[1] AL-JEFRI M, LEE J, JAMES M. Predicting acute kidney injury after surgery. Annu Int Conf IEEE Eng Med Biol Soc. 2020;2020:5606-5609.
[2] HOSTE EAJ, KELLUM JA, SELBY NM, et al. Global epidemiology and outcomes of acute kidney injury. Nat Rev Nephrol. 2018;14(10):607-625.
[3] SAEEDI P, PETERSOHN I, SALPEA P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res Clin Pract. 2019;157:107843.
[4] SAEEDI A, BAGHESTANI A, HASHEMI-NAZARI SS, et al. Prediction of mortality incidence in patients with chronic kidney disease based on influential prognostic factors with competing risks approach. Galen Med J. 2020;9:e1798.
[5] LI JS, LI B. Renal injury repair: how about the role of stem cells. Adv Exp Med Biol. 2019;1165:661-670.
[6] MIANEHSAZ E, MIRZAEI HR, MAHJOUBIN-TEHRAN M, et al. Mesenchymal stem cell-derived exosomes: a new therapeutic approach to osteoarthritis? Stem Cell Res Ther. 2019;10(1):340.
[7] MENDT M, REZVANI K, SHPALL E. Mesenchymal stem cell-derived exosomes for clinical use. Bone Marrow Transplant. 2019;54(Suppl 2): 789-792.
[8] XUNIAN Z, KALLURI R. Biology and therapeutic potential of mesenchymal stem cell-derived exosomes. Cancer Sci. 2020;111(9): 3100-3110.
[9] NIKFARJAM S, REZAIE J, ZOLBANIN NM, et al. Mesenchymal stem cell derived-exosomes: a modern approach in translational medicine. J Transl Med. 2020;18(1):449.
[10] ELSHARKASY OM, NORDIN JZ, HAGEY DW, et al. Extracellular vesicles as drug delivery systems: Why and how? Adv Drug Deliv Rev. 2020;159: 332-343.
[11] SHAH R, PATEL T, FREEDMAN JE. Circulating extracellular vesicles in human disease. N Engl J Med. 2018;379(10):958-966.
[12] VINAIPHAT A, SZE SK. Advances in extracellular vesicles analysis. Adv Clin Chem. 2020;97:73-116.
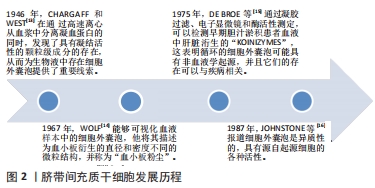
[13] CHARGAFF E, WEST R. The biological significance of the thromboplastic protein of blood. J Biol Chem. 1946;166(1):189-197.
[14] WOLF P. The nature and significance of platelet products in human plasma. Br J Haematol. 1967;13(3):269-288.
[15] DE BROE ME, BORGERS M, WIEME RJ. The separation and characterization of liver plasma membrane fragments circulating in the blood of patients with cholestasis. Clin Chim Acta. 1975;59(3):369-372.
[16] JOHNSTONE RM, ADAM M, HAMMOND JR, et al. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem. 1987;262(19): 9412-9420.
[17] HADE MD, SUIRE CN, SUO Z. Mesenchymal stem cell-derived exosomes: applications in regenerative medicine. Cells. 2021;10(8):1959.
[18] LI YJ, WU JY, WANG JM, et al. Emerging strategies for labeling and tracking of extracellular vesicles. J Control Release. 2020;328:141-159.
[19] BIDARIMATH M, LINGEGOWDA H, MILLER JE, et al. Insights into extracellular vesicle/exosome and mirna mediated bi-directional communication during porcine pregnancy. Front Vet Sci. 2021;8: 654064.
[20] HEO JS, LIM JY, YOON DW, et al. Exosome and melatonin additively attenuates inflammation by Transferring miR-34a, miR-124, and miR-135b. Biomed Res Int. 2020;2020:1621394.
[21] KELLER S, RIDINGER J, RUPP AK, et al. Body fluid derived exosomes as a novel template for clinical diagnostics. J Transl Med. 2011;9:86.
[22] RUSSELL AE, SNEIDER A, WITWER KW, et al. Biological membranes in EV biogenesis, stability, uptake, and cargo transfer: an ISEV position paper arising from the ISEV membranes and EVs workshop. J Extracell Vesicles. 2019;8(1):1684862.
[23] SOEKMADJI C, LI B, HUANG Y, et al. The future of Extracellular Vesicles as Theranostics-an ISEV meeting report. J Extracell Vesicles. 2020;9(1): 1809766.
[24] RAPOSO G, STOORVOGEL W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200(4):373-383.
[25] LOPEZ-VERRILLI MA, COURT FA. Exosomes: mediators of communication in eukaryotes. Biol Res. 2013;46(1):5-11.
[26] LABERGE A, ARIF S, MOULIN VJ. Microvesicles: intercellular messengers in cutaneous wound healing. J Cell Physiol. 2018;233(8):5550-5563.
[27] XU X, LAI Y, HUA ZC. Apoptosis and apoptotic body: disease message and therapeutic target potentials. Biosci Rep. 2019;39(1):BSR20180992.
[28] SALUNKHE S, DHEERAJ, BASAK M, et al. Surface functionalization of exosomes for target-specific delivery and in vivo imaging & tracking: strategies and significance. J Control Release. 2020;326:599-614.
[29] HA D, YANG N, NADITHE V. Exosomes as therapeutic drug carriers and delivery vehicles across biological membranes: current perspectives and future challenges. Acta Pharm Sin B. 2016;6(4):287-296.
[30] RAULF N, LUCARELLI P, THAVARAJ S, et al. Annexin A1 regulates EGFR activity and alters EGFR-containing tumour-derived exosomes in head and neck cancers. Eur J Cancer. 2018;102:52-68.
[31] RIEGER AM, NELSON KL, KONOWALCHUK JD, et al. Modified annexin V/propidium iodide apoptosis assay for accurate assessment of cell death. J Vis Exp. 2011;(50):2597.
[32] DENG H, SUN C, SUN Y, et al. Lipid, Protein, and microrna composition within mesenchymal stem cell-derived exosomes. Cell Reprogram. 2018;20(3):178-186.
[33] OSTI D, DEL BENE M, RAPPA G, et al. Clinical significance of extracellular vesicles in plasma from glioblastoma patients. Clin Cancer Res. 2019; 25(1):266-276.
[34] BHATTA B, COOKS T. Reshaping the tumor microenvironment: extracellular vesicles as messengers of cancer cells. Carcinogenesis. 2020;41(11):1461-1470.
[35] QIN ZH, XU JF, QU JM, et al. Intrapleural delivery of MSCs attenuates acute lung injury by paracrine/endocrine mechanism. J Cell Mol Med. 2012;16(11):2745-2753.
[36] CAMUSSI G, DEREGIBUS MC, TETTA C. Paracrine/endocrine mechanism of stem cells on kidney repair: role of microvesicle-mediated transfer of genetic information. Curr Opin Nephrol Hypertens. 2010;19(1):7-12.
[37] LIU B, DING FX, LIU Y, et al. Human umbilical cord-derived mesenchymal stem cells conditioned medium attenuate interstitial fibrosis and stimulate the repair of tubular epithelial cells in an irreversible model of unilateral ureteral obstruction. Nephrology (Carlton). 2018;23(8):728-736.
[38] MA Y, DONG L, ZHOU D, et al. Extracellular vesicles from human umbilical cord mesenchymal stem cells improve nerve regeneration after sciatic nerve transection in rats. J Cell Mol Med. 2019;23(4):2822-2835.
[39] XU H, WANG Z, LIU L, et al. Exosomes derived from adipose tissue, bone marrow, and umbilical cord blood for cardioprotection after myocardial infarction. J Cell Biochem. 2020;121(3):2089-2102.
[40] WANG ZG, HE ZY, LIANG S, et al. Comprehensive proteomic analysis of exosomes derived from human bone marrow, adipose tissue, and umbilical cord mesenchymal stem cells. Stem Cell Res Ther. 2020; 11(1):511.
[41] BÖRGER V, BREMER M, FERRER-TUR R, et al. Mesenchymal stem/stromal cell-derived extracellular vesicles and their potential as novel immunomodulatory therapeutic agents. Int J Mol Sci. 2017;18(7):1450.
[42] LAI RC, TAN SS, TEH BJ, et al. Proteolytic potential of the MSC exosome proteome: implications for an exosome-mediated delivery of therapeutic proteasome. Int J Proteomics. 2012;2012:971907.
[43] BURGOS-RAVANAL R, CAMPOS A, DÍAZ-VESGA MC, et al. Extracellular vesicles as mediators of cancer disease and as nanosystems in theranostic applications. Cancers (Basel). 2021;13(13):3324.
[44] LEVEY AS, JAMES MT. Acute kidney injury. Ann Intern Med. 2017;167(9): ITC66-ITC80.
[45] BRUNO S, GRANGE C, DEREGIBUS MC, et al. Mesenchymal stem cell-derived microvesicles protect against acute tubular injury. J Am Soc Nephrol. 2009;20(5):1053-1067.
[46] REIS LA, BORGES FT, SIMÕES MJ, et al. Bone marrow-derived mesenchymal stem cells repaired but did not prevent gentamicin-induced acute kidney injury through paracrine effects in rats. PLoS One. 2012;7(9):e44092.
[47] BRUNO S, GRANGE C, COLLINO F, et al. Microvesicles derived from mesenchymal stem cells enhance survival in a lethal model of acute kidney injury. PLoS One. 2012;7(3):e33115.
[48] ZHU G, PEI L, LIN F, et al. Exosomes from human-bone-marrow-derived mesenchymal stem cells protect against renal ischemia/reperfusion injury via transferring miR-199a-3p. J Cell Physiol. 2019;234(12):23736-23749.
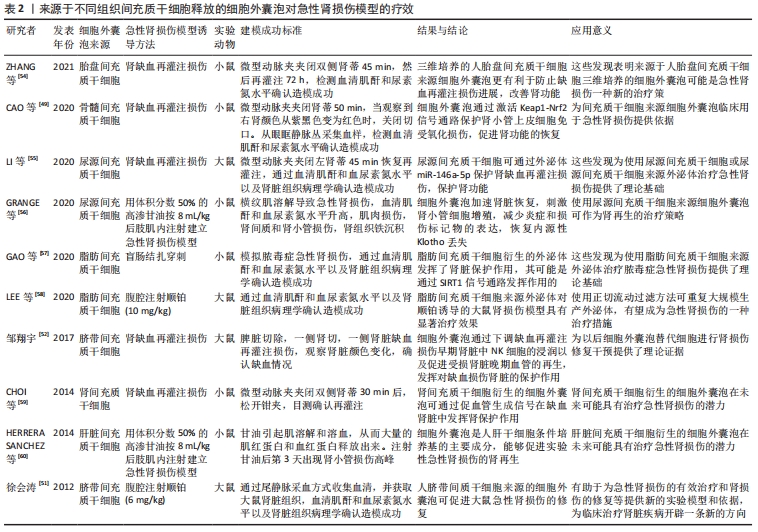
[49] CAO H, CHENG Y, GAO H, et al. In vivo tracking of mesenchymal stem cell-derived extracellular vesicles improving mitochondrial function in renal ischemia-reperfusion injury. ACS Nano. 2020;14(4):4014-4026.
[50] ZHANG ZY, HOU YP, ZOU XY, et al. Oct-4 enhanced the therapeutic effects of mesenchymal stem cell-derived extracellular vesicles in acute kidney injury. Kidney Blood Press Res. 2020;45(1):95-108.
[51] 徐会涛.脐带间质干细胞来源的囊泡对顺铂诱导的急性肾损伤修复的实验研究[D].镇江:江苏大学,2012.
[52] 邹翔宇.细胞外囊泡减轻大鼠缺血再灌注肾损伤的实验研究[D].上海:上海交通大学,2017.
[53] KILPINEN L, IMPOLA U, SANKKILA L, et al. Extracellular membrane vesicles from umbilical cord blood-derived MSC protect against ischemic acute kidney injury, a feature that is lost after inflammatory conditioning. J Extracell Vesicles. 2013. doi: 10.3402/jev.v2i0.21927.
[54] ZHANG X, WANG N, HUANG Y, et al. Extracellular vesicles from three dimensional culture of human placental mesenchymal stem cells ameliorated renal ischemia/reperfusion injury. Int J Artif Organs. 2021. doi: 10.1177/0391398820986809.
[55] LI X, LIAO J, SU X, et al. Human urine-derived stem cells protect against renal ischemia/reperfusion injury in a rat model via exosomal miR-146a-5p which targets IRAK1. Theranostics. 2020;10(21):9561-9578.
[56] GRANGE C, PAPADIMITRIOU E, DIMUCCIO V, et al. Urinary extracellular vesicles carrying klotho improve the recovery of renal function in an acute tubular injury model. Mol Ther. 2020;28(2):490-502.
[57] GAO F, ZUO B, WANG Y, et al. Protective function of exosomes from adipose tissue-derived mesenchymal stem cells in acute kidney injury through SIRT1 pathway. Life Sci. 2020;255:117719.
[58] LEE JH, HA DH, GO HK, et al. Reproducible large-scale isolation of exosomes from adipose tissue-derived mesenchymal stem/stromal cells and their application in acute kidney injury. Int J Mol Sci. 2020; 21(13):4774.
[59] CHOI HY, MOON SJ, RATLIFF BB, et al. Microparticles from kidney-derived mesenchymal stem cells act as carriers of proangiogenic signals and contribute to recovery from acute kidney injury. PLoS One. 2014;9(2):e87853.
[60] HERRERA SANCHEZ MB, BRUNO S, GRANGE C, et al. Human liver stem cells and derived extracellular vesicles improve recovery in a murine model of acute kidney injury. Stem Cell Res Ther. 2014;5(6):1639.
[61] GARLA V, KANDURI S, YANES-CARDOZO L, et al. Management of diabetes mellitus in chronic kidney disease. Minerva Endocrinol. 2019; 44(3):273-287.
[62] BUYADAA O, MAGLIANO DJ, SALIM A, et al. Risk of rapid kidney function decline, all-cause mortality, and major cardiovascular events in nonalbuminuric chronic kidney disease in type 2 diabetes. Diabetes Care. 2020;43(1):122-129.
[63] STEPHENS JW, BROWN KE, MIN T. Chronic kidney disease in type 2 diabetes: Implications for managing glycaemic control, cardiovascular and renal risk. Diabetes Obes Metab. 2020;22 Suppl 1:32-45.
[64] JIANG ZZ, LIU YM, NIU X, et al. Exosomes secreted by human urine-derived stem cells could prevent kidney complications from type I diabetes in rats. Stem Cell Res Ther. 2016;7:24.
[65] NAGAISHI K, MIZUE Y, CHIKENJI T, et al. Mesenchymal stem cell therapy ameliorates diabetic nephropathy via the paracrine effect of renal trophic factors including exosomes. Sci Rep. 2016;6:34842.
[66] WANG Y, GUO YF, FU GP, et al. Protective effect of miRNA-containing extracellular vesicles derived from mesenchymal stromal cells of old rats on renal function in chronic kidney disease. Stem Cell Res Ther. 2020;11(1):274.
[67] KHOLIA S, HERRERA SANCHEZ MB, CEDRINO M, et al. Human liver stem cell-derived extracellular vesicles prevent aristolochic acid-induced kidney fibrosis. Front Immunol. 2018;9:1639.
[68] 陈晶,刘清岩,张海宁,等.脂肪干细胞来源的外泌体对2型糖尿病肾病大鼠的保护作用[J].解剖科学进展,2020,26(4):369-373.
[69] CHOI HY, KIM TY, LEE M, et al. Kidney mesenchymal stem cell-derived extracellular vesicles engineered to express erythropoietin improve renal anemia in mice with chronic kidney disease. Stem Cell Rev Rep. 2021. doi: 10.1007/s12015-021-10141-x.
[70] GREGORINI M, CORRADETTI V, PATTONIERI EF, et al. Perfusion of isolated rat kidney with mesenchymal stromal cells/extracellular vesicles prevents ischaemic injury. J Cell Mol Med. 2017;21(12):3381-3393.
[71] WU X, YAN T, WANG Z, et al. Micro-vesicles derived from human Wharton’s Jelly mesenchymal stromal cells mitigate renal ischemia-reperfusion injury in rats after cardiac death renal transplantation. J Cell Biochem. 2018;119(2):1879-1888.
[72] NASSAR W, EL-ANSARY M, SABRY D, et al. Umbilical cord mesenchymal stem cells derived extracellular vesicles can safely ameliorate the progression of chronic kidney diseases. Biomater Res. 2016;20:21.
[73] DOOLEY K, MCCONNELL RE, XU K, et al. A versatile platform for generating engineered extracellular vesicles with defined therapeutic properties. Mol Ther. 2021;29(5):1729-1743.
[74] LAMICHHANE TN, JAY SM. Production of extracellular vesicles loaded with therapeutic cargo. Methods Mol Biol. 2018;1831:37-47.
[75] MELLING GE, CAROLLO E, CONLON R, et al. The challenges and possibilities of extracellular vesicles as therapeutic vehicles. Eur J Pharm Biopharm. 2019;144:50-56.
[76] VAN DER MEEL R, FENS MH, VADER P, et al. Extracellular vesicles as drug delivery systems: lessons from the liposome field. J Control Release. 2014;195:72-85.
[77] O’BRIEN K, BREYNE K, UGHETTO S, et al. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat Rev Mol Cell Biol. 2020;21(10):585-606.
[78] NASIRI KENARI A, CHENG L, HILL AF. Methods for loading therapeutics into extracellular vesicles and generating extracellular vesicles mimetic-nanovesicles. Methods. 2020;177:103-113.
[79] POMATTO MAC, BUSSOLATI B, D’ANTICO S, et al. Improved loading of plasma-derived extracellular vesicles to encapsulate antitumor miRNAs. Mol Ther Methods Clin Dev. 2019;13:133-144.
[80] TAO SC, RUI BY, WANG QY, et al. Extracellular vesicle-mimetic nanovesicles transport LncRNA-H19 as competing endogenous RNA for the treatment of diabetic wounds. Drug Deliv. 2018;25(1):241-255.
[81] LAINŠČEK D, KADUNC L, KEBER MM, et al. Delivery of an artificial transcription regulator dcas9-vpr by extracellular vesicles for therapeutic gene activation. ACS Synth Biol. 2018;7(12):2715-2725.
[82] JIN J, QIAN F, ZHENG D, et al. Mesenchymal stem cells attenuate renal fibrosis via exosomes-mediated delivery of microRNA Let-7i-5p antagomir. Int J Nanomedicine. 2021;16:3565-3578.
|