sirtuin 3 on titanium particle-induced osteogenic inhibition by regulating the NLRP3 inflammasome via the GSK-3β/β-catenin signalling pathway. Bioact Mater. 2021;6(10):3343-3357.
[45] ZHANG Y, YAN M, NIU W, et al. Tricalcium phosphate particles promote pyroptotic death of calvaria osteocytes through the ROS/NLRP3/Caspase-1 signaling axis in amouse osteolysis model. Int Immunopharmacol. 2022; 107:108699.
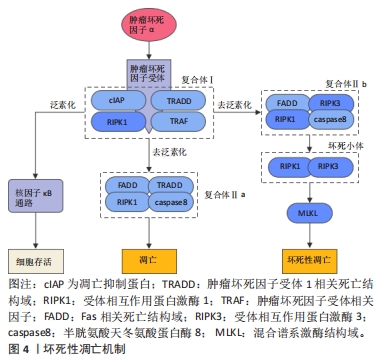
[46] GALLUZZI L, KEPP O, CHAN FKM, et al. Necroptosis: Mechanisms and Relevance to Disease. Annu Rev Pathol. 2017;12:103-130.
[47] JACO I, ANNIBALDI A, LALAOUI N, et al. MK2 Phosphorylates RIPK1 to Prevent TNF-Induced Cell Death. Mol Cell. 2017;66(5):698-710.e5.
[48] HU X, WANG Z, KONG C, et al. Necroptosis: A new target for prevention of osteoporosis. Front Endocrinol (Lausanne). 2022;13:1032614.
[49] CAI Z, JITKAEW S, ZHAO J, et al. Plasma membrane translocation of trimerized MLKL protein is required for TNF-induced necroptosis. Nat Cell Biol. 2014;16(1):55-65.
[50] MOUJALLED DM, COOK WD, OKAMOTO T, et al. TNF can activate RIPK3 and cause programmed necrosis in the absence of RIPK1. Cell Death Dis. 2013;4(1):e465-e465.
[51] CHAGANTI RK, PURDUE E, SCULCO TP, et al. Elevation of Serum Tumor Necrosis Factor α in Patients with Periprosthetic Osteolysis: A Case-Control Study. Clin Orthop Relat Res. 2014;472(2):584-589.
[52] SCHWARZ EM, LOONEY RJ, O’KEEFE RJ. Anti-TNF-α therapy as a clinical intervention for periprosthetic osteolysis. Arthritis Res. 2000;2(3):165-168.
[53] SHI G, JIA P, CHEN H, et al. Necroptosis occurs in osteoblasts during tumor necrosis factor-α stimulation and caspase-8 inhibition. Braz J Med Biol Res. 2018;52(1):e7844.
[54] MOEN IN, WESTHRIN M, HÅLAND E, et al. Smac-mimetics reduce numbers and viability of human osteoclasts. Cell Death Discov. 2021;7(1):36.
[55] MULLIN BH, TICKNER J, ZHU K, et al. Characterisation of genetic regulatory effects for osteoporosis risk variants in human osteoclasts. Genome Biol. 2020;21(1):80.
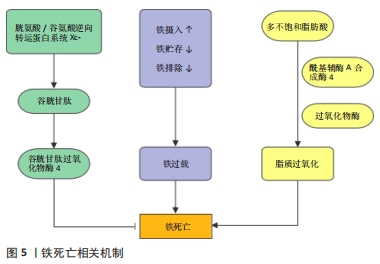
[56] DIXON SJ, LEMBERG KM, LAMPRECHT MR, et al. Ferroptosis: An Iron-Dependent Form of Non-Apoptotic Cell Death. Cell. 2012;149(5):1060-1072.
[57] YANG WS, SRIRAMARATNAM R, WELSCH ME, et al. Regulation of Ferroptotic Cancer Cell Death by GPX4. Cell. 2014;156(1-2):317-331.
[58] WANG YQ, CHANG SY, WU Q, et al. The Protective Role of Mitochondrial Ferritin on Erastin-Induced Ferroptosis. Front Aging Neurosci. 2016;8:308.
[59] GENG N, SHI BJ, LI SL, et al. Knockdown of ferroportin accelerates erastin-induced ferroptosis in neuroblastoma cells. Eur Rev Med Pharmacol Sci. 2018;22(12):3826-3836.
[60] DOLL S, PRONETH B, TYURINA YY, et al. Acsl4 Dictates Ferroptosis Sensitivity by Shaping Cellular Lipid Composition. Nat Chem Biol. 2017;13(1): 91.
[61] CLEMENTE SM, MARTÍNEZ-COSTA OH, MONSALVE M, et al. Targeting Lipid Peroxidation for Cancer Treatment. Molecules. 2020;25(21):5144.
[62] STOCKWELL BR. Ferroptosis turns 10: Emerging mechanisms, physiological functions, and therapeutic applications. Cell. 2022;185(14):2401-2421.
[63] JENEY V. Clinical Impact and Cellular Mechanisms of Iron Overload-Associated Bone Loss. Front Pharmacol. 2017;8:77.
[64] JING X, DU T, LI T, et al. The detrimental effect of iron on OA chondrocytes: Importance of pro-inflammatory cytokines induced iron influx and oxidative stress. J Cell Mol Med. 2021;25(12):5671-5680.
[65] NI S, KUANG Y, YUAN Y, et al. Mitochondrion-mediated iron accumulation promotes carcinogenesis and Warburg effect through reactive oxygen species in osteosarcoma. Cancer Cell Int. 2020;20(1):399.
[66] XU Y, SANG W, ZHONG Y, et al. CoCrMo‐Nanoparticles induced peri‐implant osteolysis by promoting osteoblast ferroptosis via regulating Nrf2‐ARE signalling pathway. Cell Prolif. 2021;54(12):e13142.
[67] DOYARD M, FATIH N, MONNIER A, et al. Iron excess limits HHIPL-2 gene expression and decreases osteoblastic activity in human MG-63 cells. Osteoporos Int. 2012;23(10):2435-2445.
[68] YANG J, DONG D, LUO X, et al. Iron Overload-Induced Osteocyte Apoptosis Stimulates Osteoclast Differentiation Through Increasing Osteocytic RANKL Production In Vitro. Calcif Tissue Int. 2020;107(5):499-509. |