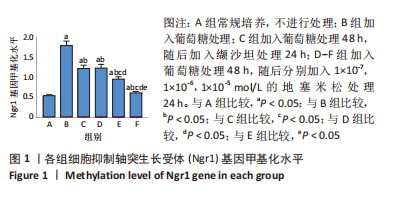
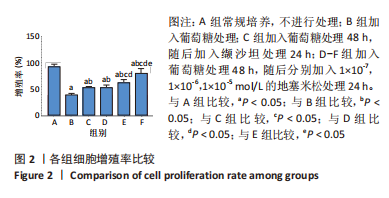
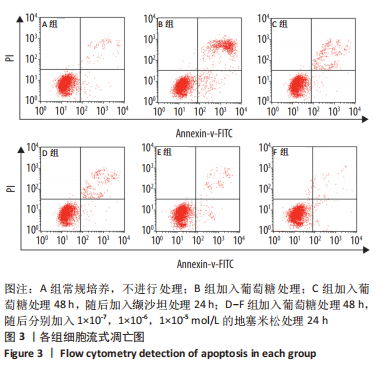
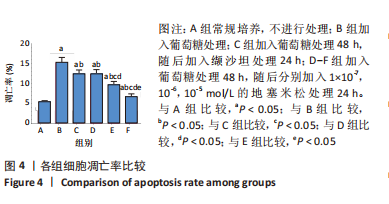
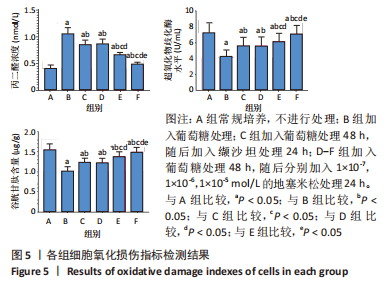
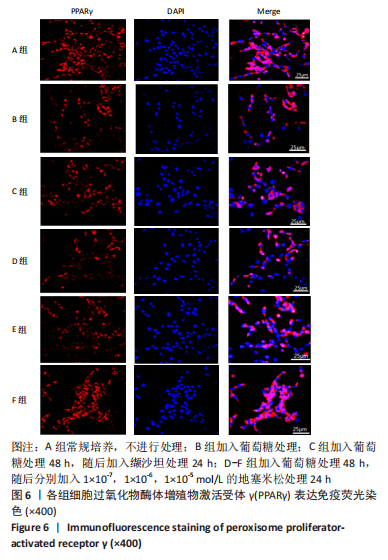
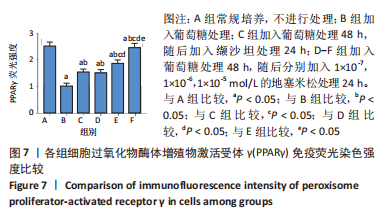
[1] SAWAF H, THOMAS G, TALIERCIO JJ, et al. Therapeutic Advances in Diabetic Nephropathy. J Clin Med. 2022;11(2):378.
[2] 王薇,匡丹丹.糖尿病肾病患者血清纤维化指标与肾血流的关系探究[J].中国医学创新,2022,19(2):65-68.
[3] LI X, LU L, HOU W, et al. Epigenetics in the pathogenesis of diabetic nephropathy. Acta Biochim Biophys Sin (Shanghai). 2022;54(2):163-172.
[4] TANG G, DU Y, GUAN H, et al. Butyrate ameliorates skeletal muscle atrophy in diabetic nephropathy by enhancing gut barrier function and FFA2-mediated PI3K/Akt/mTOR signals. Br J Pharmacol. 2022;179(1): 159-178.
[5] LIU L, BAI F, SONG H, et al. Upregulation of TIPE1 in tubular epithelial cell aggravates diabetic nephropathy by disrupting PHB2 mediated mitophagy. Redox Biol. 2022;50:102260.
[6] CAO Z, HUANG D, TANG C, et al. Pyroptosis in diabetes and diabetic nephropathy. Clin Chim Acta. 2022;531:188-196.
[7] HUANG C, ZHOU Y, HUANG H, et al. Islet Transplantation Reverses Podocyte Injury in Diabetic Nephropathy or Induced by High Glucose via Inhibiting RhoA/ROCK/NF-κB Signaling Pathway. J Diabetes Res. 2021;2021:9570405.
[8] 张锦华,张媛,高明,等.环孢素A对糖尿病肾病大鼠足细胞氧化应激损伤的作用研究[J].现代药物与临床,2022,37(1):19-24.
[9] ZHONG Y, LIU J, SUN D, et al. Dioscin relieves diabetic nephropathy via suppressing oxidative stress and apoptosis, and improving mitochondrial quality and quantity control. Food Funct. 2022;13(6): 3660-3673.
[10] YAN R, WANG Y, SHI M, et al. Regulation of PTEN/AKT/FAK pathways by PPARγ impacts on fibrosis in diabetic nephropathy. J Cell Biochem. 2019. doi: 10.1002/jcb.27937.
[11] 桑苗苗,吴伟斌,高琴,等.高尿酸血症对大鼠肾小球足细胞的影响及其机制研究[J].解剖科学进展,2021,27(6):718-722.
[12] LIU XQ, JIANG L, LI YY, et al. Wogonin protects glomerular podocytes by targeting Bcl-2-mediated autophagy and apoptosis in diabetic kidney disease. Acta Pharmacol Sin. 2022;43(1):96-110.
[13] HUANG LM, MAO JH. Glomerular podocyte dysfunction in inherited renal tubular disease. World J Pediatr. 2021;17(3):227-233.
[14] KARTI O, SAATCI AO. Place of intravitreal dexamethasone implant in the treatment armamentarium of diabetic macular edema. World J Diabetes. 2021;12(8):1220-1232.
[15] USMANI SZ, QUACH H, MATEOS MV, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): updated outcomes from a randomised, multicentre, open-label, phase 3 study. Lancet Oncol. 2022;23(1):65-76.
[16] RICHARDSON PG, PERROT A, SAN-MIGUEL J, et al. Isatuximab plus pomalidomide and low-dose dexamethasone versus pomalidomide and low-dose dexamethasone in patients with relapsed and refractory multiple myeloma (ICARIA-MM): follow-up analysis of a randomised, phase 3 study. Lancet Oncol. 2022;23(3):416-427.
[17] HUSSAIN LODHI A, AHMAD FU, FURWA K, et al. Role of Oxidative Stress and Reduced Endogenous Hydrogen Sulfide in Diabetic Nephropathy. Drug Des Devel Ther. 2021;15:1031-1043.
[18] WINIARSKA A, KNYSAK M, NABRDALIK K, et al. Inflammation and Oxidative Stress in Diabetic Kidney Disease: The Targets for SGLT2 Inhibitors and GLP-1 Receptor Agonists. Int J Mol Sci. 2021;22(19): 10822.
[19] VICTOR P, UMAPATHY D, GEORGE L, et al. Crosstalk between endoplasmic reticulum stress and oxidative stress in the progression of diabetic nephropathy. Cell Stress Chaperones. 2021;26(2):311-321.
[20] 于生友.PTEN介导的PINK1/Parkin信号通路在肾小球足细胞线粒体功能障碍中的作用[D].广州:南方医科大学,2021.
[21] PODGÓRSKI P, KONIECZNY A, LIS Ł, et al. Glomerular podocytes in diabetic renal disease. Adv Clin Exp Med. 2019;28(12):1711-1715.
[22] DING N, XIE L, MA F, et al. miR-30a-5p promotes glomerular podocyte apoptosis via DNMT1-mediated hypermethylation under hyperhomocysteinemiaa. Acta Biochim Biophys Sin (Shanghai). 2022; 54(2):1-11.
[23] KE G, CHEN X, LIAO R, et al. Receptor activator of NF-κB mediates podocyte injury in diabetic nephropathy. Kidney Int. 2021;100(2):377-390.
[24] JIN J, SHI Y, GONG J, et al. Exosome secreted from adipose-derived stem cells attenuates diabetic nephropathy by promoting autophagy flux and inhibiting apoptosis in podocyte. Stem Cell Res Ther. 2019;10(1):95.
[25] 刘泉,邵倞琦,尹卫东.白术多糖通过影响miR-320c调控脂联素受体1的表达保护高糖诱导的肾小球足细胞损伤[J].中国老年学杂志,2021,41(9):1947-1952.
[26] SUN F, JIANG D, CAI J. Effects of valsartan combined with α-lipoic acid on renal function in patients with diabetic nephropathy: a systematic review and meta-analysis. BMC Endocr Disord. 2021;21(1):178.
[27] 邱兵,高霞,崔维静,等.地塞米松对阿霉素诱导的肾小球足细胞活动力的影响[J].四川大学学报(医学版),2019,50(2):197-202.
[28] LI C, SUN R, CHEN J, et al. Different training patterns at recovery stage improve cognitive function in ischemic stroke rats through regulation of the axonal growth inhibitor pathway. Behav Brain Res. 2022;421:113730.
[29] 陈林娜,张鹏,王汉民.DNA甲基化与糖尿病肾病的研究进展[J].医学研究杂志,2019,48(12):11-13+132.
[30] TEH YM, MUALIF SA, LIM SK. A comprehensive insight into autophagy and its potential signaling pathways as a therapeutic target in podocyte injury. Int J Biochem Cell Biol. 2022;143:106153.
[31] EL-LATEEF AEA, EL-SHEMI AGA, ALHAMMADY MS, et al. LncRNA NEAT2 Modulates Pyroptosis of Renal Tubular Cells Induced by High Glucose in Diabetic Nephropathy (DN) by via miR-206 Regulation. Biochem Genet. 2022. doi: 10.1007/s10528-021-10164-6.
[32] 左袁博教.地塞米松联合黄芪甲苷对足细胞损伤的保护作用机制[D].衡阳:南华大学,2018.
[33] 敖立云,谢艳云.氧化应激介导糖尿病肾病足细胞损伤机制的研究进展[J].中南大学学报(医学版),2021,46(12):1403-1408.
[34] SU WY, LI Y, CHEN X, et al. Ginsenoside Rh1 Improves Type 2 Diabetic Nephropathy through AMPK/PI3K/Akt-Mediated Inflammation and Apoptosis Signaling Pathway. Am J Chin Med. 2021;49(5):1215-1233.
[35] 蒯巧林,于然,王维,等.白藜芦醇对嘌呤霉素氨基核苷诱导的足细胞氧化应激损伤的保护作用[J].江苏医药,2021,47(9):865-868+ 860.
[36] AMIN F, MEMARZIA A, RAD HK, et al. Retraction Note: Carvacrol and PPARγ agonist, pioglitazone, affects inhaled paraquat-induced lung injury in rats. Sci Rep. 2022;12(1):1226.
[37] 王婷,冯琦,龙喜国.基于Sirt1/PPARγ途径探究没食子酸对肾病综合征大鼠肾功能与足细胞损伤的影响[J].实用药物与临床,2021, 24(7):591-596.
[38] 姚翡,郁秋荣,龚叶,等.缬沙坦联合他汀类治疗糖尿病伴冠心病患者疗效及其对PPAR-γ的影响[J].疑难病杂志,2018,17(7):663-666.
|