中国组织工程研究 ›› 2022, Vol. 26 ›› Issue (3): 446-455.doi: 10.12307/2022.074
• 骨与关节综述 bone and joint review • 上一篇 下一篇
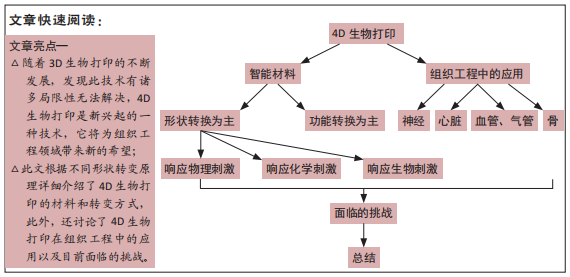
4D生物打印在组织工程的应用
关 健1,2,贾燕飞2,张葆鑫2,赵国中1,2
- 1内蒙古医科大学,内蒙古自治区呼和浩特市 010110;2内蒙古医科大学第二附属医院,内蒙古自治区呼和浩特市 010030
-
收稿日期:2021-02-25修回日期:2021-02-27接受日期:2021-04-17出版日期:2022-01-28发布日期:2021-10-29 -
通讯作者:贾燕飞,主任医师,内蒙古医科大学第二附属医院,内蒙古自治区呼和浩特市 010030 -
作者简介:关健,男,1993年生,内蒙古自治区通辽市人,蒙古族,内蒙古医科大学在读硕士,医师,主要从事骨科学方面的研究。
Application of 4D bioprinting in tissue engineering
Guan Jian1, 2, Jia Yanfei2, Zhang Baoxin 2, Zhao Guozhong1, 2
- 1Inner Mongolia Medical University, Hohhot 010110, Inner Mongolia Autonomous Region, China; 2Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China
-
Received:2021-02-25Revised:2021-02-27Accepted:2021-04-17Online:2022-01-28Published:2021-10-29 -
Contact:Jia Yanfei, Chief physician, Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China -
About author:Guan Jian, Master candidate, Physician, Inner Mongolia Medical University, Hohhot 010110, Inner Mongolia Autonomous Region, China; Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China
摘要:
文题释义:
4D生物打印:4D打印本质上是一种三维打印技术,其中包含的第四维被称为“时间”,这项技术需要具有随时间变化的多材料打印能力,或者需要一种可以从一种形状改变为另一种形状的定制材料系统。对于 4D 打印一个较为全面的定义是:一个三维印刷结构被暴露于预定刺激下(如温度、水、光、pH值等),其功能、形状、性能可随时间发生变化。因此,与 3D 打印技术相比,4D 打印最大的突破在于其随时间变化的能力,而4D生物打印则是打印材料及打印产品具有生物相容性。
智能材料:泛指将传感元件、驱动元件以及有关的信号处理和控制电路集成在材料结构中,通过机、热、光、化、电、磁等刺激和控制,使其不仅具有承受载荷的能力,而且具有识别、分析、处理及控制等多种功能,能进行自诊断、自适应、自学习及自修复。
背景:随着科技的不断发展3D生物打印已经成为组织工程学的重点研究领域,然而目前仍存在一些局限性无法解决。4D生物打印是最新兴起的一种技术,它成为了组织工程下一代的主要解决手段。
目的:根据不同形状转变原理而分别介绍4D生物打印的材料和方式,探讨4D生物打印在组织工程中的应用以及目前面临的挑战。
方法:以“4D bioprinting,4D biofabriation,printing,smart materials,smart scaffold,shape memory polymers,tissue engineering”或“4D生物打印,4D生物制造,打印,智能材料,智能支架,形状记忆聚合物,组织工程”等作为检索词,应用互联网在CNKI、PubMed、SCIE数据库检索2016-01-01/2021-01-01发表的相关文献共127篇,经过第一作者筛除并追加收录优质参考文献,共纳入106篇文章进行综述分析。
结果与结论:①4D生物打印是最新兴起的一种技术,它将时间的概念和3D生物打印相结合作为第四维度,4D生物打印能够制造复杂的具有功能性的结构;②它可以通过使用智能材料制作动态的三维生物结构,这些结构可以在各种刺激下改变形状;③印刷细胞结构的功能转变和成熟也被认为是4D生物打印其中的一种形式;④该技术为组织工程提供了前所未有的潜力,虽然该技术在生物医学领域备受瞩目,但由于其尚处于兴起阶段,若想实现临床应用还需要更多的研究和发展。
https://orcid.org/0000-0001-8034-5954 (关健)
中国组织工程研究杂志出版内容重点:人工关节;骨植入物;脊柱;骨折;内固定;数字化骨科;组织工程
中图分类号:
引用本文
关 健, 贾燕飞, 张葆鑫, 赵国中. 4D生物打印在组织工程的应用[J]. 中国组织工程研究, 2022, 26(3): 446-455.
Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455.
有多种方式能使打印出的原始结构变形,包括手动变形或细胞牵引力,也可以利用刺激智能材料来实现形状的变化。细胞牵引力在生物科学当中最关键的作用,是可以调节细胞和组织,如伤口愈合、血管生成、转移、炎症[24]。之后基于细胞牵引力开发出“细胞折叠技术”,在这种技术中,细胞通过将二维元素折叠成预定义的三维形状[6,25],但是此类转化方法的缺点为难以精确控制打印体的折叠[26]。因此,为追求更加精细的受控制形状转换,刺激响应型生物材料已经在很大程度上取代了基于细胞牵引力的打印方式。刺激响应材料可以在特定的条件下发生构象变化,如温度、pH值、湿度、电、磁场、光、声学或这些刺激的互相组合[27]。在接下来的部分中,此文将介绍几种潜在的响应刺激智能材料,包括它们的自组装或形状记忆能力,以及它们在4D生物打印中的潜在应用。根据激发4D过程的刺激类型,这些智能材料可分为物理、化学和生物响应刺激材料。
2.1.1 响应物理刺激智能材料
(1)响应温度的刺激材料:温度是生物打印结构中实现形状变形最常用的物理刺激[28]。当环境温度低于溶解临界温度时,该聚合物转变为液相;而当温度高于临界温度时,聚合物会变为凝胶状态。响应温度刺激的材料有聚(N-异丙基丙烯酰胺)、聚(乙烯基己内酰胺)、明胶、胶原和聚乙二醇等等,以及一种特殊的材料,称为形状记忆聚合物,包括聚(ε-己内酯)二甲基丙烯酸酯、聚己内酯三醇、豆油环氧化丙烯酸酯、聚醚氨酯和聚乳酸;以上两类材料是4D生物打印中最常见的温敏型聚合物[29]。
形状记忆聚合物属于智能聚合物,可以同时响应物理和化学刺激,而化学刺激主要受pH值影响[30-31]。当施加此类刺激后可以变换成“记忆”的形状,还可以再经过外部刺激变换回最初始的形状,即使“记忆”的形状与刺激之前的原始打印形状完全不同[31-32]。例如最近开发出的载细胞形状记忆水凝胶,就可以根据需要实现其形状和功能的可逆变化[33],而且它改善了固态形状记忆聚合物的弊端,使细胞不仅只可以置入打印材料的表面,还可以均匀地分布在打印材料的内部。形状记忆聚合物形状记忆的方式是通过聚合物中的亲水链段与水发生强烈的相互作用,从而使材料的体积膨胀并形成水凝胶;当聚合物链段根据环境条件进行可逆水化时,可以实现智能水凝胶,这种水凝胶能够通过收缩和膨胀等方式,从而改变其体积。目前正在进行不改变水凝胶体积的研究,它可以通过形状记忆效应实现水凝胶的空间定向驱动。形状记忆效应包括样品的弹性变形,这些样品通过可逆的共价或物理交联暂时固定,从而形成临时形状[33];当然水和光也可以刺激形状记忆聚合物的响应,但温度对其刺激响应的研究更加广泛。形状记忆聚合物既有优点也有缺点,其优点包括低密度、易于形状控制和对恢复温度的良好控制、高应变恢复率以及实现所需性能(例如生物降解性)的物理和化学刺激能力;缺点是强度差、弹性模量和加工温度较低[16]。
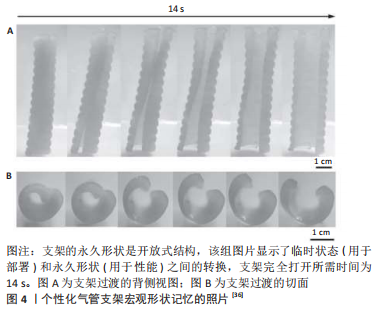
通常应用形状记忆材料进行打印时,将打印模型放在适当的条件下,使其处于亚稳态的临时形状。随后在施加外界刺激过程中逐步恢复,当施加足够的变换刺激时,临时变形的结构将变换成所需的形状,这种形状恢复特性对于治疗小骨缺损植入方式有良好的效果[34]。如SENATOV等[35]研究所示,在80 ℃压缩聚乳酸/质量分数为15%的羟基磷灰石支架后,可恢复98%的预设记忆形状,见图3。ZAREK等[36]也使用类似的方式,制造了热驱动甲基丙烯酸酯聚己内酯气管支架,见图4,该支架安放到体内气管部分后充分膨胀以适应气管,此过程既可以做到无创又可以防止在插入过程中造成伤害。

温度响应性材料是应用最广泛的转换方式,其中一种经典的温度响应材料是聚(N-异丙基丙烯酰胺)[10,28,37]。例如APSITE等[38]的研究中使用温度响应性聚己内酯和聚(N-异异丙基丙烯酰胺)制备自折叠多层多孔电纺支架,这些以聚(N-异异丙基丙烯酰胺)为基础的温度响应材料为4D生物打印提供了无限可能,但它们仍然存在一些限制,如生物相容性差、疏水性和不可降解性[30]。最重要的一点是,聚(N-异异丙基丙烯酰胺)的形状转换需要较高的温度刺激,若将其应用于4D生物打印会导致细胞功能不足甚至细胞失活。为解决这一问题有研究通过与生物活性肽结合来改善细胞黏附和生长,并且可以将刺激温度降低到人体37 ℃左右来保持细胞活力[6]。而且APSITE等[38]经过改进后,在37 ℃的水环境中支架也可以自发卷曲成管状结构,且内层为聚己内酯适合细胞包埋,这为4D生物打印提供了新的选择。
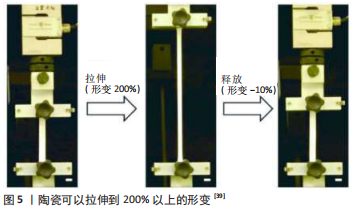
还有研究提出了一种新的方法,该研究团队利用墨水直接打印变形热处理方法打印出陶瓷临时状态,该状态储藏了陶瓷的弹性能量,通过释放该能量可以将陶瓷拉伸到原始形状的200%以上[39],见图5。虽然该实验展现了良好的优势,但是由于变形机械设施是双轴拉伸装置,因此打印陶瓷的重要缺点就是形变的精确度难以掌控,在未来随着高精度技术的引入,变形的精度有望提高。
(2)响应湿度的刺激材料:湿敏型材料是通过湿度的变化而膨胀和收缩来改变其形状和体积的[40]。该材料仅是4D打印早期选用的类型,虽然湿敏材料已经应用于4D生物打印,但是需要注意的是湿敏材料的膨胀和收缩程度在转换过程中需要精确控制,而细胞的培养应保持在恒定的高湿度和渗透压下,由于生物体内湿度和渗透压的限制,形状变换的程度也是受限的。
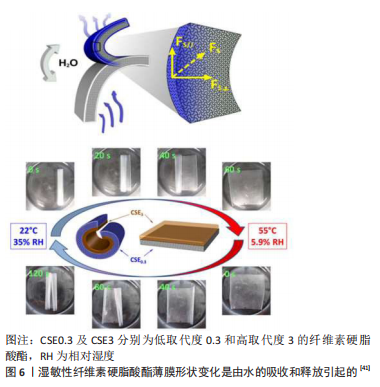
纤维素硬脂酸酯(cellulose stearoly esters,CSEs)用于制造对水分敏感的自立薄膜,由于水分子的疏水解吸,薄膜可以折叠或展开以呈现有节奏的弯曲运动,制造具有疏水表面的湿敏双层薄膜,这种双层膜在溶液中可以进行快速的可逆弯曲运动和连续的形状转变[41]。该实验制备了一种由低取代度0.3(CSE0.3)和高取代度3(CSE3)的纤维素硬脂酸酯组成的双层膜,当CSE0.3一侧暴露于湿度时,其表面吸收水分子并产生水平和垂直膨胀力;相反,CSE3具有疏水性和温度响应性,这样双层膜便具有亲水和疏水两种表面。最初CSE0.3/CSE3双层膜保持在55 ℃和5.9%的相对湿度,以实现两层的稳定状态,并将膜放置于稳定的平面;随后,当温度降低至22 ℃,相对湿度增加至35%,双层膜开始从CSE3侧弯曲,略微收缩,随后CSE0.3层由于膨胀而卷起,最终形成薄膜的紧密卷;当再次将双层膜放置于55 ℃和5.9%相对湿度的最初环境中时,双层膜便迅速恢复最初的稳定状态,见图6。当然这是4D打印技术的最早研究方式,多层结构概念也引导了4D生物打印的未来设计。然而,细胞培养环境中持续的高湿度(即在培养基中)将对材料的转化能力限制为一次,这些人体内湿度和渗透压的限制,可以通过调节湿敏材料的灵敏度来解决,这样湿敏材料便可以在细胞耐受性范围内迅速转化。
(3)响应电刺激材料:响应电刺激材料通常是聚电解质聚合物以膨胀、收缩、折叠或弯曲来响应电,而且形状的变换可以通过电场的方向和强度来调节[42],这种响应电场材料已经用于生物医学领域,如药物输送、组织工程电响应组织[43]。一些含有导电聚合物的水凝胶,如聚吡咯、聚苯胺和聚噻吩,可以表现出良好的生物相容性,为4D生物打印提供了巨大的潜力[44]。
近年来,具有电响应性的碳基纳米生物材料如石墨烯和碳纳米管,被用于制造4D生物打印的响应电刺激材料,它们具有独特的机械性能、可调节的表面和良好的导电性。碳纳米管是一类既具有优异导电性又具有生物相容性的材料,SHIN等[45]展示了利用碳纳米管材料打印出的二维结构并经过刺激形状转换成三维形状。还有研究展示,用形状记忆聚合物Veriflex?涂覆于1.8 g碳纳米管纳米纸表面,在0.09 A的电流作用下,该形状记忆复合材料形状得以恢复[46],见图7。该研究中扁平形状记忆试样被弯曲成“U”形的临时形状,但在电流释放后的120 s内恢复到其原始形状,此过程证实了碳纳米管可以显著提高形状记忆复合材料的电导率和电活性响应速度,表明电响应材料可能会改善其他材料的电学性能,并在形状变换或记忆方面有应用价值。

此外石墨烯也可以成为4D生物打印的基础材料。例如,SERVANT等[47]开发了一种以石墨烯为材料的大孔支架,它具有增强的机械性能、响应电场能力和热性能,在这篇文献中表明,当施加低电压时,药物可以从石墨烯支架中释放出来。而且,含有石墨烯材料的支架支持人骨髓间充质干细胞向神经源性分化,具有特殊的操作特性,可以在手术中操作,是神经再生的潜在平台[34]。
除了使用电活性材料外,另一种方法是利用在电刺激下可以驱动的细胞结构来实现运动。电场能够将细胞遥控到预定的位置或将细胞引导到特定方向,这些现象分别称为趋电性和电向性[34]。CVETKOVIC 等[48]的工作证明了这一概念,该研究使用了在立体平版3D打印的聚乙二醇双丙烯酸酯水凝胶上培养的充满细胞的骨骼肌条,在实现功能性集成细胞系统方面向前迈进了一步,在可编程肌肉组织工程和仿生机设计方面具有广泛的应用前景。
总体而言,电刺激材料给肌肉或神经等组织再生带来了无限的可能[49]。然而,当施加的电流过大时,可能会出现局部加热、膜破裂或细胞死亡等问题,因此在使用电场作为刺激时,应采取预防措施和精确操控。
(4)响应磁场刺激材料:响应磁场刺激材料有磁性微粒和纳米颗粒,磁性纳米颗粒由铁(Fe)、钴(Co)、镍(Ni)或它们的氧化物等磁性元素组成,这些材料已广泛应用于生物医学领域,如控制药物释放和组织工程。例如,通过将Fe3O4纳米颗粒与聚乙二醇琼脂水凝胶相结合,可以制成响应磁场刺激的药物输送系统,这是软组织损伤治疗的一种有前途的治疗方案[50]。还有研究应用于软骨组织工程领域当中,研究员利用磁性的功能排列胶原纤维的生物打印新策略,该研究利用链霉亲和素包裹的铁纳米颗粒将其嵌入到具有不同浓度的低温度凝胶琼脂糖和Ⅰ型胶原的可打印生物墨水中。然后在水凝胶中直接应用磁性机制可以在最大琼脂质量浓度为0.5%的水凝胶混合物中排列胶原纤维;相反,琼脂糖浓度越高,胶原纤维的分布就越随机;结果显示,规律排列纤维和随机排列纤维交替层叠的打印结构明显表达出大量的Ⅱ型胶原[51]。纳米颗粒的顺磁性也可用于实现细胞或微组织的磁悬浮组装,SUN等[52]提出了一种将磁性海藻酸盐微纤维材料制造支架的创新方案来构建微血管样结构,这种材料允许细胞之间的直接相互作用,对于血管样结构的形成至关重要。还有研究使用一种细胞负载的明胶甲基丙烯酰水凝胶,浸泡在稳定的自由基溶液中时产生顺磁性,从而制造出可调谐的磁性自组装微凝胶[53]。
总之,这些创新的磁悬浮组装系统在复杂的细胞组装和组织工程方面显示出巨大的前景,为4D生物打印的应用提供了新的选择。但响应磁场刺激材料也存在一部分缺点,在生物细胞中使用纳米颗粒时,当从基质中浸出时,小于50 nm的纳米颗粒能够穿过生物膜,通过诱导炎症、产生活性氧、阻碍DNA的功能和诱导细胞凋亡而对组织的功能产生不利影响[16]。因此,响应磁场刺激性聚合物的生物相容性都与所使用的纳米颗粒的类型以及它们掺入聚合物的方法直接相关。另外,在应用之前应该使用适当的实验模型来评估响应磁场刺激材料在活体条件下的生物相容性,该模型应该考虑磁场强度、纳米颗粒的使用量以及这些纳米颗粒对外加磁场的响应性。
(5)响应光刺激材料:光响应材料可以捕获外部施加的光信号,并将其进行机械转换。光响应性生物材料可以在较宽的波长范围内被光激活,包括近红外、红外和紫外区域。红外辐射对活体组织的吸收率比紫外辐射低,因此红外的光毒性较低并且组织渗透性较高[54]。光刺激最早应用于4D打印时,只作为改变打印品的颜色的功能,后来光刺激也用来改变打印品的形状。
光响应材料最常见的响应机制是聚合物链的光异构化和光降解,这早已被广泛应用于4D打印,该类型的材料具有高应变形状记忆能力和自愈合功能,为4D生物打印技术的发展铺平了道路[10]。此外,NASEER等[55]向光响应水凝胶中添加光降解元素,例如香豆素或邻硝基苄基醚基团,是调节水凝胶生物降解速率的一种新思路。由于生物材料利用光的空间控制性,可以在细胞存在的情况下实现生物材料的光降解,为动态组织培养提供了有利条件。
LUO等[56]提出了一种简便的方法,使用基于藻酸盐生物材料创建具有近红外触发形状变形的细胞负载结构,使用浓缩海藻酸盐聚多巴胺材料制备双层支架在近红外诱导脱水过程中显示出折叠形状的变化。该支架能够保持其变形的结构,水凝胶中的印刷细胞在培养基中培养至少14 d仍保持较高的活性,使这种简便的方法在4D生物打印领域具有巨大的应用潜力。
由于多种光诱导剂的光毒性,限制了其在组织工程中的应用,而且光穿过生物组织时对光的吸收导致光强烈衰减也是一项艰难的挑战。幸运的是,可以通过尝试使用超红外光来解决,超红外光具有更低的活组织吸光度和光毒性,以及更有效的组织穿透。因此,响应光刺激的生物材料在4D生物打印当中更加吸引研究人员的关注。
(6)响应声刺激材料:超声波刺激该材料会引起物理或化学变化,响应声刺激材料已经应用于控制药物释放和组织工程。声波刺激是一种具有无限潜能的技术,它能以非接触的方式快速且精确地刺激细胞进行形状变换[30]。HUEBSCH等[57]研制了一种以藻酸盐为基础材料的水凝胶,这种材料可以受声波刺激控制,它被用于输送药物。此外,NASEER等[55]报道了一种利用表面声波使明胶甲基丙烯酰水凝胶内的细胞快速排列的技术,这种声波刺激能够以安全和无创的方式使细胞进行形态学改变。但是该材料目前还仅限制于只能以线性模式来变换,且只能构建同质细胞体,未来将需要更复杂的方法来构建复杂的异质组织细胞结构。
2.1.2 响应化学刺激智能材料
(1)响应pH刺激材料:响应pH刺激材料其结构中含有弱酸性或碱性基团的聚电解质,如羧基、吡啶、磺酸、磷酸盐等,当环境的pH值改变时,这些基团会释放或接受质子,从而导致结构或性质的变化,此类材料因其独特的性质已被用于药物输送、基因输送和葡萄糖传感器等生物医学应用。在pH响应材料中,碱性单体聚合物在酸性条件下表现为阳离子聚合物,酸性单体聚合物在碱性条件下表现为阴离子聚合物[16]。打印时可以在刺激响应材料中添加2种聚合物中的1种或2种聚合物的组合,pH响应型聚合物的来源可以是天然的,也可以是合成的;人工合成的pH响应型聚合物在生物打印的研究领域很少,而天然蛋白的应用受到了极大的关注[58-59]。例如,3种不同的电荷五肽序列被用来制造一种强健的pH响应型水凝胶[60],通过调节五肽序列的电荷分布和浓度可以显著调节水凝胶的力学性能;海藻酸钠也已广泛用于生物打印当中[61]。尽管在天然蛋白质方面进行了大量研究,但重点放在获得生物活性印刷结构以改善细胞的相互作用,而该类材料独特的pH响应性尚未解锁。
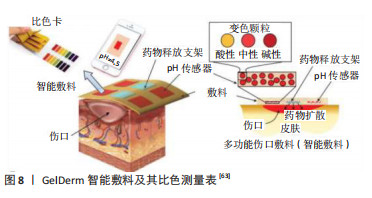
除了天然聚合物外,多数人工合成聚合物已经被用于药物传输领域。LARUSH等[62]打印丙烯酸基水凝胶以获得在高pH值环境下能够更快释放药物的片剂,在该项研究中制备的片剂显示出抗胃特性和pH响应性药物释放模式,使其成为一种很有前途的肠道给药系统。人工合成聚合物不仅应用于药物传输领域,其pH响应性在实现多功能敷料方面显示出非常的优势,例如新研制的GelDerm是一种智能敷料,见图8,它可以通过pH依赖性比色测量表提示细菌感染,并在伤口处释放抗生素制剂[63];该敷料以介孔树脂珠为载体,掺杂响应pH材料,通过对海藻酸纤维进行染色以获得比色特性。 总的来说,天然pH响应性聚合物在实际应用中受到其机械性能较差的限制,目前的方案是将天然聚合物与其他材料混合使用,形成具有足够结构完整性的混合结构。至于人工合成聚合物,尽管在探索其在生物医学方面的潜在用途方面已经建立了丰富的实验,但是该类材料在转变过程中会在细胞培养基中释放一些化合物,若将此类材料应用于生物打印中,释放的化合物一定不能影响细胞行为和功能[25],并且需要明确该材料的化学成分、生物降解性和长期结构稳定性。
(2) 响应离子刺激材料:有研究报道,已打印出坚硬的支架作为细胞负载结构[64]。一些多价离子Ca2+或Zn2+已经应用于生物打印,以获得具有合适强度和可调机械性能的支架[65-66],例如海藻酸盐和氯化钙之间钠钙离子交换的交联机制已应用于生物打印中[67],基于氢键-钙离子相互作用的水凝胶在可逆的钙离子作用下表现出形状记忆能力。此外,KIRILLOVA等[68]报道了一种先进的4D生物打印方法,该方法可用于海藻酸盐和透明质酸水凝胶中,海藻酸盐和透明质酸水凝胶具有可负载细胞、刺激触发、形状变换的特性,并用甲基丙烯酸酯基团对其进行修饰,使其具有响应光刺激性,该响应光水凝胶具有较强的钙离子浓度依赖性的特性。细胞可以耐受细胞外环境中钙离子的变化,使钙离子成为具有生物相容性的变形刺激,响应Ca2+刺激的方法是非常有研究价值的4D生物制造方法,因为材料具有生物相容性而且可生物降解,最重要的是细胞可以置于材料表面也可以掺杂在其中[30]。目前对响应离子浓度材料的研究相对较少,但仍需注意生理电解质水平也是各种疾病的关键指标,如血液中钙含量过高,便成为高钙血症。因此,若要将生物学与材料技术的最新进展结合起来,必须要研究材料对离子浓度变化的反应行为,这才能在未来的研究中获取更有效的治疗选择。
(3)响应生物刺激材料:在人体生理过程当中通常依赖于反馈调节,包括生物分子,如蛋白质、核酸、多肽和酶,因此除了通过离子交联过程形成凝胶外,水凝胶还可以结合可调的共价生物正交交联策略和特定肽折叠介导的相互作用,用于动态调节交联和功能化。有研究人员为了能够使用先进的添加剂制造技术进行加工并模拟细胞外基质的动态性质,细胞外基质的复杂微环境并不是从传统的水凝胶静态微环境复制而来,但生物打印的演变有助于适应水凝胶体系的动态调节和空间异质性[69-70]。ARONSSON 等[69]展示了一种模块化和3D打印的水凝胶,它结合了高弹性模量但可调的共价生物正交交联策略和特定的肽折叠介导的相互作用,用于动态调节交联和功能化。以多臂聚乙二醇为原料,通过菌株促进的炔叠氮环加成反应,实现了透明质酸水凝胶的共价交联。此外,重新设计的螺旋-环-螺旋多肽被连接到透明质酸骨架上,以实现交联密度、动力学以及水凝胶功能的特异性折叠调节,因此开发了一系列具有不同功能的互补多肽,并将其用作超分子调节细胞-水凝胶相互作用和控制酶介导的生物矿化过程的策略。模块化多肽系统能够动态修改3D打印结构的特性,展示了一条设计更复杂的生物材料用于四维生物打印的新途径。
刺激酶反应材料在药物输送和组织工程领域也引起了研究人员的极大关注。酶是具有高度特异性和选择性的分子,调节许多生物活性过程,包括蛋白质表达、细胞粘连和肌肉收缩。实际上大量的酶已经存在于体内,酶的刺激本质上是由生物环境本身和环境所触发的刺激,自然地由身体状态来调节。响应酶刺激材料之所以引起重视,是因其制成的植入物能够在完成它的作用后自然地降解。目前的研究已经证明4D打印水凝胶在负载多个活性酶的3D复杂形状方面的有效性,这些酶可以单独应用也可以多种酶共同引用[71]。基于此目的,有研究展示,印刷微管道系统并在其中嵌入蛋白酶降解肽用于支撑血管化结构,结果显示微管道内皮细胞由于酶降解而形成“血管新生芽”[34]。目前打印响应酶刺激材料的应用仅限于酶促降解性,但酶可能还会引起其他的反应,如超分子结构的改变、或表面性质的改变;该材料挑战的关键在于控制其反应的特异性,以防止与体内发生其他交叉反应而造成负面影响,尤其是应用于DNA序列或更长的肽链时,该特异性问题务必引起重视。
2.2 以功能转换为主的4D生物打印 随着生物医学的发展,4D生物打印的定义也被补充完善,起初它仅限于3D打印结构的几何变化,如今把3D打印细胞或组织结构的功能转化和自成熟也划分为4D生物打印的范畴[15,72-73]。自然组织和器官在结构上具有不同的特性和高度组织性,现在干细胞的生物打印和分化也可被视为4D生物打印的一种形式[74]。模拟天然细胞外基质仿生结构的建立可以引导和支持干细胞在生物打印后的生长和分化,从而有可能按需制造可植入的具有功能性的多层组织,如软骨、皮肤和骨骼肌,甚至是鼓膜[75-78]。
例如BETSCH等[51]提出了一种先进的生物打印方案,该方法在琼脂糖凝胶或Ⅰ型胶原水凝胶中利用一种基于响应磁场刺激的技术来排列胶原纤维,该策略利用磁场实现了微结构的实时矩阵重构,支架模拟了天然组织纤维的形态,以非常理想的方式指导细胞的增殖和分化行为以及打印组织的成熟。此外,MIAO等[79]使用一种特殊的光刻——立体光刻串联方法和智能大豆油环氧化丙烯酸酯生物材料制造了仿生的具有层次化的4D图形,这种先进的4D动态功能具有明显的心肌再生作用,打印品的表面形态有效调节了人骨髓间充质干细胞的心肌生成行为;这些打印支架具有分化和增殖人骨髓间充质干细胞的能力,可应用于骨软骨和神经组织工程,不仅在外界刺激下表现出动态的4D形状变化,而且提供了微妙的表面变换来调节干细胞的多向分化。还有人使用阶梯状策略构建了4D各向异性骨骼肌组织,并研究了不同分布策略对人骨髓间充质干细胞向骨骼肌分化的影响[80],结果发现肌源性基因的表达增强,证实这种仿生策略可以重建高度仿生的骨骼肌组织。综上所述,通过构建排列和分级的微结构,出现了更多潜在的生物打印策略来模拟细胞培养的微环境,调节种子细胞的分化,促进复杂结构的成熟,为4D生物打印提供了新的方向。
2.3 4D生物打印在组织工程中的应用 随着科技的不断发展,4D生物打印技术已经彻底颠覆了3D打印,生物材料也将迎来一场新的“材料革命”。创新的4D生物打印为制造复杂的多层组织结构提供了潜力,为组织工程和药物输送提供了许多优势,而且智能材料还可以作为细胞、药物、蛋白质载体,它们的可注射性和形状记忆性能使其可以应用于生物打印、微创外科手术和精密医学[81]。此文重点介绍4D生物打印技术在组织工程中的应用。
2.3.1 4D生物打印在神经组织的应用 MIAO等[82]应用4D打印制造神经引导导管以修复受损的神经,他们将1根临时扁平形状的4D打印智能形状记忆神经引导导管固定在受损神经下,然后在体温下恢复到原来的管状结构,将神经模型的两个残端完全包裹起来;这根4D打印的导管以一种多响应的石墨烯混合材料为基础,展示了人骨髓间充质干细胞的神经源性分化,表明了神经再生的潜力。此外,最近有另一项研究采用静电纺丝法制备了单轴排列的聚己内酯-聚癸二酸甘油酯和随机排列的甲基丙烯酸化透明质酸纤维双层支架,并将其应用于神经细胞的培养,纤维双层在水溶液中浸泡后立即形成管状结构,通过改变各层厚度、总双层厚度和介质反离子浓度等双层参数可以控制支架的管径;该支架在经过4周的实时降解后,呈现出高稳定性的自折叠涡卷状结构[83];该研究制作了可调谐的管状神经导管,为神经损伤的临床治疗铺平了道路。另外,ESWORTHY等[84]表示,水凝胶可能是制造复杂神经组织结构的最有效的材料类型,该材料模量可以很容易地通过改变刺激量或刺激条件来改变,而且水凝胶的模数对神经干细胞的增殖和分化有重要影响。
2.3.2 4D生物打印在心脏的应用 随着生物打印技术、响应刺激材料和水凝胶中心肌细胞活性的研究进展, 4D生物打印方法的进一步发展将有助于提高控制心肌细胞空间排列和内部结构的能力[85]。例如YUAN等[86]在模拟细胞自然环境的3D纤维蛋白水凝胶环境中培养心肌细胞,该细胞可以在电起搏条件下重建结构以产生收缩力。随着科技的发展,在刺激响应性水凝胶中培养心肌细胞,可能有助于形成更多三维天然结构。此外,还有研究应用简单而高效打印技术将二维设计转变为花状结构的4D动态形状变化,先进的4D动态功能可以提供与受损组织或器官的无缝集成,并首次证明了心脏再生的4D补片的概念。经证实,这种4D心脏补片具有明显的心肌再生作用,表明其在未来组织和器官再生应用中具有广阔的应用前景[79]。
2.3.3 4D生物打印在血管和气管中的应用 由于管状结构相对简陋,因此4D打印最早的形状变换便是转换为管状结构,但它却在人体中扮演着非常重要的角色,如血管,人体多种组织再生都需要血管化来营养再生的组织,不同组织和器官的成功血管化高度依赖于血管细胞和其他组织特异性细胞类型之间的相互作用[87]。因此,4D生物打印血管是打印其他再生组织的基础。
4D生物打印应用中最重要的方向之一就是制造血管,该技术也是解决组织血管化问题的一种很有前途的方法,例如具有高分辨率的中空管状结构和仿生毛细血管[88],它可以通过聚合物薄膜的自折叠来制造高分辨率的中空管状结构,这是4D打印独特的优势。例如,已经以高分辨率制造了4D空心自折叠管,其内径低至20 μm,这是其他3D打印技术难以达到的分辨率[68]。不同类型的细胞已经成功地被单层或多层自折叠微图案化水凝胶包裹,显示出在构建复杂的多细胞血管组织方面的巨大潜力。在另一项研究中,反应灵敏的自折叠或自折叠聚合物薄膜被用来在直径小于200 μm的管内对细胞进行温度控制的封装和释放[89]。
KIM等[90]基于数字光处理和光固化丝素蛋白水凝胶打印双层光固化丝素蛋白水凝胶气管支架,它在生理条件下可通过调节其内部或外部性质来控制其形状变化,使用有限元分析来探索复杂结构中可能发生的变化。最后,利用该4D生物打印系统制作模拟气管组织,结果表明4D生物打印系统可以在生物学上制作出组织仿生支架。
2.3.4 4D生物打印在骨组织工程中的应用 一些可注射的热响应性多糖水凝胶已被开发,可作为不同细胞、生长因子或无机复合材料(如羟基磷灰石、磷酸钙骨水泥和生物活性玻璃)的载体,用于骨组织工程[91]。羟丙基甲基纤维素、羟丁基壳聚糖、羟丙基瓜尔胶接枝聚(N-乙烯基己内酰胺)等改性生物材料在生理温度和操作温度之间具有较低的临界溶解温度,在体温下可转变为凝胶状态[10]。经典的热响应材料聚(N-异丙基丙烯酰胺)已经与透明质酸和壳聚糖结合在一起,制成了一种可注射的骨组织再生水凝胶[92-95]。类似地还有,壳聚糖可以通过与β-甘油磷酸盐结合形成温度响应性水凝胶,在凝胶形成过程中调节氢键和静电、疏水交联[96]。这种壳聚糖水凝胶可在体温下表现出良好的可注射性和快速凝胶性,通过加入不同的矿物成分,如磷酸三钙、纳米羟基磷灰石和生物活性玻璃,机械强度和承载能力都得到了提高[10]。这些可注射的杂化水凝胶具有良好的流变特性和体内自固化能力,是成骨细胞理想的载体,可以改善碱性磷酸酶活性和钙沉积,使其能够填充微小的不规则形状的缺损,并在体温下形成凝胶,在骨缺损的微创修复方面具有重要的潜力。
4D打印结构的形状变形特征可以实现个性化的骨缺损修复,因此更多的研究人员开始探索4D打印支架应用于骨缺损。例如MIAO等[97]制造了高度生物相容性的天然衍生智能聚合物四维打印新型仿生梯度组织支架,他们以最小的侵入性植入以实现无缝和动态集成时装配式结构固有的智能形状转换;为此合成了一系列具有良好生物相容性和可调变形效果的新型形状记忆聚合物,并在三维印刷牺牲模板存在下固化,然后将其溶解,在支架内产生可控和渐变的孔隙率。该支架与聚己内酯为对照,人骨髓间充质干细胞在新型智能聚合物上的黏附、增殖和分化显著增加。ARONSSON等[98]还合成了可生物降解的水基形状记忆聚氨酯,将其作为构建骨支架打印材料的主要成分,研究表示打印聚氨酯支架具有形状记忆性能、生物降解性和成骨效应,可作为骨组织工程的定制化骨替代物应用于微创外科手术。
还有研究人员设计了一种全新的支架——微孔聚乳酸支架[99],该支架的特点是在承重、抗压缩的框架结构中具有生物功能的多孔亚单位,开放式、大孔和微孔的内部结构通过扩散优化了向植入物内部区域提供氧气和营养的条件。
此外,研究人员的目光不仅仅放置于骨骼的修复,还开展了骨骼肌的生物打印,例如APSITE等[100]采用静电纺丝法制备了一层由各向异性甲基丙烯酸酯海藻酸纤维和取向聚己内酯纤维组成的双层支架,并诱导其自折叠包裹成肌细胞。双层垫子在含水缓冲液中经历形状转变,这一过程取决于它们的总厚度、每层的厚度和垫子的几何形状,正确选择这些参数可以制造包裹成肌细胞的涡卷状管。成肌细胞沿着各向异性聚己内酯纤维的轴线排列,并进一步分化成排列整齐的肌管,在电刺激下收缩,这种方法的意义在于制造中空的管状结构,这种结构可以进一步发展,以形成血管化的功能性肌肉。还有研究开发了一种电场辅助的3D细胞打印工艺,他们使用明胶甲基丙烯酰基负载细胞,在电刺激中诱导细胞排列,该研究组成功地研究了明胶的溶胀性能和折叠能力的形状变化机制,并使用一种4D概念化的明胶薄膜对甲基丙烯酰基超细纤维进行了捆绑,细胞负载的甲基丙烯酰基结构在电场刺激下可有效加速肌管形成和成熟[101]。目前关于这些材料的研究为4D生物打印应用于骨组织工程奠定了基础,在未来骨与软骨组织工程若想实现完全的缺损修复,与4D生物打印相结合是最有前途的道路[102]。
2.4 当前挑战 4D生物打印以“时间”作为3D生物打印的第4个维度,被众多研究人员认为是下一代组织工程的主流技术。近年来,随着刺激响应型的智能生物材料的发展和对组织再生认识的加深,4D生物打印技术在生物医学和临床领域中受到了极大的关注。尽管4D生物印刷可以设计和控制特定形状、大小、作用时间和作用部位的印刷构件,满足组织工程和临床应用的要求,但仍处于起步阶段,还面临多重挑战以待解决。
首先第一个挑战便是材料开发,目前将刺激响应性智能生物材料转化为优化的生物打印材料仍然具有挑战性[6]。根据力学性能的不同,用于4D生物打印的材料可分为软性材料和硬性材料两大类。软材料中最有前途的为形状记忆水凝胶,但在生物细胞组织的环境下可生物降解性、生物相容性和刺激响应性的水凝胶相当有限,而且力学性能较差,强烈膨胀的水凝胶非常柔软而且易碎。硬性材料其力学性能令人满意,是骨科植入物一种有前途的材料[18],但其生物相容性尚未测试。对材料的另一个要求是对外部刺激的反应,目前大多数智能材料只能对一种刺激做出反应,而人体内的细胞活动是复杂的,它受到多种刺激的调节,如自身调节、神经调节和体液调节。因此,应开发能够对多种生理信号刺激做出反应的智能材料。
其次,现有4D打印结构的形状转换过程仍然是简单的变形,不能满足临床应用中复杂的需要,在组织工程应用中,需要更专注于提高形状变换和打印分辨率的精确时空控制。对于打印二维结构再通过形状转换为三维结构类型的4D打印,已经存在数字建模计算机软件,它会以数字化计算模型来准确预测4D生物打印的演变。关于直接打印三维结构的4D打印,若想实现更高分辨率并非易事,因为它需要施加高剪切力,这会对细胞活力产生负面影响,有一种可能解决分辨率的方法是熔化静电纺丝技术和熔融电写技
术[103-104]。另一个与形状相关的问题是管状分支网络的制造,到目前为止,大多数是单根直管的打印报道,ZHU等[105]利用图灵不稳定性的模拟模型来研究肺分支结构的生物制造,填补了分支结构制造的空白。
此外,打印物反复折叠、展开会导致印刷结构的力学性能明显下降,而且印刷结构的机械强度通常无法承受高压,不能直接用作动脉血管,因为它们无法承受血压。因此,有必要开发具有健壮的形状转换特性的4D生物打印结构,特别是在需要重复响应的情况下。
在进一步应用于临床之前,还需相对温和的4D转化刺激机制或刺激措施,使4D打印技术对宿主环境更加友好,在不给细胞和组织带来负面影响的情况下,有效地刺激打印物形状转换。同时,还需要进一步的工作来以微创的方式精确控制施加到生物打印结构上的刺激物。
| [1] VIJAYAVENKATARAMAN S, YAN WC, LU WF, et al. 3D bioprinting of tissues and organs for regenerative medicine. Adv Drug Deliv Rev. 2018;132:296-332. [2] RODRÍGUEZ-SALVADOR M, RIO-BELVER RM, GARECHANA-ANACABE G. Scientometric and patentometric analyses to determine the knowledge landscape in innovative technologies: The case of 3D bioprinting. PLoS One. 2017;12(6):e0180375. [3] THAYER P, MARTINEZ H, GATENHOLM E. History and Trends of 3D Bioprinting. Methods Mol Biol. 2020;2140:3-18. [4] DIAS JR, RIBEIRO N, BAPTISTA-SILVA S, et al. In situ Enabling Approaches for Tissue Regeneration: Current Challenges and New Developments. Front Bioeng Biotechnol. 2020;8:85. [5] GHILAN A, CHIRIAC AP, NITA LE, et al. Trends in 3D Printing Processes for Biomedical Field: Opportunities and Challenges. J Polym Environ. 2020;28(5):1345-1367. [6] LI YC, ZHANG YS, AKPEKk A, et al. 4D bioprinting: the next-generation technology for biofabrication enabled by stimuli-responsive materials. Biofabrication. 2016;9(1):012001. [7] DORISHETTY P, DUTTA NK, CHOUDHURY NR. Bioprintable tough hydrogels for tissue engineering applications. Adv Colloid Interface Sci. 2020;281:102163. [8] RASTOGI P, KANDASUBRAMANIAN B. Review of alginate-based hydrogel bioprinting for application in tissue engineering. Biofabrication. 2019; 11(4):042001. [9] ANGELATS LOBO D, GINESTRA P. Cell Bioprinting: The 3D-Bioplotter™ Case. Materials (Basel). 2019;12(23):4005. [10] WAN Z, ZHANG P, LIU Y, et al. Four-dimensional bioprinting: Current developments and applications in bone tissue engineering. Acta Biomater. 2020;101:26-42. [11] YANG GH, YEO M, KOO YW, et al. 4D Bioprinting: Technological Advances in Biofabrication. Macromol Biosci. 2019;19(5):e1800441. [12] LUKIN I, MUSQUIZ S, EREZUMA I, et al. Can 4D bioprinting revolutionize drug development? Expert Opin Drug Discov. 2019;14(10):953-956. [13] ECHAVE MC, HHERNÁEZ-MOYA R, ITURRIAGA L, et al. Recent advances in gelatin-based therapeutics. Expert Opin Biol Ther. 2019;19(8): 773-779. [14] LI YE. Sustainable Biomass Materials for Biomedical Applications. ACS Biomater Sci Eng. 2019;5(5):2079-2092. [15] GAO B, YANG Q, ZHAO X, et al. 4D Bioprinting for Biomedical Applications. Trends Biotechnol. 2016;34(9):746-756. [16] TAMAY DG, DURSUN Usal T, ALAGOZ AS, et al. 3D and 4D Printing of Polymers for Tissue Engineering Applications. Front Bioeng Biotechnol. 2019;7:164. [17] YANG Q, GAO B, XU F. Recent Advances in 4D Bioprinting. Biotechnol J. 2020;15(1):e1900086. [18] SHAKIBANIA S, GHAZANFARI L, RAEEZADEH-SARMAZDEH M, et al. Medical application of biomimetic 4D printing. Drug Dev Ind Pharm. 2021:1-14. [19] CHANG J, HE J, MAO M, et al. Advanced Material Strategies for Next-Generation Additive Manufacturing. Materials (Basel). 2018;11(1):166. [20] CLEGG JR, WAGNER AM, SHIN SR, et al. Modular Fabrication of Intelligent Material-Tissue Interfaces for Bioinspired and Biomimetic Devices. Prog Mater Sci. 2019;106:100589. [21] HEINRICH MA, LIU W, JIMENEZ A, et al. 3D Bioprinting: from Benches to Translational Applications. Small. 2019;15(23):e1805510. [22] STANTON MM, SAMITIER J, SÁNCHEZ S. Bioprinting of 3D hydrogels. Lab Chip. 2015;15(15):3111-3115. [23] XUN KHOO Z, EE MEI TEOH J, LIU Y, et al. 3D printing of smart materials: A review on recent progresses in 4D printing. Virt Phys Prototyp. 2015; 10(3):103-122. [24] FRANZE K. The mechanical control of nervous system development. Development. 2013;140(15):3069-3077. [25] ASHAMMAKHI N, AHADIAN S, ZENGJIE F, et al. Advances and Future Perspectives in 4D Bioprinting. Biotechnol J. 2018;13(12):e1800148. [26] 白凯淞,陈光华,由长城,等.4D生物打印技术的研究进展[J].医学综述,2019,25(16):3189-3194. [27] ASHAMMAKHI N, KAARELA O. Stimuli-Responsive Biomaterials: Next Wave. J Craniofac Surg. 2017;28(7):1647-1648. [28] BAKARICH SE, GORKIN R 3rd, IN HET PANHUIS M, et al. 4D Printing with Mechanically Robust, Thermally Actuating Hydrogels. Macromol Rapid Commun. 2015;36(12):1211-1217. [29] ZHOU W, QIAO Z, NAZARZADEH ZARE E, et al. 4D-Printed Dynamic Materials in Biomedical Applications: Chemistry, Challenges, and Their Future Perspectives in the Clinical Sector. J Med Chem. 2020;63(15): 8003-8024. [30] IONOV L. 4D Biofabrication: Materials, Methods, and Applications. Adv Healthc Mater. 2018;7(17):e1800412. [31] BEDELL ML, NAVARA AM, DU Y, et al. Polymeric Systems for Bioprinting. Chem Rev. 2020;120(19):10744-10792. [32] SU JW , TAO X , DENG H , et al. 4D printing of a self-morphing polymer driven by a swellable guest medium. Soft Matter. 2018;14(5):765-772. [33] LÖWENBERG C, BALK M, WISCHKE C, et al. Shape-Memory Hydrogels: Evolution of Structural Principles To Enable Shape Switching of Hydrophilic Polymer Networks. Acc Chem Res. 2017;50(4):723-732. [34] LUI YS, SOW WT, TAN LP, et al. 4D printing and stimuli-responsive materials in biomedical aspects. Acta Biomater. 2019;92:19-36. [35] SENATOV FS, NIAZA KV, ZADOROZHNYY MY, et al. Mechanical properties and shape memory effect of 3D-printed PLA-based porous scaffolds. J Mech Behav Biomed Mater. 2016;57:139-148. [36] ZAREK M, MANSOUR N, SHAPIRA S, et al. 4D Printing of Shape Memory-Based Personalized Endoluminal Medical Devices. Macromol Rapid Commun. 2017;38(2). doi: 10.1002/marc.201600628. [37] LANZALACO S, ARMELIN E. Poly(N-isopropylacrylamide) and Copolymers: A Review on Recent Progresses in Biomedical Applications. Gels. 2017;3(4):36. [38] APSITE I, STOYCHEV G, ZHANG W, et al. Porous Stimuli-Responsive Self-Folding Electrospun Mats for 4D Biofabrication. Biomacromolecules. 2017;18(10):3178-3184. [39] LIU G, HE Y, LIU P, et al. Development of Bioimplants with 2D, 3D, and 4D Additive Manufacturing Materials. Engineering. 2020;6(11):1232-1243. [40] DE HAAN LT, VERJANS JM, BROER DJ, et al. Humidity-responsive liquid crystalline polymer actuators with an asymmetry in the molecular trigger that bend, fold, and curl. J Am Chem Soc. 2014;136(30): 10585-10588. [41] ZHANG K, GEISSLER A, STANDHARDT M, et al. Moisture-responsive films of cellulose stearoyl esters showing reversible shape transitions. Sci Rep. 2015;5:11011. [42] BORISOVA OV, BILLON L, RICHTER RP, et al. pH- and Electro-Responsive Properties of Poly(acrylic acid) and Poly(acrylic acid)-block-poly(acrylic acid-grad-styrene) Brushes Studied by Quartz Crystal Microbalance with Dissipation Monitoring. Langmuir. 2015;31(27):7684-7694. [43] AHADIAN S, DAVENPORT HUYER L, ESTILI M, et al. Moldable elastomeric polyester-carbon nanotube scaffolds for cardiac tissue engineering. Acta Biomater. 2017;52:81-91. [44] SONG E, CHOI JW. Conducting Polyaniline Nanowire and Its Applications in Chemiresistive Sensing. Nanomaterials (Basel). 2013;3(3):498-523. [45] SHIN SR, FARZAD R, TAMAYOL A, et al. A Bioactive Carbon Nanotube-Based Ink for Printing 2D and 3D Flexible Electronics. Adv Mater. 2016;28(17):3280-3289. [46] LI YC, ZHANG YS, AKPEK A, et al. 4D bioprinting: the next-generation technology for biofabrication enabled by stimuli-responsive materials. Biofabrication. 2016;9(1):012001. [47] SERVANT A, LEON V, JASIM D, et al. Graphene-based electroresponsive scaffolds as polymeric implants for on-demand drug delivery. Adv Healthc Mater. 2014;3(8):1334-1343. [48] CVETKOVIC C, RAMAN R, CHAN V, et al. Three-dimensionally printed biological machines powered by skeletal muscle. Proc Natl Acad Sci U S A. 2014;111(28):10125-10130. [49] KAI D, TAN MJ, PRABHAKARAN MP, et al. Biocompatible electrically conductive nanofibers from inorganic-organic shape memory polymers. Colloids Surf B Biointerfaces. 2016;148:557-565. [50] ZHANG L, ZUO X, LI S, et al. Synergistic therapy of magnetism-responsive hydrogel for soft tissue injuries. Bioact Mater. 2019;4:160-166. [51] BETSCH M, CRISTIAN C, LIN YY, et al. Incorporating 4D into Bioprinting: Real-Time Magnetically Directed Collagen Fiber Alignment for Generating Complex Multilayered Tissues. Adv Healthc Mater. 2018;7(21): e1800894. [52] SUN T, SHI Q, HUANG Q, et al. Magnetic alginate microfibers as scaffolding elements for the fabrication of microvascular-like structures. Acta Biomater. 2018;66:272-281. [53] TASOGLU S, YU CH, GUNGORDU HI, et al. Guided and magnetic self-assembly of tunable magnetoceptive gels. Nat Commun. 2014;5:4702. [54] BAGHERI A, ARANDIYAN H, BOYER C, et al. Lanthanide-Doped Upconversion Nanoparticles: Emerging Intelligent Light-Activated Drug Delivery Systems. Adv Sci (Weinh). 2016;3(7):1500437. [55] NASEER SM, MANBACHI A, SAMANDARI M, et al. Surface acoustic waves induced micropatterning of cells in gelatin methacryloyl (GelMA) hydrogels. Biofabrication. 2017;9(1):015020. [56] LUO Y, LIN X, CHEN B, et al. Cell-laden four-dimensional bioprinting using near-infrared-triggered shape-morphing alginate/polydopamine bioinks. Biofabrication. 2019;11(4):045019. [57] HUEBSCH N, KEARNEY CJ, ZHAO X, et al. Ultrasound-triggered disruption and self-healing of reversibly cross-linked hydrogels for drug delivery and enhanced chemotherapy. Proc Natl Acad Sci U S A. 2014;111(27):9762-9767. [58] NADGORNY M, AMELI A. Functional Polymers and Nanocomposites for 3D Printing of Smart Structures and Devices. ACS Appl Mater Interfaces. 2018;10(21):17489-17507. [59] CHEN C, ZHAO ML, ZHANG RK, et al. Collagen/heparin sulfate scaffolds fabricated by a 3D bioprinter improved mechanical properties and neurological function after spinal cord injury in rats. J Biomed Mater Res A. 2017;105(5):1324-1332. [60] CLARKE DE, PARMENTER CDJ, SCHERMAN OA. Tunable Pentapeptide Self-Assembled β-Sheet Hydrogels. Angew Chem Int Ed Engl. 2018; 57(26):7709-7713. [61] DU M, CHEN B, MENG Q, et al. 3D bioprinting of BMSC-laden methacrylamide gelatin scaffolds with CBD-BMP2-collagen microfibers. Biofabrication. 2015;7(4):044104. [62] LARUSH L, KANER I, FLUKSMAN A, et al. 3D printing of responsive hydrogels for drug-delivery systems. 3D Printing. 2017;1(4):219-229. [63] MIRANI B, PAGAN E, CURRIE B, et al. An Advanced Multifunctional Hydrogel-Based Dressing for Wound Monitoring and Drug Delivery. Adv Healthc Mater. 2017;6(19):10.1002/adhm.201700718. [64] HOCKADAY LA, KANG KH, COLANGELO NW, et al. Rapid 3D printing of anatomically accurate and mechanically heterogeneous aortic valve hydrogel scaffolds. Biofabrication. 2012;4(3):035005. [65] TABRIZ AG, HERMIDA MA, LESLIE NR, et al. Three-dimensional bioprinting of complex cell laden alginate hydrogel structures. Biofabrication. 2015;7(4):045012. [66] REN Z , ZHANG Y , LI Y , et al. Hydrogen bonded and ionically crosslinked high strength hydrogels exhibiting Ca2+-triggered shape memory properties and volume shrinkage for cell detachment. J Mater Chem B. 2015;3(30):6347-6354. [67] JUNGST T, SMOLAN W, SCHACHT K, et al. Strategies and Molecular Design Criteria for 3D Printable Hydrogels. Chem Rev. 2016;116(3): 1496-539. [68] KIRILLOVA A, MAXSON R, STOYCHEV G, et al. 4D Biofabrication Using Shape-Morphing Hydrogels. Adv Mater. 2017;29(46). doi: 10.1002/adma.201703443. [69] ARONSSON C, JURY M, NAEIMIPOUR S, et al. Dynamic peptide-folding mediated biofunctionalization and modulation of hydrogels for 4D bioprinting. Biofabrication. 2020;12(3):035031. [70] UNAGOLLA JM, JAYASURIYA AC. Hydrogel-based 3D bioprinting: A comprehensive review on cell-laden hydrogels, bioink formulations, and future perspectives. Appl Mater Today. 2020;18:100479. [71] DEVILLARD CD, MANDON CA, LAMBERT SA, et al. Bioinspired Multi-Activities 4D Printing Objects: A New Approach Toward Complex Tissue Engineering. Biotechnol J. 2018;13(12):e1800098. [72] CASTRO NJ, MEINERT C, LEVETT P, et al. HUTMACHER. Current developments in multifunctional smart materials for 3D/4D bioprinting. Current Opinion in Biomedical Engineering. 2017;2:67-75. [73] GLADMAN AS, MATSUMOTO EA, NUZZO RG, et al. Biomimetic 4D printing. Nat Mater. 2016;15(4):413-418. [74] IRVINE SA, VENKATRAMAN SS. Bioprinting and Differentiation of Stem Cells. Molecules. 2016;21(9):1188. [75] COSTANTINI M, TESTA S, MOZETIC P, et al. Microfluidic-enhanced 3D bioprinting of aligned myoblast-laden hydrogels leads to functionally organized myofibers in vitro and in vivo. Biomaterials. 2017;131:98-110. [76] KOFFLER J, ZHU W, QU X, et al. Biomimetic 3D-printed scaffolds for spinal cord injury repair. Nat Med. 2019;25(2):263-269. [77] MAHARAJAN N, CHO GW, JANG CH. Application of mesenchymal stem cell for tympanic membrane regeneration by tissue engineering approach. Int J Pediatr Otorhinolaryngol. 2020;133:109969. [78] DEVILLARD CD, MANDON CA, LAMBERT SA, et al. Bioinspired Multi-Activities 4D Printing Objects: A New Approach Toward Complex Tissue Engineering. Biotechnol J. 2018;13(12):e1800098. [79] MIAO S, CUI H, NOWICKI M, et al. Photolithographic-stereolithographic-tandem fabrication of 4D smart scaffolds for improved stem cell cardiomyogenic differentiation. Biofabrication. 2018;10(3):035007. [80] MIAO S, NOWICKI M, CUI H, et al. 4D anisotropic skeletal muscle tissue constructs fabricated by staircase effect strategy. Biofabrication. 2019;11(3):035030. [81] HUANG HJ, TSAI YL, LIN SH, et al. Smart polymers for cell therapy and precision medicine. J Biomed Sci. 2019;26(1):73. [82] MIAO S, CUI H, NOWICKI M, et al. Stereolithographic 4D Bioprinting of Multiresponsive Architectures for Neural Engineering. Adv Biosyst. 2018;2(9):1800101. [83] APSITE I, CONSTANTE G, DULLE M, et al. 4D Biofabrication of fibrous artificial nerve graft for neuron regeneration. Biofabrication. 2020;12(3):035027. [84] ESWORTHY TJ, MIAO S, LEE SJ, et al. Advanced 4D Bioprinting Technologies for Brain Tissue Modeling and Study. Int J Smart Nano Mater. 2019;10(3):177-204. [85] ONG CS, NAM L, ONG K, et al. 3D and 4D Bioprinting of the Myocardium: Current Approaches, Challenges, and Future Prospects. Biomed Res Int. 2018;2018:6497242. [86] YUAN YE K, SULLIVAN KE, BLACK LD. Encapsulation of cardiomyocytes in a fibrin hydrogel for cardiac tissue engineering. J Vis Exp. 2011;55:3251. [87] SHAH MOHAMMADI M, BUCHEN JT, PASQUINA PF, et al. Critical considerations for regeneration of vascularized composite tissues. Tissue Eng Part B Rev. 2020. [88] MIRI AK, KHALILPOUR A, CECEN B, et al. Multiscale bioprinting of vascularized models. Biomaterials. 2019;198:204-216. [89] STROGANOV V, AL-HUSSEIN M, SOMMER JU, et al. Reversible thermosensitive biodegradable polymeric actuators based on confined crystallization. Nano Lett. 2015;15(3):1786-1790. [90] KIM SH, SEO YB, YEON YK, et al. 4D-bioprinted silk hydrogels for tissue engineering. Biomaterials. 2020;260:120281. [91] GRAHAM S, MARINA PF, BLENCOWE A. Thermoresponsive polysaccharides and their thermoreversible physical hydrogel networks. Carbohydr Polym. 2019;207:143-159. [92] YANG J, VAN LITH R, BALER K, et al. A thermoresponsive biodegradable polymer with intrinsic antioxidant properties. Biomacromolecules. 2014;15(11):3942-3952. [93] CHEN JP, TSAI MJ, LIAO HT. Incorporation of biphasic calcium phosphate microparticles in injectable thermoresponsive hydrogel modulates bone cell proliferation and differentiation. Colloids Surf B Biointerfaces. 2013;110:120-129. [94] CHEN X, ZHAO Y, GENG S, et al. In vivo experimental study on bone regeneration in critical bone defects using PIB nanogels/boron-containing mesoporous bioactive glass composite scaffold. Int J Nanomedicine. 2015;10:839-846. [95] ZHAO C, ZENG Z, QAZVINI NT, et al. Thermoresponsive Citrate-Based Graphene Oxide Scaffold Enhances Bone Regeneration from BMP9-Stimulated Adipose-Derived Mesenchymal Stem Cells. ACS Biomater Sci Eng. 2018;4(8):2943-2955. [96] WU J, ZHENG K, HUANG X, et al. Thermally triggered injectable chitosan/silk fibroin/bioactive glass nanoparticle hydrogels for in-situ bone formation in rat calvarial bone defects. Acta Biomater. 2019;91: 60-71. [97] MIAO S, ZHU W, CASTRO NJ, et al. Four-Dimensional Printing Hierarchy Scaffolds with Highly Biocompatible Smart Polymers for Tissue Engineering Applications. Tissue Eng Part C Methods. 2016;22(10): 952-963. [98] ARONSSON C, JURY M, NAEIMIPOUR S, et al. Dynamic peptide-folding mediated biofunctionalization and modulation of hydrogels for 4D bioprinting. Biofabrication. 2020;12(3):035031. [99] SÖHLING N, NEIJHOFT J, NIENHAUS V, et al. 3D-Printing of Hierarchically Designed and Osteoconductive Bone Tissue Engineering Scaffolds. Materials (Basel). 2020;13(8):1836. [100] APSITE I, URIBE JM, POSADA AF, et al. 4D biofabrication of skeletal muscle microtissues. Biofabrication. 2019;12(1):015016. [101] YANG GH, KIM W, KIM J, et al. A skeleton muscle model using GelMA-based cell-aligned bioink processed with an electric-field assisted 3D/4D bioprinting. Theranostics. 2021;11(1):48-63. [102] QASIM M, CHAE DS, LEE NY. Advancements and frontiers in nano-based 3D and 4D scaffolds for bone and cartilage tissue engineering. Int J Nanomedicine. 2019;14:4333-4351. [103] YANG Z, SONG Z, NIE X, et al. A smart scaffold composed of three-dimensional printing and electrospinning techniques and its application in rat abdominal wall defects. Stem Cell Res Ther. 2020;11(1):533. [104] CONSTANTE G, APSITE I, ALKHAMIS H, et al. 4D Biofabrication Using a Combination of 3D Printing and Melt-Electrowriting of Shape-Morphing Polymers. ACS Appl Mater Interfaces. 2021;13(11):12767-12776. [105] ZHU X, YANG H. Turing Instability-Driven Biofabrication of Branching Tissue Structures: A Dynamic Simulation and Analysis Based on the Reaction⁻Diffusion Mechanism †. Micromachines (Basel). 2018;9(3):109. [106] KWOK JKS, LAU RWH, ZHAO ZR, et al. Multi-dimensional printing in thoracic surgery: current and future applications. J Thorac Dis. 2018; 10(Suppl 6):S756-S763. |
| [1] | 张立创, 徐 浩, 马迎辉, 熊梦婷, 韩海慧, 鲍嘉敏, 翟伟韬, 梁倩倩. 免疫调控淋巴回流功能治疗类风湿关节炎的机制及前景[J]. 中国组织工程研究, 2022, 26(9): 1459-1466. |
| [2] | 姚晓玲, 彭建城, 许岳荣, 杨志东, 张顺聪. 可变角度零切迹前路椎间融合内固定系统治疗脊髓型颈椎病:30个月随访[J]. 中国组织工程研究, 2022, 26(9): 1377-1382. |
| [3] | 朱 婵, 韩栩珂, 姚承佼, 周 倩, 张 强, 陈 秋. 人体唾液成分与骨质疏松/骨量低下[J]. 中国组织工程研究, 2022, 26(9): 1439-1444. |
| [4] | 金 涛, 刘 林, 朱晓燕, 史宇悰, 牛建雄, 张同同, 吴树金, 杨青山. 骨关节炎与线粒体异常[J]. 中国组织工程研究, 2022, 26(9): 1452-1458. |
| [5] | 王 景, 熊 山, 曹 金, 冯林伟, 王 信. 白细胞介素3在骨代谢中的作用及机制[J]. 中国组织工程研究, 2022, 26(8): 1260-1265. |
| [6] | 朱 婵, 韩栩珂, 姚承佼, 张 强, 刘 静, 邵 明. 针刺治疗帕金森病:动物实验显示的作用机制[J]. 中国组织工程研究, 2022, 26(8): 1272-1277. |
| [7] | 郭 嘉, 丁琼桦, 刘 泽, 吕思懿, 周泉程, 高玉花, 白春雨. 间充质干细胞来源外泌体的生物学特性及免疫调控作用[J]. 中国组织工程研究, 2022, 26(7): 1093-1101. |
| [8] | 吴玮玥, 郭晓东, 包崇云. 工程化外泌体在骨修复再生中的应用[J]. 中国组织工程研究, 2022, 26(7): 1102-1106. |
| [9] | 周洪琴, 吴丹丹, 杨 琨, 刘 琪. 传递特定miRNA的外泌体可调控成骨并促进成血管[J]. 中国组织工程研究, 2022, 26(7): 1107-1112. |
| [10] | 张璟琳, 冷 敏, 朱博恒, 汪 虹. 干细胞源外泌体促进糖尿病创面愈合的机制及应用[J]. 中国组织工程研究, 2022, 26(7): 1113-1118. |
| [11] | 黄晨玮, 费彦亢, 朱梦梅, 李鹏昊, 于 兵. 谷胱甘肽在干细胞“干性”及调控中的重要作用[J]. 中国组织工程研究, 2022, 26(7): 1119-1124. |
| [12] | 惠小珊, 白 京, 周思远, 王 阶, 张金生, 何庆勇, 孟培培. 中医药调控干细胞诱导分化的理论机制[J]. 中国组织工程研究, 2022, 26(7): 1125-1129. |
| [13] | 安维政, 何 萧, 任 帅, 刘建宇. 肌源干细胞在周围神经再生中的潜力[J]. 中国组织工程研究, 2022, 26(7): 1130-1136. |
| [14] | 范一鸣, 刘方煜, 张洪宇, 李 帅, 王岩松. 脊髓损伤后室管膜区内源性神经干细胞反应的系列问题[J]. 中国组织工程研究, 2022, 26(7): 1137-1142. |
| [15] | 宋玉鑫, 张同同, 牛建雄, 王增平, 文 杰, 张群立, 薛 文, 刘 林. 3D打印模型辅助与骨科机器人在脊柱畸形矫形过程中的精准置钉[J]. 中国组织工程研究, 2022, 26(6): 904-907. |
3D生物打印的一个主要缺点是它只考虑打印对象的初始状态,忽略了生物的生命和动态性[4-5],而人体组织通常会由外界刺激导致组织构象动态变化,例如心脏由副交感神经系统控制,并由引起心肌收缩的电刺激驱动,产生有节律的收缩来调节全身血液循环。这些动态的功能构象变化大多是由内在和外部刺激做出的反应,这是3D生物打印无法模仿的[6-8]。目前,3D生物打印正在向4D生物打印发展,这将是生物打印领域未来的主流[9]。
2014年麻省理工大学首次展示了4D打印技术[10],四维打印本质上是一种三维打印技术[11],其中包含的第四维被称为“时间”。这项技术需要具有随时间变化的多材料打印能力,或者需要可以从一种形状改变为另一种形状的定制材料系统,从而解决3D打印的局限性。自发明以来,4D打印迅速吸引了越来越多的关注,并发表了海量论文,这项技术为研究人员熟知,并在各个领域得到了广泛的应用,其中在生物医学领域中为组织工程和药物传递带来了无限的潜能[12-14]。
最近,4D打印已经扩展到将生物相容性材料甚至活细胞打印成复杂的结构,这被称为4D生物打印,见图1C,D。
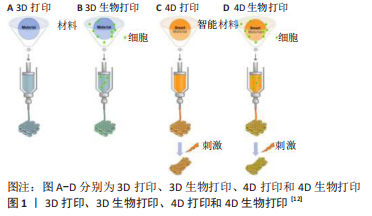 目前4D生物打印的定义仍然存在争议。4D生物打印不仅定义为能够对内部细胞力或外部刺激做出反应的加载细胞3D打印结构的产生,而且定义为3D打印结构中细胞或组织随时间的成熟和功能化,即打印结构的构型不变,而且对刺激的反应是可逆的[15-16],因此3D打印结构中材料的受控降解(在动态过程中完全消失)不应被视为4D打印[10,16]。此文认为4D生物打印是一个广义的概念,4D打印可以被称为4D生物打印,只要它满足后续条件之一:①打印物可用于生物组织工程;②打印材料具有生物相容性,可以移植到人体内;③打印制品中富含细胞[17]。
目前4D生物打印的定义仍然存在争议。4D生物打印不仅定义为能够对内部细胞力或外部刺激做出反应的加载细胞3D打印结构的产生,而且定义为3D打印结构中细胞或组织随时间的成熟和功能化,即打印结构的构型不变,而且对刺激的反应是可逆的[15-16],因此3D打印结构中材料的受控降解(在动态过程中完全消失)不应被视为4D打印[10,16]。此文认为4D生物打印是一个广义的概念,4D打印可以被称为4D生物打印,只要它满足后续条件之一:①打印物可用于生物组织工程;②打印材料具有生物相容性,可以移植到人体内;③打印制品中富含细胞[17]。从本质上讲,4D生物打印是一种新的趋势,它起源于3D生物打印,不同的是需要应用可刺激的智能材料打印[18-19],它的刺激可以包括水、pH值、温度、电、离子强度、光、磁场、压力、声波等等[20]。综上所述,4D生物打印可能会为3D生物打印开辟一条面向各种应用的新途径,随着智能生物材料和智能设计的发展,刺激3D结构转变的新策略即将出台,例如4D生物打印的智能生物机器人可以用于疾病诊断或可编程释放的药物输送。此外,智能组织结构可能通过这一概念实现,以更好地再生功能性组织和器官[21]。
4D生物打印具有较3D打印更加精细的结构以及动态适应性,从而带来了巨大的潜力,但4D生物打印技术尚未完全成熟,其概念和机制还未被研究人员充分了解。此文对CNKI、PubMed、SCIE 3个数据库发表的文章进行综述,将4D生物打印方式分为2类:第一类指以变型为主的四维生物打印,它通常以具有固有特性和功能的、可以响应外部刺激的智能材料为打印原料,根据预设定的刺激变形或重塑,如这类材料具有代表性的变形方式为自折叠;第二类指模仿天然组织和器官,打印出三维的微组织,通过细胞涂层或添加自体组织使其根据时间而成熟,最终形成预想的、完整的功能性组织结构[22-23]。此文重点介绍了4D生物打印技术在组织工程中的发展,并结合目前的研究和潜在的临床应用,介绍其在骨科中的应用;最后讨论4D生物打印发展所面临的瓶颈,并展望了这项革命性技术的未来方向和前景。
中国组织工程研究杂志出版内容重点:人工关节;骨植入物;脊柱;骨折;内固定;数字化骨科;组织工程
1.2 纳入与排除标准
纳入标准:根据检索式检索出的文章题目及摘要进行初步筛选,再对文献进行精读提炼出与此篇综述相关的4D生物打印应用于组织工程中应用的综述与论著,此外还对检索范围内文献的部分优质参考文献进行追踪收录,避免丢失高质量文章。
排除标准:文献内容与此篇综述主旨不符的、参考价值不高的、重复的文献;未应用于生物医学及组织工程学的4D打印,仅提及4D打印阐述3D打印的文章。
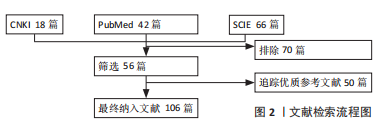
1.3 质量评估 通过该检索式检索出相关文献共126篇,根据文章内容初步筛选56篇,追踪收录优质参考文献50篇,最终收录105篇英文文献和1篇中文文献,见图2。
综上所述,4D生物打印提供了以可控的方式制造形状程序化和功能性结构的能力,目前已基本满足生物医学的要求,但若想实现临床应用还有很长的路要走。4D生物打印需要与智能材料同步发展,利用两者相结合才能打造出具有精确响应性的复杂结构,是组织工程领域的一次革新,全面推进了生物医学的概念,其最终目标是模拟人体内的生物功能。虽然已经有4D生物打印被应用于临床,但在它成为建立自然组织或器官的动态和分层结构的有效方式之前,还需要更多的研究来解决现有的挑战。可以肯定的是4D生物打印时代已经到来,它将给生物医学工程等领域带来革命性的影响。 中国组织工程研究杂志出版内容重点:人工关节;骨植入物;脊柱;骨折;内固定;数字化骨科;组织工程

文题释义:#br# 4D生物打印:4D打印本质上是一种三维打印技术,其中包含的第四维被称为“时间”,这项技术需要具有随时间变化的多材料打印能力,或者需要一种可以从一种形状改变为另一种形状的定制材料系统。对于 4D 打印一个较为全面的定义是:一个三维印刷结构被暴露于预定刺激下(如温度、水、光、pH值等),其功能、形状、性能可随时间发生变化。因此,与 3D 打印技术相比,4D 打印最大的突破在于其随时间变化的能力,而4D生物打印则是打印材料及打印产品具有生物相容性。#br# 智能材料:泛指将传感元件、驱动元件以及有关的信号处理和控制电路集成在材料结构中,通过机、热、光、化、电、磁等刺激和控制,使其不仅具有承受载荷的能力,而且具有识别、分析、处理及控制等多种功能,能进行自诊断、自适应、自学习及自修复。
通常应用形状记忆材料进行打印时,将打印模型放在在适当的条件下,使其处于亚稳态的临时形状。随后在施加外界刺激过程中逐步恢复,当施加足够的变换刺激时,临时变形的结构将变换成所需的形状,这种形状恢复特性对于治疗小骨缺损植入方式有良好的效果。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||